1Anatomical Sciences Research Center, Kashan University of Medical Sciences, Kashan, Iran
2Gametogenesis Research Center, Kashan University of Medical Sciences, Kashan, Iran
Article Publishing History
Received: 29/11/2016
Accepted After Revision: 13/11/2016
Cytochrome P450 (CYP) family enzymes have a crucial role in detoxification of environmental toxins which affected on male reproductive function. In this study we investigate the association of 3801T>C transition (rs4646903) in CYP1A1 gene with male infertility in an Iranian population which followed by a meta-analysis in Asian populations. In the case-control study, we collected blood samples from 100 idiopathic infertile and 100 healthy controls. 3801T>C genotyping was performed by PCR-RFLP method. In meta-analysis we selected eligible studies by searching in standard databases such as PubMed, Google Scholar, and etc. Our case-control study revealed that there is a significant association of CC genotype (OR= 3.00, 95%CI= 1.20-7.48, P= 0.018) and C allele (OR= 1.93, 95%CI= 1.09-3.39, P= 0.023) with male infertility. Also, meta-analysis revealed that there are significant associations between rs4646903 and male infertility in all of five genetic models with a true heterogeneity (PheterogeneityC transition can be considered as a biomarker for risk of male infertility within Asian populations.
Male Infertility; Cyp1a1; Genetic Polymorphism; Meta-Analysis
Nejati M, Karimian M. The Rs4646903 Gene Transition and Idiopathic Male Infertility: A Systematic Literature Review and Meta-Analysis. Biosc.Biotech.Res.Comm. 2016;9(4).
Nejati M, Karimian M. The Rs4646903 Gene Transition and Idiopathic Male Infertility: A Systematic Literature Review and Meta-Analysis. Biosc.Biotech.Res.Comm. 2016;9(4). Available from: https://bit.ly/2qzFfm0
INTRODUCTION
Idiopathic male infertility as a common health problem involved about 10-15% of men worldwide (De Kretser, 1997). Some causes including environmental factors, lifestyle and genetic factors may contribute in idiopathic male infertility (Nikzad et al., 2015; Karimian and Colagar 2016a). Despite the development of diagnostic tests for male infertility, especially in the molecular genetics field, many aspects are still unknown. Some evidences suggested that xenobiotic damage to genome, may help failure of spermatogenesis in about 30 percent of infertility in men (Schuppe et al., 2000).
Cytochrome P450 (CYP) family enzymes are key regulatory enzymes which catalyze the step 1 oxidation reactions of endogenous agents and xenobiotics (Gonzalez, 1992). CYP enzymes belonging to families 1–3 (among 14 families) in human are most essential for metabolic activation of numerous lipophilic compounds called as environmental or industrial pollutants (Nebert et al., 1996). The CYP1A1 (P4501A1) enzyme is considered in extra hepatic metabolism of agents including polycyclic aromatic hydrocarbons (PAHs) (Guengerich, 1992). Given to environmental compounds such as PAHs and their possible reproductive poisonousness through metabolic activation, polymorphisms in CYP1A1 gene may contribute in male infertility susceptibility (Crofts et al., 1994; Schuppe et al., 2000).
The CYP1A1 gene is located on chromosome 15 (15q24.1) and containing 7 exons. This gene is known as a polymorphic gene with several single nucleotide polymorphisms (SNPs) (Nebert et al., 1996). Among its polymorphisms, the 3801T>C (also known as, 2A, m1 or rs4646903) is the most common. This polymorphism results in T to C substitution at 3’ untranslated region (3’UTR) of the gene, and may change the expression of CYP1A1 (Luo et al., 2014). The association of CYP1A1 3801T>C polymorphism with male infertility has been studied only once (Salehi et al., 2012). Also, the results of genetic association studies in Asian populations are inconclusive. Therefor the aim of this study was to investigate the association of CYP1A1 3801T>C polymorphism with male infertility within an Iranian population which followed by a meta-analysis in Asian population.
MATERIALS AND METHODS
Subjects And Snp Genotyping
Subjects of this study were selected randomly from an Iranian population and included 100 idiopathic infertile and 100 aged-match healthy fertile men from individuals refer to IVF center (Kashan, Iran). The causes of male infertility of the subjects were remained unknown. The inclusion and exclusion criteria of fertile and infertile subjects were described in our previous study (Jamali et al., 2016). According to WHO 1999 criteria all of infertile subjects were classified as oligozospermia with less than 20 million sperm per ml in ejaculated semen. After obtaining informed consent form from all subjects, we collected 2ml blood samples on EDTA from all of them.
