1Department of Biotechnology Gulbarga University Kalaburagi
2Luqman College of Pharmacy, Gulbarga, Karnataka 585101, India
Corresponding author Email: ckelmani@gmail.com
Article Publishing History
Received: 04/01/2019
Accepted After Revision: 20/03/2019
Salmonella typhi specifi c bacteriophage i.e. GRCST exhibited potential bacteriolytic activity against n=4, ESBL producing S. typhi isolates in vitro. The GRCST possesses an icosahedral head with 50 nm size and contractile tail belongs to Myoviridae Vi01-like family. The experimental outcome of in vivo studies in BALB/c mice induced with S. typhi bacteraemia treated with 1.5×107 PFU GRCST showed 100% survival with zero causality was recorded. On contrary, only 67% and 83% survival rate was observed in the group of mice which received standard antibiotic ciprofl oxacin. The IgG and IgM titres of anti-phage GRCST antibodies were detected, with increased 4100 fold, 600 fold respectively. This result demonstrates that the antibodies elicited by GRCST are non-neutralizing.
Balb/C Mice, Esbl, ϕGrcst, In Vivo, Igg, Igm, Salmonella Typhi
Narasanna R, Chavadi M, Ahmed L, Sannauallah S, Chandrakanth K. Experimental protection of ESBL producing Salmonella typhi bacteremic induced mice model by ϕ GRCST; a therapeutic approach. Biosc.Biotech.Res.Comm. 2019;12(1).
Narasanna R, Chavadi M, Ahmed L, Sannauallah S, Chandrakanth K. Experimental protection of ESBL producing Salmonella typhi bacteremic induced mice model by ϕ GRCST; a therapeutic approach. Biosc.Biotech.Res.Comm. 2019;12(1). Available from: https://bit.ly/2HSXuHZ
Copyright © Narasanna et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
Introduction
The Viruses or Bacteriophage which infects bacteria were discovered in 1915 by Frederick Twort. The era of “bacteriophage” was begun with the seminal publication by Felix D’Herelle in 1917, demonstrating “un bacteriophage obligatoire” means “a bacteriophage mandatory”. Total of 13 microbiologists worked together integrate the applications of phages in the fi eld of medicine. Till date, over 6000 various bacteriophages were discovered, which includes 6196 bacterial and 88 archaeal viruses, identifi ed morphologically and classifi cation was accomplished (Ackermann et al., 2012). Morphologically, the majority of these phages consisting contractile tail with polyhedral, fi lamentous or pleomorphic head types. The classifi cation of phages so far has been achieved signifi cantly based on their genetic content (DNA vs. RNA), their morphology, and their limited host range i.e. specifi c host (Deghorain et al., 2012).
Increasing case studies of antimicrobial resistance and relented discoveries and development have propelled the researchers to search for an alternative therapy has led to the revitalization of bacteriophage (phage) studies in the Western world. Recently, WHO Listed, a global priority pathogens consist of 12 bacterial species categorized into critical, high and medium priority based on their level of resistance and availability of therapeutics pathogens (Tacconelli et al., 2018). While this is a contentious fi gure (De Kraker et al., 2016), it nonetheless highlights the serious problem we face regarding therapeutic options for multi-drug resistant (MDR) bacterial infections (Bassetti et al., 2017).
Phage therapy; obligatory lytic phages were employed to kill the specifi c bacterial hosts, without causing damage to human host cells and nullifying the impact on commensal bacteria. Rapid evolving of phage therapy has resulted in resolving life-threatening clinical cases. Currently, antibiotic alternative facing the regulations and policies surrounding clinical use and application beyond compassionate cases (Furfaro et al., 2018).
In the year 1919, phage therapy was the fi rst time practiced in human beings at the hospital des Enfants Malades in Paris, France, when D’Herelle successfully treated many children’s who were suffering from severe dysentery by using phages as a therapeutics, he has isolated these infective phages in Pasteur Institute, from stools of soldiers (Sulakvelidze et al., 2005).
Salmonella bacteria are often health hazards, associated with a million food borne illnesses per year in the US. Bacteriophages have been specifi cally used to identify Salmonella species and may also be useful in therapy and prophylaxis of Salmonella infections. The phage FelixO1 was fi rst used in 1943 by Felix and Callow as part of a “phage-typing” system for the identifi – cation Salmonella typhi (Anderson et al., 1953).
First commercial phage produced by Theodore Mazure, in which contains, cocktails—Bacté-Coli-Phage, Bacté-Intesti-Phage, Bacté-Dysentérie-Phage, Bacté- Pyo-Phage and Bacté-Rhino-Phage (Abedon et al., 2011). Henri de Montclos, chief clinical microbiologist at Pasteur Institute of Lyon, for 10 years, his research team has produced fi rst anti-staphylococcal vaccines and therapeutic phages in the year early 1990s. The bacteriophages were administered to treat the “acute colitis” due to infections of Shigella or Salmonella in Georgia (Mikeladze et al., 1936). Potential administration of Bacté- Pyo-Phage and Bacté-Intesti-Phage, undiluted resulted in drastic reduction of mortality rate from 85% to 20%.
