1Department of Biotechnology, Koneru Lakshmaiah Education Foundation, Guntur, India
2,3,4,5,6Department of Petroleum Engineering, KoneruLakshmaiah Education Foundation, Guntur, India
Corresponding author Email: kkchandrika@kluniversity.in
Article Publishing History
Received: 06/04/2019
Accepted After Revision: 15/06/2019
The mathematical modelling of the growth of microalgae (Nannochloropsis) was developed under flue gas in a photo-bioreactor. Carbon dioxide (CO2) was the major growth parameter used to model algal growth. The critical SOx concentration and CO2mass transfer rate were considered under the growth inhibitory model (GIM) which played a big role in pH based inter-conversion of the bicarbonate. The Nannochloropsis profile was validated at 0.04% of CO2 in air and in a range of 2% to 12% (v/v) CO2 and the predicted values were observed consistent with the measured values. Sensitivity analysis was used to justify the constants used in the GIM. The growth inhibitory model was defined as ±0.5Lmin-1 of the calibrated flow rate of 3.0 Lmin-1 .The growth model was used to predict the growth of microalgae under flue gas to generate biomass as a feed stock for the production of biofuels.
Microalgae, Nannochloropsis, Growth Inhibitory Model, Sensitivity Analysis.
Chandrika K, Majok E. M, Das A, Paul S. K, Sandeep T. N, Kumar P. A. The Mathematical Modelling of Algal Growth for the Production of Biofuels. Biosc.Biotech.Res.Comm. 2019;12(2).
Chandrika K, Majok E. M, Das A, Paul S. K, Sandeep T. N, Kumar P. A. The Mathematical Modelling of Algal Growth for the Production of Biofuels. Biosc.Biotech.Res.Comm. 2019;12(2). Available from: https://bit.ly/2QInzgC
Copyright © Chandrika et al.,, This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
Introduction
The generation of biofuels from microalgae is as a result of a high thrill for alternative energy sources. The increasing population which has led to the increase number of vehicles and high energy demand has left the world market with scarcity of fossil fuels (Minneapolis, MN, USA, 2011). Biodiesel can be obtained from any vegetable oils like sunflower, cotton seeds, peanut, safflower and microalgae lipids, but microalgae has the highest lipid yield (Demirbas and Demirbas, 2013). Algae cultivation is done in open ponds and bioreactors, but to control the growth parameters, algae is cultivated in photobioreactors. Nannochloropsis are algae species of cold-water marine, with high triglycerides (triglyceride is an ester derived from glycerol and three fatty acids) and high growth rate (Archambault, 2014) and therefore, it is most opted as the source of biofuel, (Islam et al., 2013, Mondal et al., 2017 and Sharma et al., 2019).
Factors affecting microalgae growth include light intensity, PH, presence of nitrogen (Danesi et al., 2002; Colla et al., 2007; Celekli and Yavuzatmaca, 2009), presence of contaminants (Vonshak, 1997), initial biomass concentration (Costa et al., 2002), population density (Vonshak, 1997), temperature, salinity and growth dynamics and much more. All these parameters can be controlled and improved only when the algae cultivation is done in a photobioreactor, instead of an open pond.
It is important to understand growth modelling of organisms and also its behaviour under vast different environmental conditions which include PH, temperature, nutrients (Zwietering et al., 1990; Celekli et al., 2008; Celekli and Yavuzatmaca, 2009) and light intensity.Differentmodels like Gompertz model were used to describe the curve of microbial growth and biovolume production (Zwietering et al., 1990; Celekli et., 2008). These models are also used to predict microbial biomass production and optimizing the growth conditions (Costa et al., 2002; Celekli et al., 2008; Celekli and Yavuzatmaca, 2009). Gompertz model is modified to give lag time, specific rate of growth and stationary phase (maximum population biovolume) (Zwietering et al., 1990; Celekli et al., 2008). Other growth models have been put on the table to describe growth during the cultivation (Rangel-Yagui et al., 2004; Chojnacka and Noworyta, 2004, Mondal et al., 2017 Singh and Singh, 2019).
With the escalating climate change, global warming has been a major concern worldwide. Presence of much greenhouse gas concentrations in the atmosphere is the major cause of ozone layer depletion that leads to global warming, in which carbon dioxide takes a lion’s share with an estimation of 76.7% (v/v) and this concentration has been increasing rampantly from industrial revolution(Sharma et al., 2019). It is estimated that emission of carbon dioxide from coal thermal plants is about 7% (v/v) of global CO2 emission and that emitted from flue gas of power plants is 10% (v/v) to 15% (v/v), and these exclude the emissions from transportation sector (automobiles)(Natural Gas Production, 2012).