Genomic DNA was extracted from blood samples by salting out procedure. CYP1A1 3801T>C SNP genotyping was performed by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP). The protocol of SNP genotyping was described previously (Islam et al., 2013). Briefly, the CYP1A1 gene containing 3801T>C polymorphism was amplified by PCR method with the following specific primers: F: 5′-CAGTGAAGAGGTGTAGCCGCT-3′ and R: 5′-TAGGAGTCTTGTCTCATGCCT-3′. The 340-bp amplified fragments were treated by MspI restriction enzyme. After digestion procedure and separation by 1% agarose gel electrophoresis, the samples with 340-bp fragment were normal homozygote while the samples with 200-bp and 140-bp fragments were mutant homozygote. Moreover, the samples with 340-bp, 200-bp, and 140-bp fragments were heterozygote.
Meta-Analysis
We employed standard databases including PubMed, Google Scholar, and ScienceDirect, EMBase, and Medline till October 2016 for publication investigating the association CYP1A1 3801T>C polymorphism with male infertility. The keywords that used to search were as follow: male infertility, CYP1A1, 3801T>C, rs4646903, SNP, and polymorphism. The studies must met the following criteria: 1- case-control plan, 2- evaluating the association of CYP1A1 3801T>C transition with risk of male infertility, and 3- containing sufficient data to estimate odd ratios (ORs) with 95% confidence interval (95%CI) for all alleles and genotypes. The following data were excluded from eligible studies: country, author’s last name, year of publication, and genotypes frequency.
Statistical Analysis
In case-control study, the Hardy-Weinberg equilibrium (HWE) for genotypes frequencies was calculated by a Chi-square test. For evaluation of association of CYP1A1 3801T>C transition with male infertility, odd ratios and 95%CI were calculated by binary regression logistic. A P-value less than 0.05 was considered as statistically significant. These calculations were performed by SPSS ver.19 software.
In meta-analysis, the pooled OR was calculated in the five following genetic models: allelic (C vs. T), homozygous (CC vs. TT), and heterozygous (TC vs. TT), dominant (CC+TC vs. TT), and recessive (CC vs. TC+TT). Heterogeneity was measured by the chi square based Q test and I2 score, and a p-value less than 0.1 was considered as statistically significant. If there was a significant heterogeneity, ORs and 95%CI was calculated based on a random-effect model. Otherwise, a fixed-effects model was used. Egger’s test and Begg’s funnel plots were employed to calculate possible publication bias. The sensitivity analysis, relative effect of individual study on the pooled OR, was calculated by eliminating one study at a time. Statistical Meta-analyzes were performed by Comprehensive Meta Analysis software.
RESULTS
Distribution of Cyp1a1 3801t>C In Case-Control Study
The data from Hardy-Weinberg test revealed that the distribution of genotypes in case group was deviated from HWE (÷2= 6.39, P= 0.011), whereas this distribution in control group met HWE (÷2= 3.39, P= 0.066). The results of genetic association in case-control study are given in table 1. For example: CC genotype is associated with male infertility (OR= 3.00, 95%CI= 1.20-7.48, P= 0.018). Also, carriers of C allele were at a high risk for male infertility (OR= 1.93, 95%CI= 1.09-3.39, P= 0.023). In addition, C allele is significantly associated with male infertility (OR= 1.91, 95%CI= 1.23-2.97, P= 0.004).