Therapeutic application of bacteriophage started in Eastern Europe and the former Soviet Union, currently it’s been applied widespread as a part of health care systems. However, the effi ciency of phage therapy is investigated according to rigorous scientifi c standards and presented a list of key criteria for consideration and reporting of phage therapy studies (Kutter et al., 2010; Abedon, 2017; Villarroel et al., 2017). Information critical to the success of clinical trials includes the adequate characterization and selection of phages as well as of the subjects (humans) and the target bacteria., in addition to that, the choice of appropriate disease targets for phage therapy (Harper, 2018). On the other hand, it may be that broad-host range phages are more common than is currently believed, due in part to biases in phage isolation methods (De Jonge et al., 2018); this disparity deserves much further research.
However, recent research and its outcomes suggest that bacteriophage therapy is the appropriate treatment to cure Salmonella associated infections. Majorly typhoid fever was treated with bacteriophages by Tsouloukidze et al., 1936 (Tsulukidze et al., 1936); who successfully treated twenty patients suffering from peritonitis due to intestinal perforations in typhoid fever (Abedon et al., 2011). There are some published reports of successful treatment against Salmonella-associated disease with prophylactic phage therapy in treating Russian soldiers suffering from dysentery during and after World War II (Kutter et al., 2009). The reports suggest that, it has been already practiced in broiler chickens. The bacteriophages were able to reduce S. enteritidis counts on chicken skin at refrigeration temperature and short contact time (Atterbury et al., 2007). In addition, the decrease of S. enteritidis count on artifi cially-contaminated chicken skin after phage treatment corresponded to the reduction achieved by chemical agents commonly used in the poultry industry. A signifi cant breakthrough is, bacteriophages were used as biocontrol agents in Pigs to control the infection, according to the study conducted by Albino et al., 2014; and the outcome of the study was a signifi cant reduction in the colonization of Salmonella in pigs administered with pool of bacteriophages (Albino et al., 2014).
Materials and Methods
Phage Isolation, Production, and Titration
ESBL resistant strain S. typhi BST 51 was used to specifi chost isolate bacteriophage from raw sewage samples. The sewage sample was collected from various places of Kalaburagi. The sample was fi ltered with sewage was fi ltered with fi lter paper, and subsequently 40 ml of sewage was added to the 10 ml of 10X LB broth, inoculated with S. typhi BST 51 strain and incubated for 18-24 hr. The media was centrifuged at 10000 rpm for 10 min and the supernatant was collected and subsequently fi ltered using a 0.2 μm syringe fi lter (Melo et al., 2014b).
Screening of GRCST (G: Gulbarga, R: Rahul, C: Chandrakanth Kelmani, S: Salmonella, T: Typhi) bacteriophage was accomplished by plaque assay method i.e. agar overlay technique. The LB agar plates were prepared, 0.1 ml of supernatant was serially diluted in 0.9 ml of LB media from 101 -1010 in 1.5 ml eppendorf tubes 0.5 ml of test culture S. typhi BST 51 with 0.1 O. D was equally distributed to another set of 10 eppendorf and labelled for each dilution tubes subsequently 0.1 ml serially diluted fi ltrate was added to the respective tubes containing 0.5 ml of S. typhi BST51 bacterial culture labelled with respective dilution, incubated for 10-15 min. Each labelled tube was taken and uniformly mixed with LB soft agar containing 0.6% agar in a molten state at a temperature of 40 0C- 45 0C. Thereafter soft agar was overlaid on LB hard agar plates containing 2% agar and kept for incubation at 18-24 hr. Plaque formation on agar plates indicates bacteriophage positive (Mazzocco et al., 2009; Kropinski et al., 2009).
Phage purifi cation and storage
An isolated colony of S. typhi BST 51 strain was inoculated into the LB broth, the culture was allowed to attain an OD of 0.1, and then infected with ϕGRCST of 2X107 PFU, the culture was co-cultivated for 18 hr at 37 0C in a shaking incubator (240 rpm). Polyethylene glycol-8000 (PEG) or NaCl was added to the lysate to a fi nal concentration of 20% or 0.5 M respectively and incubated at 1 hr at 4 0C. After centrifugation at 10,000 rpm (16 min at 4 0C) in a sorvall RC5B centrifuge, polyethylene glycol (PEG-8000) was added to the supernatant to a fi nal concentration of 10%. The lysate was incubated overnight at 4 0C with gentle stirring. Polyethylene glycol-precipitated phage was collected by centrifugation at 15,000 rpm for 20 min. The resulting pellets were resuspended in 3 ml of SM phage buffer (20 mM Tris-HCL [PH 7.4], 100 mM NaCl, 10 mM MgSO4), fi ltered through 0.2 μm bacterial fi lters and phage fi ltrate was recovered and dialyzed against phage buffer. Purifi ed phage GRCST was stored in aliquots of phage buffer at- 20 0C (Sambrook and Russell, 2001)).
Transmission Electron Microscopy
The morphology of ϕGRCST particles was observed by transmission electron microscopy, as previously described (Melo et al., 2014b). A drop of Purifi ed phage GRCST suspension was fi xed with fi xative. Samples were dehydrated with series of ethanol series, passed through a “transition solvent” such as propylene oxide and then infi ltrated and embedded in a liquid resin such as epoxy and LR White resin. The processed suspension was applied to a Farmvar carbon coated grid for 5 min; subsequently stained with 2% uranyl acetate. The grids were examined in a Transmission Electron Microscope at 200kv (2000X – 1500000 X) (Ayache et al., 2010).