As the population increases, there is an increase in the number of vehicles with high demand of petroleum products which contribute to the emission of greenhouse gases (CO2 in particular). This has brought the attention to the scientist and researchers to look for ways of mitigating the concentrations of CO2 from the atmosphere (Sharma et al., 2019).
It’s has been found in some studies that growth of microalgae is much more influenced by carbon dioxide and sunlight; nutrients are also required for biomass production (Mondal et al., 2017). The concern of this paper is to prepare a model that maximizes biomass production from microalgae by use flue gas, for the production of bio-fuel, (Majid et al., 2014).
Flue gas is used to make this model. In flue gas, toxins like SOx and NOx have to be put into consideration, the presence of these toxins inhibit algal growth. The use of flue gas as a source of CO2 for algal growth would reduce the content of emitted CO2 from automobiles and industries, in the atmosphere. Air contains less percentage (0.04%) of CO2 and the growth of microalgae that gets CO2 from the air would be gradual and biomass production minimal. On the other hand, high concentrations of CO2 would drive microalgae to death phase (Singh and Singh, 2019).
Hence, determining the maximum concentration of CO2for the growth of algae is more preeminent.
Methodology
Organism and culture conditions for model development
Nannochloropsis limnetica 18.99 was obtained from a culture collection of Algae, SAG, Germany. The culture was grown in a basal medium (Shen and Yuan, 2012) at 30oC and a pH of 8.0. In all experiments, the initial cell density was maintained at 0.01 g L-1. Cool white fluorescent lamps were mounted on either side of the glass reactor (bubble column: 30 L; 220 mm dia ; 3:1 H/D) with 100 μmol m-2s-1 light intensity. The temperature, pH, and algal growth were monitored every 24 hours. Algal growth was estimated in terms of cell count using a Neubaur chamber (Rohem, India) .The number of cells was converted to equivalent dry weight values by the regression equation.
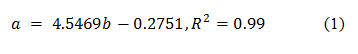
Where α is the dry is weight (g L-1); is the cell number expressed in cells L-1.
Effective growth rate and SOx inhibitory parameters
A series of experiments were made to determine effective growth rate (µeff), consisting CO2 (% v/v) inputs at different concentrations of SOx. The PH was adjusted in Erlenmeyer flasks (250mL) with dilute H2SO4 (18.4M) to simulate the effect of SOxin the growth medium, therefore different concentrations of SO42-(0-150 mg L-1) were generated. The effective growth rate (µeff) under a concentration of SO42-(MSOx) was measured in each experimental flask by recording cell concentration with respect to time.
A linear plot logarithmic cell concentration versus time was then used to derive µeff from the slope of the curve. A supplementary linear plot, comprising µeff, versusSO42- concentration wasalso used to derive the constant c which is equivalent to specific growth rate ( ) from the Y-intercept and the SOx inhibitory constant from the slope of the plot following Eq. (10). The critical SO42- concentration (Mt) was also determined from the same plot where the SO42- concentration corresponding to the µeff = 0 was recorded to be the Mt for the algae.
Growth Model development
The model consists the following factors: (i) The growth inhibition in the presence of flue gas that depends on partial pressures of CO2 and SOx;(ii) The substrate (HCO3–) concentration is influenced by the pH of the medium; (iii) The effective growth rate (µeff) is influenced by critical toxin (Mt) in the medium.
To minimize the complexity, some of the assumptions were made; no external mass transfer influence due to a diffusion process from the head space into the culture, the total pressure of the system is constant at operational gas velocities; the light intensity, temperature and O2 inhibitory were not considered as but kept constant. The CO2 in the feed was assumed to be consistent throughout stages of growth and sequestered CO2 is consistent during the entire exponential phase.
Gas phase CO2 and SOx mass transfer and their solubility
The rate of mass transfer of CO2 and SOx into the liquid is estimated with an assumption of a steady state and the mass balance of the inlet and outlet volumetric flow rate of flue gas comprising mole fraction of CO2 and SOx is as below;

Where and are the mass transfer rates of CO2 and SOx across air-water interface; G is gas the flow rate, and are the inlet mole fraction of CO2 and SOx respectively; and are the outlet mole fraction of CO2 and SOx in the flue gas; (mol.w)CO2 and (mol.w)Sox are molecular weights of CO2 and SO2 respectively; H is the total sparging time of flue gas in a day; Pt is total pressure; and R,T&V are universal constant, absolute temperature and total volume of the liquid respectively.