| Table 1: Genotype and allele frequencies of CYP1A1 3801T>C in cases and controls. | ||||
| Genotype/Allele | Control (%) (n= 100) | Cases (%) (n= 100) | OR (95% CI) | P-value |
| TT | 64 (64%) | 48 (48%) | – | – |
| TC | 28 (28%) | 34 (34%) | 1.62 (0.87-3.02) | 0.131 |
| CC | 8 (8%) | 18 (18%) | 3.00 (1.20-7.48) | 0.018 |
| TC+CC | 36 (36%) | 52 (52%) | 1.93 (1.09-3.39) | 0.023 |
| T | 156 (78%) | 130 (65%) | – | – |
| C | 44 (22%) | 70 (35%) | 1.91 (1.23-2.97) | 0.004 |
| OR, Odds Ratio; CI, Confidence Interval. The significant differences are bolded | ||||
Meta-Analysis
The initial search of database and screening of relevant articles, 5 eligible studies were included in meta-analysis (Lu et al. 2008; Vani et al. 2009; Chen et al. 2010; Salehi et al. 2011; Peng et al. 2012). Moreover, the data from current study was added to the meta-analysis. The characteristics of included studies in meta-analysis are detailed in table 2. These studies included:3, 2, and 1 papers involved Chinese, Iranian, and Indian populations, respectively. This meta-analysis involved a total of 957 cases and 1098 controls. The results of genetic association in meta-analysis revealed that there is a significant associations between CYP1A1 3801T>C polymorphism and male infertility in all five genetic models (C vs. T: OR= 1.53, 95%CI= 1.16-2.02, P= 0.003; CC vs. TT: OR= 2.43, 95%CI= 1.33-4.42, P= 0.004; TC vs. TT: OR= 1.36, 95%CI: 1.13-1.64, P= 0.001; CC+TC vs. TT: OR= 1.57, 95%CI= 1.17-2.11, P= 0.003; CC vs. TC+TT: OR= 2.02, 95%CI= 1.21-3.37, P= 0.007) (Table 3 and Figure 1). The heterogeneity analysis revealed that there are true heterogeneities for association of 3801T>C with male infertility risk in C vs. T (Pheterogeneity< 0.001, I2= 77%), CC vs. TT (Pheterogeneity= 0.003, I2= 73%), CC+TC vs. TT (Pheterogeneity= 0.021, I2= 62%), CC vs. TC+TT (Pheterogeneity= 0.010, I2= 67%) models within Asian populations (Table 3). The data from publication bias test revealed that there are publication biases in CC vs. TT (PEgger= 0.018) and CC vs. TC+TT (PEgger= 0.017) genetic models (Figure 2).
| Table 2: Characteristics of included studies in meta-analysis | |||||||||||
| Country | Genotype frequencies | Allele frequencies | Reference | ||||||||
| Control | Case | Control | Case | ||||||||
| TT | TC | CC | TT | TC | CC | T | C | T | C | ||
| China | 95 | 104 | 27 | 69 | 96 | 27 | 294 | 158 | 234 | 150 | Lu et al. 2008 |
| India | 146 | 80 | 4 | 108 | 80 | 18 | 372 | 88 | 296 | 116 | Vani et al. 2009 |
| China | 88 | 45 | 7 | 35 | 47 | 23 | 221 | 59 | 117 | 93 | Chen et al. 2010 |
| Iran | 85 | 91 | 24 | 58 | 72 | 20 | 261 | 139 | 188 | 112 | Salehi et al. 2012 |
| China | 78 | 94 | 30 | 72 | 93 | 39 | 250 | 154 | 237 | 171 | Peng et al. 2012 |
| Iran | 64 | 28 | 8 | 48 | 34 | 18 | 156 | 44 | 130 | 70 | Current study |
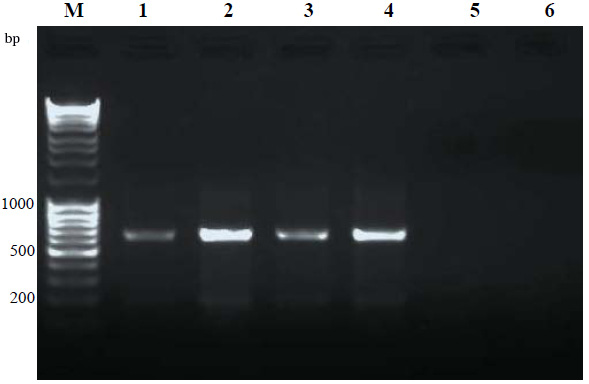 Figure 1: Forest plot for the association of CYP1A1 3801T>C polymorphism and male infertility. Findings of quantitative data synthesis under TC vs. TT model (A) and CC+TC vs. TT model (B) Figure 1: Forest plot for the association of CYP1A1 3801T>C polymorphism and male infertility. Findings of quantitative data synthesis under TC vs. TT model (A) and CC+TC vs. TT model (B) |
 Figure 2: Funnel plot for the association of CYP1A1 3801T>C polymorphism and male infertility. Possible publication bias under TC vs. TT model (A) and CC+TC vs. TT model (B) Figure 2: Funnel plot for the association of CYP1A1 3801T>C polymorphism and male infertility. Possible publication bias under TC vs. TT model (A) and CC+TC vs. TT model (B) |
| Table 3: Results of meta-analysis for association of 3801T>C with male infertility risk | ||||||||
| Genetic model | Analysis model | OR (95%CI) | P-value | tau2 | Q(df=5) | Ph | I2 | Pe |
