1School of Life Sciences, Central University of Gujarat, Gandhinagar, Gujarat, India.
2School of Life Sciences, Jawaharlal Nehru University, New Delhi, India.
Corresponding author Email: kunaljii06@gmail.com
Article Publishing History
Received: 20/07/2019
Accepted After Revision: 18/09/2019
Usnic acid is a secondary metabolite obtained from various species of lichen. Previous studies have shown various biological activities of usnic acid such as anti-oxidant, anti-microbial, anti-viral, anti-protozoal, anti-inflammatory and anti-proliferative activities in different models. Its anti-proliferative activities in gastric cancer cells are still unexplored. Herein, we have investigated the effects of usnic acid on cell proliferation and viability and associated molecular alterations using MTT {3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide} assay, RT-PCR (Reverse-Transcriptase-Polymerase Chain Reaction), and western blotting assay in human gastric carcinoma AGS cells. The treatment of usnic acid (2.5-25 µM) dose-dependently reduced cell proliferation. The mRNA expression of tumor suppressor gene PTEN (phosphatase tensin homolog) in the usnic acid-treated AGS cells was increased, which may play a role in the inhibition of cell proliferation and induction of cell death. We also observed a decrease in the expression of proliferating cell nuclear antigen (PCNA) that regulate cell proliferation by playing an important role in DNA replication. The expression of cyclin-dependent kinase inhibitor p21 which may play a role in cell cycle and proliferation inhibition was found uninfluenced with usnic acid treatment. Thus, collectively these results for the first time revealed that usnic acid inhibits the cell proliferation of AGS cells through downregulating the expression of PCNA and can be further evaluated in vivo models for its therapeutic potentials.
Cell proliferation, Cancer, PCNA, Tumor-suppressor Genes
Kumar K, Mishra J. P. N, Singh R. P. Usnic acid inhibits cell proliferation via downregulation of proliferating cell nuclear antigen (PCNA) expression in gastric carcinoma AGS cells. Biosc.Biotech.Res.Comm. 2019;12(3).
Kumar K, Mishra J. P. N, Singh R. P. Usnic acid inhibits cell proliferation via downregulation of proliferating cell nuclear antigen (PCNA) expression in gastric carcinoma AGS cells. Biosc.Biotech.Res.Comm. 2019;12(3). Available from: https://bit.ly/2kzRWuo
Copyright © Kumar et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
Introduction
Gastric cancer is the fifth most commonly diagnosed and third leading cause of cancer-related death throughout the world. The recent reports have revealed that gastric cancer incidence is twofold higher in men than women. Despite significant advancement in diagnostic and surgical oncology in recent decades and the use of various adjuvant therapy, gastric cancer remain a major cause of mortality due to metastasis. Various natural agents having the capacity to inhibit the cancer cell proliferation or carcinogenesis or tumorigenesis or reverse/arrest the progression of malignant cells have been used for cancer management ( Bray et al 2018, Zhang et al., 2018). Usnic acid, a dibenzofuran derivative, is obtained from a number of lichen species, has shown its potential in various biological activities including anti-inflammatory and anti-proliferative activities (Ingolfsdottir, 2002). Previous studies have demonstrated that usnic acid has the potential to inhibit tumor cell proliferation, angiogenesis, invasion and migration in various types of tumor cells (Singh et al., 2013, Geng et al., 2018).
However, the mechanism of usnic acid-induced anti-proliferative and cells death-inducing properties in gastric cancer cells is yet to be explored. Uncontrolled cell proliferation and division are the primary characteristics of cancer cells (Farber, 1995). Cell proliferation plays a key role in initiation, promotion or progression during cancer development. Abnormal regulations of cell division can lead to over proliferation and accumulation of abnormal cells. A number of key genes and proteins that regulate cell proliferation and the process of cell division have been identified (Valdespino-Gomez et al., 2015). It is now clear that mutations in key genes affect the action of regulating proteins and enzymes and lead to unregulated cell proliferation which is seen in cancer. Different types of cancers have different mutational signatures. However, the study of multiple types of tumor has revealed that certain genes are mutated in cancer cells more often than others. Other cancer-related mutations inactivate the genes that suppress cell proliferation or those that signal to program cell death. These genes are known as tumor suppressor genes that normally function as a brake on cell proliferation and are involved in the protection of cells from a single event or multiple events that lead to cancer (Wang et al., 2018b).