Animal Experiment
Selection of Animals
Disease free, healthy and active BALB/c mice breed were selected for in vivo studies. Both female and male mice were chosen for the experimental purpose with animals weighing in the range of 20-30 gm. Animals were obtained from Sri Venkateswara Enterprises, Bangalore, approved by the institute of Animal Ethics Committee (237/99/CPCSEA). Animals were nourished under controlled climate conditions and fed with standard pellet (VRK Nutrition and Solutions, Sangli, Maharashtra, India Ltd.), and provided suffi cient amount of potable water for drinking. Animals were kept for 10 days before experimentation to acclimatize for laboratory conditions. The animals were housed and the entire experiment was carried out in Luqman Pharmacy College, Kalaburagi).
Selection of pathogen and induction of bacteraemia
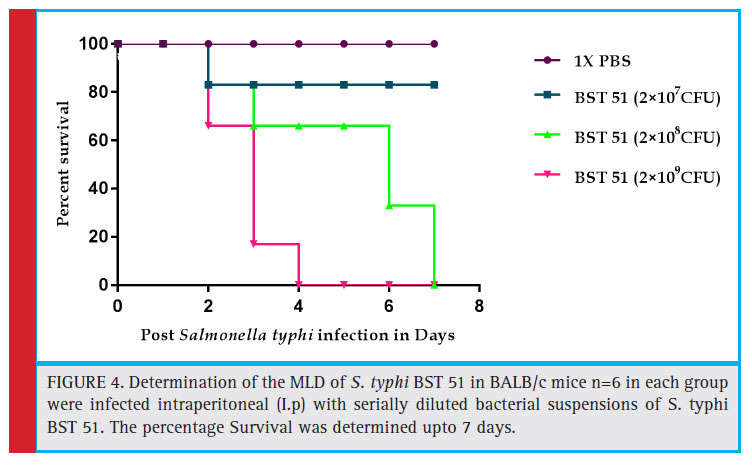
Salmonella typhi BST 51 (Blood Salmonella typhi 51) selected for induction of typhoid fever in experimental mice. S. typhi BST 51 strain has been chosen based on its resistance power to n=7, antibiotics and exhibited a high range of MIC to cefetoxime and also capable of producing ESBL. The selected pathogen was inoculated in LB broth, after 8-12 hr incubation, growth reached 0.2 O.D. Thereafter it was serially diluted in 0.1M PBS and CFU (Colony forming units) was calculated. Subsequently, 107-109 CFU was administered to experimental mice intraperitoneal to determine the MLD (Minimum Lethal Dose).
Effi cacy of bacteriophage in challenged BALB/c mice
Experimental animals (BALB/c) mice were divided into six groups and each group consist of 6 animals each. The doses were fi xed and prepared in PBS and administered intraperitoneal (i.p). Mice from the group I received only PBS as a control, Group II animals administered with S. typhi BST 51 (2×109 CFU) diluted in PBS, Group III animals administered with only GRCST phage (1.5×107 PFU) to check the lethality of phage on Mice. Group IV animals (Mice) represents (Test group), Group V and VI animals represent (Standard) challenged with S. typhi BST 51 (2×109 CFU) by intraperitoneal injection to induce typhoid. Thereafter, 20 mins induction, Group IV (Test) Mice received a GRCST (1.5×107 PFU), and similarly Group V and VI animals received standard ciprofloxacin antibiotic substituting bacteriophage. In Group VI ciprofloxacin were administered in multiple doses, daily up to 7 days. All the six groups were kept in hygienic condition with a continuous supply of food and water for 14 days. The signifi cant observation made and results were recorded (Table 1).
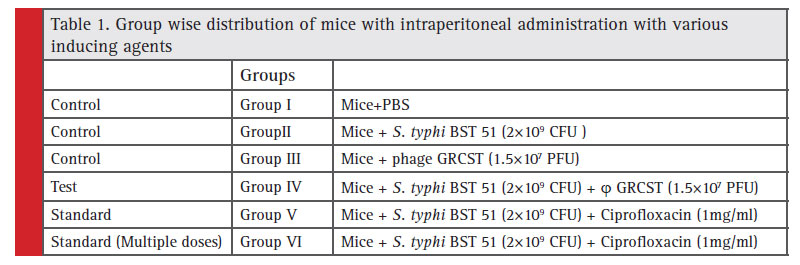 |
Table 1: Group wise distribution of mice with intraperitoneal administration with various inducing agents |
Determination of immunologic response against ϕGRCST in mice
Introduction of bacteriophage in human body as a therapeutic agent cause signifi cant stimulation of humoral immunity subsequently leads to production of antibodies. According to previous reports it is a potent antigen causes no toxic effect on health of humans. During experiment BALB/c mice were treated with ϕ GRCST (1.5×107 PFU) through i.p injection. At various time point, mice blood was collected from optic vein and subsequently subjected for ELISA (Enzyme Linked Immunosorbent Assay) for the detection of antibody titres of IgG and IgM antibody in serum of experimental mice described by Biswas et al., 2002. ELISA is a semi-quantitative method used to determine the concentration of primary antibody in serum in antigen coated wells. In ELISA detection was done based on positive enzyme-substrate reaction makes change in colour.