| C vs. T (Allelic model) | Random effect | 1.53 (1.16-2.02) | 0.003 | 0.092 | 21.56 | < 0.001 | 77% | 0.057 |
| CC vs. TT (Homozygous model) | Random effect | 2.43 (1.33-4.42) | 0.004 | 0.40 | 18.35 | 0.003 | 73% | 0.018 |
| TC vs. TT (Heterozygous model) | Fixed effect | 1.36 (1.13-1.64) | 0.001 | – | 7.24 | 0.203 | 31% | 0.141 |
| CC+TC vs. TT (Dominant model) | Random effect | 1.57 (1.17-2.11) | 0.003 | 0.083 | 13.25 | 0.021 | 62% | 0.118 |
| CC vs. TC+TT (Recessive model) | Random effect | 2.02 (1.21-3.37) | 0.007 | 0.263 | 15.06 | 0.010 | 67% | 0.017 |
| OR, odds ratio; CI, confidence interval; Ph, P-values for heterogeneity from Q test; Pe: PEgger (p< 0.05) was considered as a significant difference. | ||||||||
DISCUSSION
In this study we investigated the association of CYP1A1 3801T>C polymorphism with male infertility in an Iranian population and a meta-analysis in Asian populations. Our case-control study indicated a significant association between 3801CC genotype and 3801C allele and male infertility in study population. Moreover, results of meta-analysis revealed that there are significant association between CYP1A1 3801T>C and male infertility in all of five genetic models. But, heterogeneity analysis demonstrated a true heterogeneity in four genetic models. The different results from various studies may be due to differences in environmental conditions and ethnicities. Also, a publication bias was found in TC vs. TT and CC vs. TC+TT genetic models. This publication bias may arise from small sample sizes and more chance of positive outputs for publications.
Numerous studies suggested a time-dependent reduction of quality of human sperm (Adamopoulos et al., 1996; Van Waeleghem et al., 1996). The causes of probable alterations in quality of human semen and the cumulative rate of male infertility were not fully understood. Among the several suggestions in this field, the possible roles of environmental exposures and their effects on functions and development of male reproductive system less regarded. (Sharpe and Skakkebaek, 1993). Specifically chemical compounds disrupting the endocrine system such as polychlorinated biphenyls (PCBs), pesticides, and etc. have been debated as possible risks of fertility (Sonnenschein and Soto, 1998). Since the xenobiotics including chlorinated hydrocarbons are indissoluble, they could accumulate in ecosystem and reproductive system of males affected by them. (Wagner et al., 1990). Therefore the protective procedures in human body were amplified. In order to prevent the accumulation of xenobiotic materials in human body, a wide range of enzymes with variable catalytic activity are involved in the process of detoxification (Nebert et al., 1996). The cytochrome P450 (CYP) superfamily are important enzymes in phase I oxidation/ reactions for metabolizing both endogenous substrates and xenobiotic agents (Nelson et al., 1996). The CYP familiy enzymes are member of 1-3 families (from 14 cytochrome families), that are very important for catabolism of lipophilic chemicals (Gonzalez, 1992). Among these, CYP1A1 enzyme metabolizes extra-hepatic catabolism of heterocyclic amines and polycyclic aromatic hydrocarbons (PAHs) (Guengerich, 1992). Given to the complex procedure of spermatogenesis (Weinbauer & Wessels, 1999), xenobiotics accumulation in different sections of the testicular tissues may be damaging for the process. In addition, immune activation of cells by reactive metabolites can lead to testicular immunopathology. The capacity of testicular tissue for metabolizing of xenobiotics was investigated in experimental models. For instance Leydig cells in rats are able to metabolize PAHs (Georgellis and RydstroÈm, 1987). Also, in certain strains of rats, the expression of CYP enzymes in the testes altered at caloric restriction (Seng et al., 1996).