In the present study, we have explored the effect of usnic acid on gastric carcinoma AGS cells. Our study revealed that usnic acid inhibits cell proliferation via modulating the expression of tumor suppressor genes. The up-regulation of PTEN and the down-regulation of PCNA expression clearly indicates the relevance of this study in highlighting the usnic acid as a probable cancer targeting agent in gastric cancer cells. However, further validation of usnic acid in in vivo model to develop as a potential anti-cancer agent is obligatory.
Material and Methods
Cell Culture and Reagents
Gastric carcinoma AGS cells (ATCC, USA) were grown in Ham’s F-12 nutrient mixture medium (Gibco BRL, Grand Island, NY) supplemented with 1% antibiotic-antimycotic solution (HiMedia) and 10% Fetal Bovine Serum (Gibco Life Technology) in a humidified 37°C incubator. (+)- Usnic acid and MTT dye were procured from Sigma Aldrich, USA, and SRL, India respectively. Procurement of anti-PCNA, anti-p21 (CIP) and anti-GAPDH was done from Cell Signaling Technology, USA.
Cell Viability Assay
Cell viability of gastric carcinoma AGS cells was examined through MTT assay as described earlier (Mohan et al., 2016). Briefly, AGS cells (8,000/well) were seeded in a flat bottom 96 well plate and after overnight incubation treated with 0, 2.5, 5, 10, 15 and 25 µM usnic acid for 72 h. Further, cells were processed for MTT assay.
Semi-quantitative RT-PCR Assay
Cells (5×105/plate) were seeded in 100 mm plates and after 24 h incubation treated with 0, 10, 15 and 25 µM usnic acid for 48 h. Once the treatment period was over, cells harvested for RNA isolation followed by cDNA synthesis, and RT-PCR as detailed earlier (Jaiswal et al., 2018). Briefly, Total RNA isolation was done by Trizol Methods, then 5 µg RNA was used for cDNA synthesis. The final volume of the PCR reaction mixture was 25 µl with 3 µg of cDNA, forward and reverse primers (20 pM) and all required components. PCR tube was kept in a thermal cycler (Eppendorf, India) for 25 cycles. Agarose gel (1%) with (1µg/ml) ethidium bromide separate the amplified DNA and visualized in the GelDoc system. The following primer sequence was used: for PTEN forward primer- ACC AGG ACC AGA GGA AAC CT, reverse primer- GCT AGC CTC TGG ATT TGA CG, for GAPDH forward primer- AAG GCT GAG AAC GGG AAG CTT GTC ATC AAT, reverse primer- TTC CCG TTC AGC TCA GGG ATG ACC TTG CCC.
Immunoblotting
Cells were seeded and treated as described in the previous section. After 48 h incubation, cells were lysed to prepare whole cells lysate as described (Kumar et al., 2019). The protein concentration was estimated by the Bradford method. Further, the protein was resolved in 10% denaturing SDS-PAGE gel and transferred on PVDF membrane. Skimmed milk (3%) in blocking buffer was used to block the membrane. Subsequently, the membrane was incubated in specific antibody (diluted in skimmed milk) followed by appropriate HRP-linked secondary antibody and the bands were detected by ECL and capture on X-ray film. The band density was analyzed by scanning the film with high-resolution scanner and Image J program (NIH, Bethesda, MD).
Statistical Analysis
For statistical significance, GraphPad Prism-5 program was used. Student’s t-test was applied to detect the degree of significance between control and treated samples. P-value of < 0.05 was considered significant.
Results and Discussion
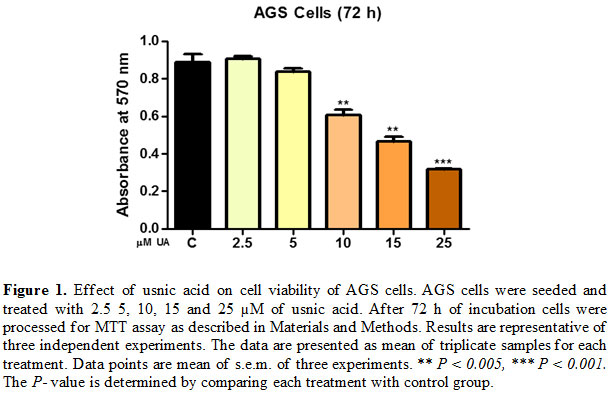
Effect of usnic acid on cell viability of AGS cells
With the aim to evaluate the effects of usnic acid on cell proliferation and viability, gastric carcinoma AGS cells were exposed to increasing concentrations of usnic acid ranging from 2.5 to 25 µM for 72 h. Treatment with 2.5 µM usnic acid resulted in no marked decrease in cell viability however at (5-25) µM concentrations showed (6-65%) decrease in cell viability after 72 h [Fig. 1].