Results
Isolation and Morphology of Salmonella typhi specifi c ϕGRCST
The plaque formation indicates the presence of S. typhi specifi c bacteriophage i.e. ϕGRCST (Fig 1). TEM results revealed that ϕGRCST (G: Gulbarga, R: Rahul, C: Chandrakanth Kelmani, S: Salmonella, T: Typhi) possesses an icosahedral head with 50 nm size and contractile tail as shown in fi gure belongs to Myoviridae Vi01-like family (Fig 2).
Broad Host Range Screening
In order to investigate the broad host specifi city of ϕGRCST exhibited potential bacteriolytic activity against n=4 (BST 43, BST 94, BST 130, BST 141), ESBL producing S. typhi isolates among n=9 selected isolates. The plaque formation was observed against (n=4) tested isolates (Fig 3 and
Table 2).
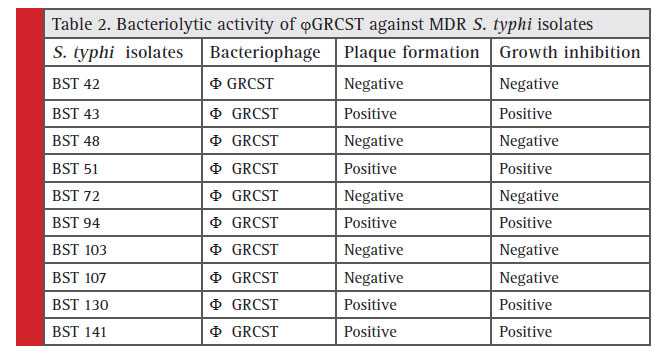 |
Table 2: Bacteriolytic activity of ϕ GRCST gainst MDR S. typhi isolates |
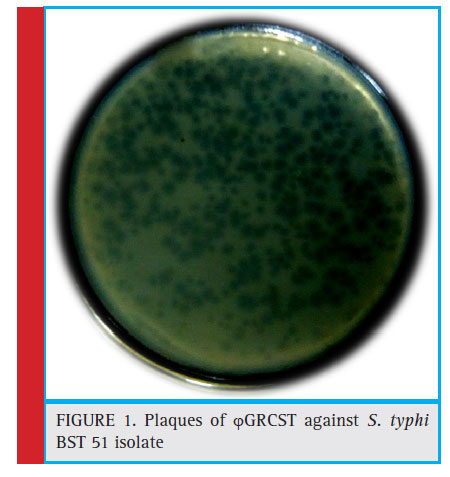 |
Figure 1: Plaques of ϕGRCST against S. typhi BST 51 isolate |
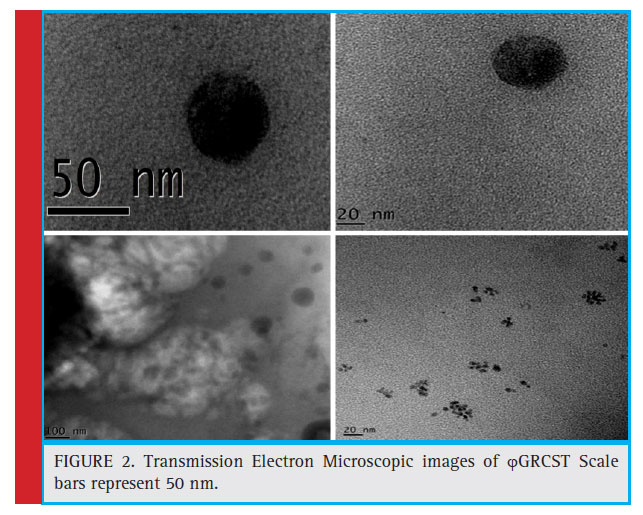 |
Figure 2: Transmission Electron Microscopic images of ϕGRCST Scale bars represent 50 nm. |
Animal Experiment
Experimental induction of S. typhi BST 51 strain in BALB/c mice and determination of Minimum Lethal dose (MLD)
No causality was reported in the fi rst group mice, which received 1XPBS and were proactive and healthy. Consequently, only 83 % of mice survived in group II mice which received until the 7th day of experimentation. However, we observed the 100% mortality in III group on day 7 but in contrast, 100% mortality was recorded on 4th day itself in group IV. Based on the observation 2×109 CFU was determined as MLD (Fig 4).