Therefore CYP1A1 has a protective role in male reproductive function any changes in the structure and expression of CYP1A1 gene could alter the risk of male infertility. SNPs could alter the functions and expression of genes depending on their locations on the genes (Karimian and Colagar 2016b; Karimian et al., 2015). Therefore CYP1A1 3801T>C gene transition which located at 3’UTR of CYP1A1 may alter the gene expression. Our previous studies revealed that bioinformatics tools are suitable for evaluation of the effects of SNPs on several molecular aspects (Raygan et al., 2016; Soleimani et al., 2016). Then we suggest that the further studies were focused on effects of 3801T>C on molecular aspects of CYP1A1 by in silico analysis.
Finally there are some limitation in this study that should be declared. The gene-gene and gene-environment interactions were not considered in case-control study, since these issue may moderate the effects of 3801T>C CYP1A1 on male infertility. Moreover, original data of included studies in meta-analysis such as biochemical characteristics and BMI were not available, therefor we could not able to justify the role of 3801T>C CYP1A1 in male infertility by mentioned parameters. Moreover, the number of studies which included in meta-analysis was very low, therefore more studies with larger sample size are needed to gain more precise data.
REFERENCES
Adamopoulos, D. A., Pappa, A., Nicopoulou, S., Andreou, E., Karamertzanis, M., Michopoulos, J. & Simou, M. (1996). Andrology: Seminal volume and total sperm number trends in men attending subfertllity clinics in the Greater Athens area during the period 1977–1993. Human Reproduction, 11(9), 1936-1941.
Chen, W., Kang, X., Huang, Z., Wei, Y., Pan, Y. (2010). CYP1A1 gene polymorphism in oligozoospermic infertile patients of Zhuang population in Guangxi area. Guangdong medical journal, 13, 1669–1671.
De Kretser, D.M. (1997) Male infertility. The Lancet, 349(9054), 787-790.
Georgellis, A., & Rydström, J. (1989). Cell-specific metabolic activation of 7, 12-dimethylbenz [a] anthracene in rat testis. Chemico-biological interactions, 72(1-2), 65-78.
Gonzalez, F. J. (1992). Human cytochromes P450: problems and prospects. Trends in pharmacological sciences, 13, 346-352.
Guengerich, F. P. (1992). Metabolic activation of carcinogens. Pharmacology & therapeutics, 54(1), 17-61.
Islam, M. S., Ahmed, M. U., Sayeed, M. S. B., Al Maruf, A., Mostofa, A. G. M., Hussain, S. M. A. & Hasnat, A. (2013). Lung cancer risk in relation to nicotinic acetylcholine receptor, CYP2A6 and CYP1A1 genotypes in the Bangladeshi population. Clinica chimica acta, 416, 11-19.
Jamali, S., Karimian, M., Nikzad, H., & Aftabi, Y. (2016). The c.− 190 C> A transversion in promoter region of protamine1 gene as a genetic risk factor for idiopathic oligozoospermia. Molecular biology reports, 43(8), 795-802.
Karimian, M., & Colagar, A. H. (2016a). Association of C677T transition of the human methylenetetrahydrofolate reductase (MTHFR) gene with male infertility. Reproduction, fertility and development, 28(6), 785-794.
Karimian, M., & Colagar, A. H. (2016b). Methionine synthase A2756G transition might be a risk factor for male infertility: Evidences from seven case-control studies. Molecular and cellular endocrinology, 425, 1-10.
Karimian, M., Nikzad, H., Azami-Tameh, A., Taherian, A., Darvishi, F. Z. & Haghighatnia, M. J. (2015). SPO11-C631T gene polymorphism: association with male infertility and an in silico-analysis. Journal of family & reproductive health, 9(4), 155.
Lu, N., Wu, B., Xia, Y., Wang, W., Gu, A., Liang, J. & Zhang, Z. (2008). Polymorphisms in CYP1A1 gene are associated with male infertility in a Chinese population. International journal of andrology, 31(5), 527-533.
Luo, H., Li, H., Yao, N., Hu, L., & He, T. (2014). Association between 3801T> C polymorphism of CYP1A1 and idiopathic male infertility risk: a systematic review and meta-analysis. PloS one, 9(1), e86649.