These results indicated that usnic acid caused a concentration-dependent reduction in cell viability of AGS cells.
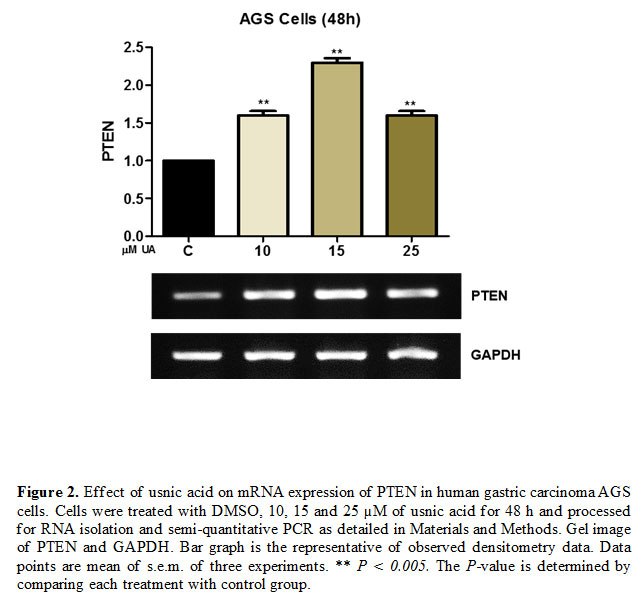
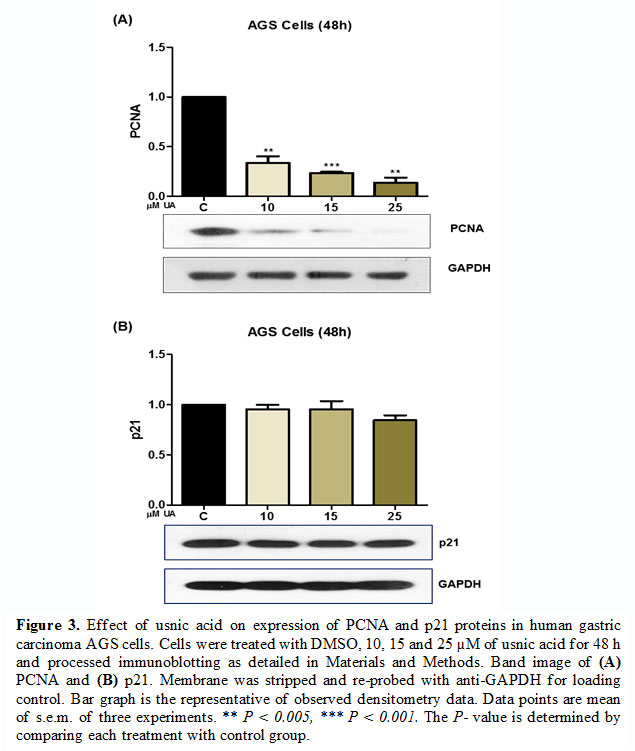
Gene and protein expression modulation by usnic acid treatment in AGS cells
To access the effect of usnic acid on the expression of tumor suppressor gene and proliferation-inducing proteins, AGS cells were treated with
(10-25 µM) of usnic acid for 48 h and processed for semi-quantitative PCR and western blot analysis. The result of semi-quantitative PCR revealed that usnic acid induces the expression of PTEN at 48 h time point [Fig. 2]. Similarly, the immunoblotting analysis revealed that usnic acid decreased the expression of PCNA which has an important role in DNA replication and hence cell proliferation [Fig. 3 (A)]. However, at a lower concentration of usnic acid the expression of p21 has no effect but at higher concentration (25 µM), its expression was decreased [Fig. 3(B)].
In various cases of gastric cancer, the tumor suppressor protein PTEN (phosphatase Tensin Homolog) has been detected functionally inactive and shown to be closely associated with the development, progression, and prognosis of the disease (Xu et al., 2014, Lu et al., 2016). PTEN can induce apoptosis and suppress cell proliferation by antagonizing the phosphatidylinositol 3- kinase (PI3K)/Akt signaling pathway. Our results have shown that usnic acid induces the gene expression of PTEN. Which might be helpful in controlling the cell proliferation and inducing cell death of AGS cells.
On the other hand, PCNA, a DNA clamp that is essential for DNA replication and has been closely associated with cell proliferation (Wang et al., 2018a). It has an active role in cyclin-dependent kinase regulations during cell division (Strzalka and Ziemienowicz, 2011). Its expression is associated with metastasis and malignancies (Peng et al., 2019). Hence, the expression of PCNA can be regarded as a marker of proliferating cells. Our result has shown that usnic acid dose-dependently decreases the expression PCNA in gastric carcinoma AGS cells. However, the expression of cyclin-dependent kinase inhibitor p21 (Cip) relatively ineffective with usnic acid treatments. These results indicated that usnic acid targets the PCNA expression and hence cell proliferation without modulating the expression of other cell cycle inhibitors at least p21.