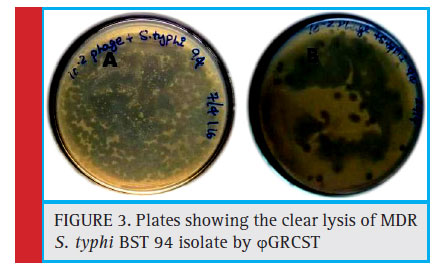 |
Figure 3: Plates showing the clear lysis of MDR S. typhi BST 94 isolate by ϕGRCST |
Treatment and rescue of experimentally challenged BALB/c mice with ϕGRCST
The comparative study was carried out to evaluate the effi cacy of phage GRCST with Standard antibiotic (ciprofloxacin). The observation made on daily basis up to 14 days and data was recorded and represented in the graph. According to the observation made, there was 100% rescue was accomplished challenged BALB/c mice treated with 1.5×107 PFU ϕGRCST, comparatively 67% and 83% survival rate was observed in the group
of mice which received standard antibiotic ciprofl oxacin in a single dose and in multiple doses for treatment. The multiple doses were given on daily basis and results obtained were represented in statistical graph (Fig 5).
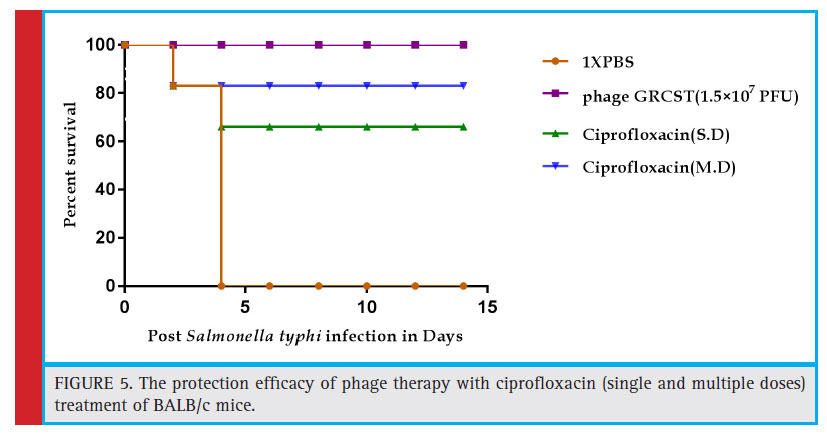 |
Figure 5: The protection effi cacy of phage therapy with ciprofl oxacin (single and multiple doses) treatment of BALB/c mice. |
The host immune response against phage GRCST in BALB/c mice
The blood was collected from the optical vein of BALB/c mice (n=6) and serum was separated, both IgG, IgM titres of anti-phage GRCST antibodies were detected, subsequently increased with 4100 fold, 600 fold respectively in both the cases (Fig 6 and Fig 7). Incubation of phage GRCST with an excess of mice anti-phage GRCST antibodies did not interfere with phage’s capacity to lyse susceptible bacteria. This result demonstrates that the antibodies elicited by ϕGRCST are non-neutralizing.
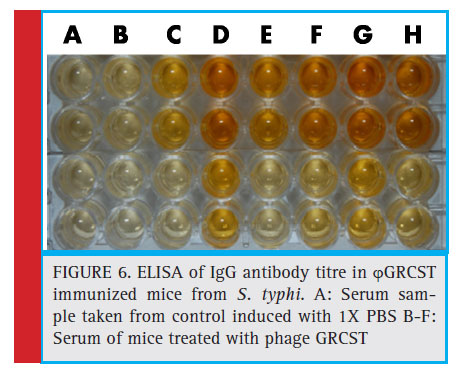 |
Figure 6: ELISA of IgG antibody titre in ϕGRCST immunized mice from S. typhi. A: Serum sample taken from control induced with 1X PBS B-F: Serum of mice treated with phage GRCST |
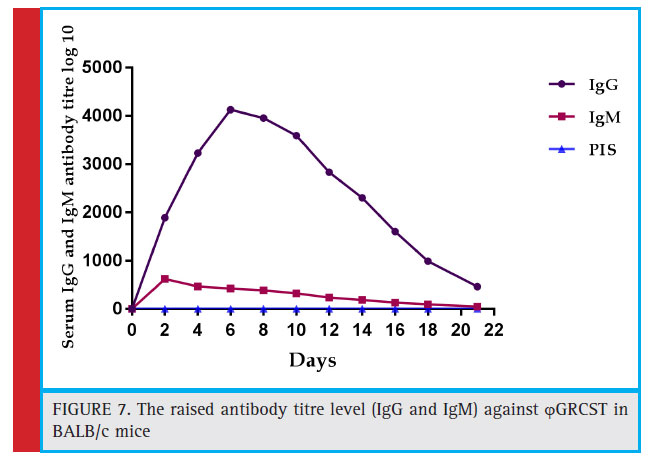 |
Figure 7: The raised antibody titre level (IgG and IgM) against ϕ GRCST in BALB/c mice |
No signifi cant difference was found in BALB/c mice IgG and IgM titres against ϕGRCST. The anaphylactic reactions were negative, and no changes in animal behaviour, no signifi cant changes in the body temperature or no other side effects were observed in both the groups.