Nebert, D. W., Mckinnon, R. A., & Puga, A. (1996). Human drug-metabolizing enzyme polymorphisms: effects on risk of toxicity and cancer. DNA and cell biology, 15(4), 273-280.
Nelson, D. R., Koymans, L., Kamataki, T., Stegeman, J. J., Feyereisen, R., Waxman, D. J. & Gunsalus, I. C. (1996). P450 superfamily: update on new sequences, gene mapping, accession numbers and nomenclature. Pharmacogenetics and Genomics, 6(1), 1-42.
Nikzad, H., Karimian, M., Sareban, K., Khoshsokhan, M. & Colagar, A. H. (2015). MTHFR-Ala222Val and male infertility: a study in Iranian men, an updated meta-analysis and an in silico-analysis. Reproductive biomedicine online, 31(5), 668-680.
Peng, L., Wang, G., Jiao, H., Li, Y., Dang, J., (2012). CYP1A1 gene (rs4646903) polymorphism in infertile patients in Ningxia area. Journal of Ningxia Medical University, 03, 208–210.
Raygan, F., Karimian, M., Rezaeian, A., Bahmani, B., & Behjati, M. (2016). Angiotensinogen-M235T as a risk factor for myocardial infarction in Asian populations: a genetic association study and a bioinformatics approach. Croatian Medical Journal, 57(4), 351.
Salehi, Z., Gholizadeh, L., Vaziri, H., & Madani, A. H. (2012). Analysis of GSTM1, GSTT1, and CYP1A1 in idiopathic male infertility. Reproductive sciences, 19(1), 81-85.
Schuppe, H. C., Wieneke, P., Donat, S., Fritsche, E., Köhn, F. M., & Abel, J. (2000). Xenobiotic metabolism, genetic polymorphisms and male infertility. Andrologia, 32(4‐5), 255-262.
Schuppe, H. C., Wieneke, P., Donat, S., Fritsche, E., Köhn, F. M., & Abel, J. (2000). Xenobiotic metabolism, genetic polymorphisms and male infertility. Andrologia, 32(4‐5), 255-262.
Seng, J. E., Gandy, J., Turturro, A., Lipman, R., Bronson, R. T., Johnson, W. & Leakey, J. E. (1996). Effects of caloric restriction on expression of testicular cytochrome P450 enzymes associated with the metabolic activation of carcinogens. Archives of biochemistry and biophysics, 335(1), 42-52.
Sharpe, R. M., & Skakkebaek, N. E. (1993). Are oestrogens involved in falling sperm counts and disorders of the male reproductive tract? The Lancet, 341(8857), 1392-1396.
Soleimani, Z., Kheirkhah, D., Sharif, M. R., Sharif, A., Karimian, M., & Aftabi, Y. Association of CCND1 Gene c. 870G> A Polymorphism with Breast Cancer Risk: A Case-Control Study and a Meta-Analysis. Pathology & Oncology Research, 1-11. Doi: 10.1007/s12253-016-0165-3.
Sonnenschein, C., & Soto, A. M. (1998). An updated review of environmental estrogen and androgen mimics and antagonists. The Journal of steroid biochemistry and molecular biology, 65(1), 143-150.
Toffoli, G., & De Mattia, E. (2008). Pharmacogenetic relevance of MTHFR polymorphisms. Pharmacogenomics, 9(9), 1195-1206.
Van Waeleghem, K., De Clercq, N., Vermeulen, L., Schoonjans, F. R. A. N. K. & Comhaire, F. (1996). Deterioration of sperm quality in young healthy Belgian men. Human Reproduction, 11(2), 325-329.
Vani, G. T., Mukesh, N., Prasad, B. S., Devi, P. R., Prasad, M. H., Rani, P. U. & Reddy, P. P. (2009). Association of CYP1A1 2A polymorphism with male infertility in Indian population. Clinica chimica acta, 410(1), 43-47.
Wagner, U., Schlebusch, H., Van der Ven, H., Van der Ven, K., Diedrich, K. & Krebs, D. (1990). Accumulation of pollutants in the genital tract of sterility patients. Journal of clinical chemistry and clinical biochemistry. Zeitschrift fur klinische Chemie und klinische Biochemie, 28(10), 683-688.
Weinbauer, G. F. & Wessels, J. (1999). Paracrine control of spermatogenesis. Andrologia, 31(5), 249-262.