Conclusion
Our results for the first time shows that usnic acid negatively modulates the cancer cell viability that can play important role in cancer prevention and therapeutics. The down-regulated expression of PCNA and up-regulated expression of PTEN is associated with inhibition of cell proliferation and induction of cell death in gastric carcinoma AGS cells. However, the cell death induction appears poorly associated with the expression of cyclin-dependent kinase inhibitor (p21).
Acknowledgments
The work was supported by the funds from the Central University of Gujarat, India, and UPE-II, UGC-RN and DST-PURSE, JNU, New Delhi. KK was supported by a fellowship from University Grants Commission, New Delhi, India.
References
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A. & Jemal, A. (2018). Global cancer statistics 2018: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin, 68, 394-424.
Farber, E. (1995). Cell proliferation as a major risk factor for cancer: A concept of doubtful validity. Cancer Res, 55, 3759-62.
Geng, X., Zhang, X., Zhou, B., Zhang, C., Tu, J., Chen, X., Wang, J., Gao, H., Qin, G. & Pan, W. (2018). Usnic acid induces cycle arrest, apoptosis, and autophagy in gastric cancer cells in vitro and in vivo. Med Sci Monit, 24, 556-566.
Ingolfsdottir, K. (2002). Usnic acid. Phytochemistry, 61, 729-36.
Jaiswal, A., Sabarwal, A., Narayan Mishra, J. P. & Singh, R. P. (2018). Plumbagin induces ros-mediated apoptosis and cell cycle arrest and inhibits emt in human cervical carcinoma cells. RSC Advances, 8, 32022-32037.
Kumar, K., Sabarwal, A. & Singh, R. P. (2019). Mancozeb selectively induces mitochondrial-mediated apoptosis in human gastric carcinoma cells through ros generation. Mitochondrion, 48, 1-10
Lu, X. X., Cao, L. Y., Chen, X., Xiao, J., Zou, Y. & Chen, Q. (2016). Pten inhibits cell proliferation, promotes cell apoptosis, and induces cell cycle arrest via downregulating the pi3k/akt/htert pathway in lung adenocarcinoma a549 cells. 2016, 2476842
Mohan, V., Agarwal, R. & Singh, R. P. (2016). A novel alkaloid, evodiamine causes nuclear localization of cytochrome-c and induces apoptosis independent of p53 in human lung cancer cells. Biochem Biophys Res Commun, 477, 1065-1071
Peng, B., Ortega, J., Gu, L., Chang, Z. & Li, G. M. (2019). Phosphorylation of proliferating cell nuclear antigen promotes cancer progression by activating the atm/akt/gsk3beta/snail signaling pathway. 294, 7037-7045
Singh, N., Nambiar, D., Kale, R. K. & Singh, R. P. (2013). Usnic acid inhibits growth and induces cell cycle arrest and apoptosis in human lung carcinoma a549 cells. Nutr Cancer, 65 Suppl 1, 36-43
Strzalka, W. & Ziemienowicz, A. (2011). Proliferating cell nuclear antigen (pcna): A key factor in DNA replication and cell cycle regulation. Ann Bot, 107, 1127-40
Valdespino-Gomez, V. M., Valdespino-Castillo, P. M. & Valdespino-Castillo, V. E. (2015). [cell signaling pathways interaction in cellular proliferation: Potential target for therapeutic interventionism]. Cir Cir, 83, 165-74
Wang, L., Kong, W., Liu, B. & Zhang, X. (2018a). Proliferating cell nuclear antigen promotes cell proliferation and tumorigenesis by up-regulating stat3 in non-small cell lung cancer. Biomed Pharmacother, 104, 595-602
Wang, L. H., Wu, C. F., Rajasekaran, N. & Shin, Y. K. (2018b). Loss of tumor suppressor gene function in human cancer: An overview. Cellular Physiology and Biochemistry, 51, 2647-2693
Xu, W. T., Yang, Z. & Lu, N. H. (2014). Roles of pten (phosphatase and tensin homolog) in gastric cancer development and progression. Asian Pac J Cancer Prev, 15, 17-24
Zhang, Q. Y., Wang, F. X., Jia, K. K. & Kong, L. D. (2018). Natural product interventions for chemotherapy and radiotherapy-induced side effects. Front Pharmacol, 9, 1253.