Discussion
Over the years, application of phages as therapeutic alternatives or complements to antibiotic therapy has been evaluated extensively (Viertel et al., 2014) and has even been listed by the US National Institute of Allergy and Infectious Diseases as one important approach to combat antibiotic resistance (Reardon, 2014) In the present study, the isolated ϕGRCST effectively infective to MDR S. typhi BST 51 strain (ESBL producing isolate) from sewage sample. In vitro experiments showed remarkable antibacterial activity against the S. typhi BST 51; plaque formation indicates the presence of bacteriophage. The broad host range study of ϕGRCST, demonstrated the effi ciency of ϕGRCST potentially lysed n=4 (44.44%), ESBL producing S. typhi isolates (Table 2). The positive plaque formation was observed in all (n=4) S. typhi strains (Fig 2). Similarly, host range screening was carried out by Wang et al., 2016; with Phage 5460 potentially lysed 12 out of 18 P. mirabilis strains (67%), three out of six P. vulgaris strains and one tested P. penneri strain; while phage 5461 killed all (100%) of the Proteus spp. tested.
Transmission Electron Microscopic (TEM) study of GRCST revealed that our phage belongs to Myoviridae family. The phages which exhibit tail were classifi ed in the order Caudavirales (dsDNA) (Ackermann et al., 2006). This study was concentrated on the bacteriophage in the field of therapeutics, which act against MDR S. typhi, the characteristic feature GRCST possesses an icosahedral head with size 50 nm with contractile tails consisting of a sheath with a central tube (Fig 2); it belongs to Myoviridae Vi01-like family of phages containing S. typhi-specifi c Vi01 (Hooton et al., 2011).
In vivo studies conducted in the BALB/c mice model (weighing from 20-30 gms) for the experimental examination of the effi cacy of ϕGRCST. The mice models were showed the effective of prevention of infection caused by antibiotic-resistant bacteria (Wang et al., 2006; Capparelli et al., 2007; Vinod kumar et al., 2008). In the present study, successfully experimented the in vivo efficacy of GRCST against S. typhi BST 51 infected mice model and obtained moderate results.
The minimum lethal dose (MLD) was determined by intraperitoneal administration of 2×107 CFU, 2×108 CFU and 2×109 CFU bacterial dose. Total of 100% survival rate was recorded in 1X PBS induced mice. Only 83% survival rate was observed in mice received 2×107 CFU bacterial dosage. The comparative study was carried out to evaluate the effi cacy of ϕ GRCST with standard antibiotic (ciprofl oxacin). The 100% rescue was accomplished in challenged BALB/c mice treated with 1.5×107 PFU phage GRCST, comparatively, 67% and 83% survival rate was observed in the group of mice which received standard antibiotic ciprofl oxacin in a single dose and in multiple doses for treatment. The multiple doses were given on daily basis (Fig 4). Relatively, the similar kind of experiment was conducted on S. paratyphi B infected mice and successfully rescued by treating with phage ɸ1 (Wang et al., 2006).
The therapeutic effect of GRCST was successfully achieved in vivo, the given phage (1.5×107 PFU) administered along with saline showed no side effects on health and behavior of the experimental animals. Thus, phage rescue experiments could be conducted by without bias (Uchiyama et al., 2008).
The immunology of phages has been a subject debate over the years. The potential induced phages in; in vivo, subsequently lead to the humoral immune responses ultimately results in the inactivation of phage virion particles. There were some early assumptions; Kucharewica- Krukowska and Slopek, 1987, phage therapy in both animals and patients subsequently affect the patients immunity, by stimulating the immune system and subsequent production of anti-phage antibodies production of neutralizing antibodies, rapid emergence of phageresistant bacterial strains (Stent, 1963; Lederberg, 1996; Cairns et al., 2009) and efficacy of phages only when administered shortly after bacterial infection (Bull et al., 2002); are the most frequent criticisms of the clinical use of phages. Recently, similar kind of studies were conducted by Wang et al., (2016); comparatively results obtained were very much similar to the activity of Phage SLPW showed a broad host range and high efficiency of plating against various types of S. aureus. Phage SLPW remained stable under a various temperatures or pH range. Further, it effi ciently lysed MRSA strains in vitro and in vivo. Intraperitoneal phage administration at 1 h post-infection cured the mice and reduced the bacterial expression of infl ammatory cytokines in mice (Wang et al., 2016).
Titre of IgG and IgM was well measured BALB/c mice after a single dose of ϕGRCST induction, the background titre was increased signifi cantly with 4100 fold, and 600 fold respectively (Fig 6 and Fig 7). Incubation of phage GRCST with an excess of mice anti-phage GRCST antibodies did not interfere with phage’s capacity to lyse susceptible bacteria. This result demonstrates that the antibodies elicited by phage GRCST are non-neutralizing. The similar kind of immune response study was conducted in phageϕ1 against Salp572 (S. paratyphi B) with an elevated level of mice anti-ϕ1 antibodies did not interfere with phage’s capacity to lyse phage-susceptible bacteria. This result demonstrates that the antibodies elicited by phageϕ1 are non-neutralizing (Capparelli et al., 2010). This study clearly emphasizes that mice does produce antibodies against induced phages but they are non-neutralizing. Indeed, Gorski et al., 2006 (Górski, et al., 2007); have provided enough evidence of a positive impact of phages on immune system functioning and have explored potential phage anti-tumour properties mediated through observed shifts in levels of various cytokines as a consequence of interactions between extra decorative head proteins with surface proteins of certain immune-system cells (Budynek et al., 2010). The immune response to GRCST was not associated with anaphylaxis or other adverse immunological reactions. The anaphylactic reactions were negative, and no changes in animal behavior, no signifi cant changes in the body temperature or no other side effects were observed in both the group
Conclusion
The bacteriophage therapy will serve for better perspective, with minimum side effects. The present investigation attempted to fi nd and characterize a bacteriophage infective against the multidrug resistant and ESBL producing S. typhi. Our explored phage was lytic against many MDR S. typhi isolates, it’s in vivo effi cacy proved as an excellent therapeutic agent. The signifi cant outcome of our conducted study a single dose of 1.5×107 PFU of phage GRCST successfully eliminated bacteria from mice circulatory system without infl uencing the host immune system and rescued infected mice, compare to antibiotics failed to rescue all infected mice. Hence, we conclude that our explored GRCST is clinically more effi cient than earlier reported; further characterization like whole genome sequencing and identifi cation and cloning of genes coding for bacterial lysis of the ϕGRCST may prove as an excellent alternative therapeutic agent. Based on our observations of this study, phage therapy can be used as an alternative therapy for those patients not responding to antibiotic treatment.
Conflict of Interest
The authors declare of no confl ict of interest in conducting this study.
References
Abedon, S. T., Kuhl, S. J., Blasdel, B. G., & Kutter, E. M. (2011). Phage treatment of human infections. Bacteriophage, 1(2), 66-85.
Ackermann, H. W. (2012). Bacteriophage electron microscopy. Adv. Virus Res, 82, 1-32.
Ackermann, H.W. (2006). Classifi cation of bacteriophages. In The Bacteriophages, Ed.Calendar R, Oxford University Press, ISBN 0-19-514850-9, New York, USA, pp. 8–16.
Albino, L. A., Rostagno, M. H., Húngaro, H. M., & Mendonça, R. C. (2014). Isolation, characterization, and application of bacteriophages for Salmonella spp. biocontrol in pigs. Foodborne pathogens and disease, 11(8), 602-609.
Anderson, E. S., & Felix, A. (1953). The Vi type-determining phages carried by Salmonella typhi. Microbiology, 9(1), 65-88.
Atterbury, R. J., Van Bergen, M. A. P., Ortiz, F., Lovell, M. A., Harris, J. A., De Boer, A., & Barrow, P. A. (2007). Bacteriophage therapy to
reduce Salmonella colonization of broiler chickens. Applied and environmental microbiology, 73(14), 4543-4549.
Ayache, J., Beaunier, L., Boumendil, J., Ehret, G., & Laub, D. (2010). Sample preparation handbook for transmission electron microscopy: techniques (Vol. 2). Springer Science & Business Media.
Bassetti, M., Poulakou, G., Ruppe, E., Bouza, E., Van Hal, S. J., and Brink,A. (2017). Antimicrobial resistance in the next 30 years, humankind,bugs and drugs: a visionary approach. Intensive Care Med. 43, 1464–1475. doi: 10.1007/s00134-017-4878-x.
Biswas, B., Adhya, S., Washart, P., Paul, B., Trostel, A. N., Powell, B., … & Merril, C. R. (2002). Bacteriophage therapy rescues mice bacteremic from a clinical isolate of vancomycinresistant Enterococcus faecium. Infection and immunity, 70(1), 204-210.
Budynek, P., Dabrowska, K., Skaradzinski, G., Górski, A. (2010). Bacteriophages and cancer. Arch.Microbiol. 192:315–320.
Bull, J. J., Levin, B. R., DeRouin, T., Walker, N., & Bloch, C. A. (2002). Dynamics of success and failure in phage and antibiotic therapy in experimental infections. BMC microbiology, 2(1), 1.
Cairns, B. J., Timms, A. R., Jansen, V. A., Connerton, I. F., & Payne, R. J. (2009). Quantitative models of in vitro bacteriophage– host dynamics and their application to phage therapy. PLoS Pathog, 5(1), e1000253.
Capparelli, R., Nocerino, N., Iannaccone, M., Ercolini, D., Parlato, M., Chiara, M., & Iannelli, D. (2010). Bacteriophage therapy of Salmonella enterica: a fresh appraisal of bacteriophage therapy. Journal of Infectious Diseases, 2011(1), 52-61.
Capparelli, R., Parlato, M., Borriello, G., Salvatore, P., & Iannelli, D. (2007). Experimental phage therapy against Staphylococcus aureus in mice. Antimicrobial agents and chemotherapy, 51(8), 2765-2773.
Chanishvili, N., Sharp, R.A. (2009). Literature Review of the Practical Application of Bacteriophage Research. Tbilisi, Georgia: Eliava Institute.
De Jonge, P. A., Nobrega, F. L., Brouns, S. J. J., and Dutilh, B. E. (2018). Molecular and evolutionary determinants of bacteriophage host range. Trends Microbiol.7:1352. doi: 10.1016/j.tim.2018.08.006
De Kraker,M. E., Stewardson, A. J., and Harbarth, S. (2016). Will 10 million people die a year due to antimicrobial resistance by 2050? PLoS Med. 13:e1002184. doi: 10.1371/journal. pmed.1002184
Deghorain M, Bobay LM, Smeesters PR, Bousbata S, Vermeersch M, Perez-Morga D, Drèze PA, Rocha EP, Touchon M, Van Melderen L (2012) Characterization of novel phages isolated in coagulase-negative staphylococci reveals evolutionary relationships with Staphylococcus aureus phages. J Bacteriol 194:5829–5839
Furfaro, L.L., Payne, M.S. and Chang, B.J., 2018. Bacteriophage therapy: Clinical trials and regulatory hurdles. Frontiers in cellular
and infection microbiology, 8. Górski, A., Borysowski, J., Miedzybrodzki, R., & Weber-Dabrowska, B. (2007). Bacteriophages in medicine (pp. 125-158). Caister Academic Press.
Harper, D. R. (2018). Criteria for selecting suitable infectious diseases for phage therapy. Viruses 10:E177. doi: 10.3390/v10040177
Hooton, S. P., Timms, A. R., Rowsell, J., Wilson, R., & Connerton, I. F. (2011). Salmonella Typhimurium-specifi c bacteriophage SH19 and the origins of species specifi city in the Vi01-like phage family. Virology journal, 8(1), 1.
Kropinski, A. M., Mazzocco, A., Waddell, T. E., Lingohr, E., and Johnson, R. P. (2009). Enumeration of bacteriophages by double
agar overlay plaque assay. Methods Mol. Biol. 501, 69–76. doi: 10.1007/978-1-60327-164-6_7
Kutter EM. Bacteriophage therapy: past and present. In: Schaecter M, editor. Encyclopedia of Microbiology. Oxford: Elsevier; 2009. pp. 258–266.
Lederberg, J. (1996). Smaller fl eas… ad infi nitum: therapeutic bacteriophage redux. Proceedings of the National Academy of Sciences, 93(8), 3167-3168.
Mazzocco, A., Waddell, T. E., Lingohr, E., & Johnson, R. P. (2009). Enumeration of bacteriophages by the direct plating plaque assay. Bacteriophages: Methods and Protocols, Volume 1: Isolation, Characterization, and Interactions, 77-80.
Melo, L. D., Sillankorva, S., Ackermann, H. W., Kropinski, A. M., Azeredo, J., and Cerca, N. (2014b). Isolation and characterization of a new Staphylococcus epidermidis broad-spectrum bacteriophage. J. Gen. Virol. 95(Pt 2), 506–515. doi: 10.1099/vir.0.060590-0
Mikeladze, C., Nemsadze, E., Alexidze, N., Assanichvili, T. (1936). On the treatment of typhoid fever and acute colitis by d’Herelle bacteriophage. La Médecine. 1936; 17:33–38. (Fre).
Reardon, S. (2014). Phage therapy gets revitalized. Nature 510, 15–16. doi: 10.1038/510015a
Sambrook, J., and Russell, D. W. (2001). Molecular Cloning: A Laboratory Manual, 3rd Edn. New York, NY: Cold Spring Harbor Laboratory Press
Stent, G. S. (1963). Molecular biology of bacterial viruses. Molecular biology of bacterial viruses. Sulakvelidze, A., Barrow, P. (2005). Phage therapy in animals and agribusiness. In: Kutter E, Sulakvelidze A, eds. Bacteriophages: Biology and Application. Boca Raton, FL: CRC Press,
335-80.
Tacconelli, E., Carrara, E., Savoldi, A., Harbarth, S., Mendelson, M., Monnet, D.L., et al. (2018). Discovery, research, and development of new antibiotics: the WHO priority list of antibiotic- resistant bacteria and tuberculosis. Lancet Infect Dis. 18, 318–327. doi: 10.1016/S1473-3099(17)30753-3
Tsulukidze, A. (1936). Sur l’application du bacteriophage dans la peritonite par perforation au cours de la fi evre typhoide. La Médecine, 17(Suppl), 41-2.
Uchiyama, J., Rashel, M., Maeda, Y., Takemura, I., Sugihara, S., Akechi, K., … & Matsuzaki, S. (2008). Isolation and characterization
of a novel Enterococcus faecalis bacteriophage EF24 Cas a therapeutic candidate. FEMS Microbiology letters, 278(2), 200-206.
Vinodkumar, C. S., Kalsurmath, S., & Neelagund, Y. F. (2008). Utility of lytic bacteriophage in the treatment of multidrug-resistant Pseudomonas aeruginosa septicemia in mice. Indian Journal of Pathology and Microbiology, 51(3), 360.
Vinogradov, E., and Perry, M. B. (2000). Structural analysis of the core region of lipopolysaccharides from Proteus mirabilis serotypes O6, O48 and O57. Eur. J. Biochem. 267, 2439– 2446. doi: 10.1046/j.1432-1327.2000. 01262.x
Wang, J., Hu, B., Xu, M., Yan, Q., Liu, S., Zhu, X., & Li, Q. Q. (2006). Use of bacteriophage in the treatment of experimental animal bacteremia from imipenem-resistant Pseudomonas aeruginosa. International journal of molecular medicine, 17(2), 309-318.
Wang, Z., Zheng, P., Ji, W., Fu, Q., Wang, H., Yan, Y., & Sun, J. (2016). SLPW: A virulent bacteriophage targeting methicillin resistant Staphylococcus aureus in vitro and in vivo. Frontiers in microbiology, 7, 934.