The concentration of the liquid phase of CO2 and SOx in equilibrium with that of CO2 and SOx in the flue gas feed is described as follows:
![]()
Where MLm is CO2 concentration at equilibrium; Pt is the total pressure; is the mole fraction of gas component; He is Henry’s constant for CO2. Similarly;
![]()
Where is mass transfer of from gas to liquid medium; R, T and He are universal gas constant, absolute temperature and Henry’s constant of respectively. Initial substrate (ML) is an important variable influencing growth rate in the model development, and an assumption of steady state between absorption and consumption of the substrate was made, while CO2 transfer from the flue gas was driven by ML gradient in the liquid which is aided by the mass transfer coefficient of the system. Therefore;
![]()
Where the volumetric mass is transfer coefficient; is the equilibrium concentration of CO2 and is the concentration of dissolved CO2 at a defined time. The was estimated experimentally by using the static method. Hence;
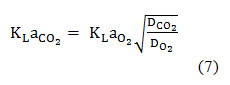
where and were the diffusivities of CO2 and O2 respectively
Inhibitory growth kinetics
To model the effective growth rate appropriately, the effects of CO2 and SOx inhibition have to be combined.
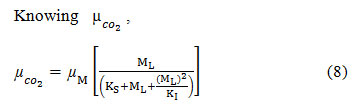
The growth time slows down when SO42 accumulates during the transfer of SOxinto the liquid medium, while considering SOx inhibition along with µCo2 then the growth rate as a function of is modeled as;
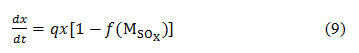
Where is the dissolved concentration of SOx in the form of SO42, q is the SOx inhibitory constant. When SOx linearly decreases growth rate,
![]()
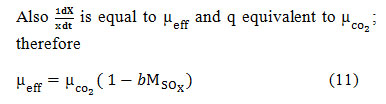
The critical time for SOx inhibition
The SOx in the flue gas is toxic to the algal cells when the reduced pH reaches the critical concentration. Thus, it’s of a great significance to correlate the defined mass transfer rate of SOx in the liquid medium and the time at which the critical concentration is reached.Therefore, toxic time and stationary phase are modelled as follows;
![]()
Where is the critical SOx concentration, is the dissolved SOx (SO42-) concentration at a defined time.
Results and Discussion
Toxic components in the flue gas have influence on algal growth, high concentration of CO2; residual SOx drastically affect the growth rate.
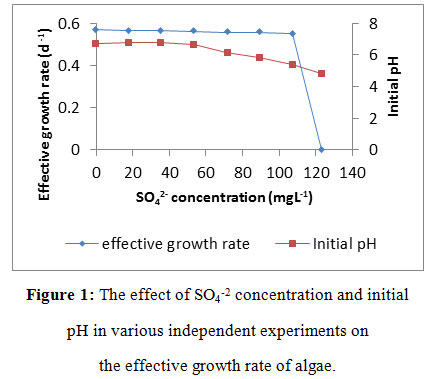 |
Figure 1: The effect of SO4-2 concentration and initial pH in various independent experiments on the effective growth rate of algae. |
Figure 1 shows graphical interpretation of effective growth rate against SOx (SO42-) concentrations with the corresponding values of initial pH. The active growth was under alkaline condition with the pH of 6.8, as SOx concentration increases, the effective growth rate (µeff) decreases linearly with a constant . The effective growth rate fluctuates slightly from 0.48 to 0.43 per day and suddenly to 0.0 at SOx critical concentration (Mt=123 mgL-1), and this leads to substrate (HCO3) depletion and thus affecting the and µeff . The change in effective growth rate profile before reaching Mt is insignificant in comparison to µeff at Mt. This supports theoretical evidence that is constant before reaching Mt with less effect of toxin (SO42), whereas sudden drop in at Mt, toxin concentration has a great effect on µeff.
Table 1: List of best fit parameter constants and their comparison with literature
| Parameter | *Best fit value | Comparable values from literature |
| µM | 0.82 day-1 | 1.0 day-1;Forján et al. (2007) |
| KI | 1.02 g L-1 | 10 mM@ ;Silva and Pirt. (1984) |
| KS | 0.07 g L-1 | 0.12 gL-1;Chen et al. (2010) |
| Ct | 123mg L-1 | 300 ppm@ (gas phase); Lee et al. (2000) |
*The best fit value is the base value in sensitivity analysis; @: KI and Ct equivalent to 0.44g L-1and 133mg L1 (liquid phase concentration) respectively.
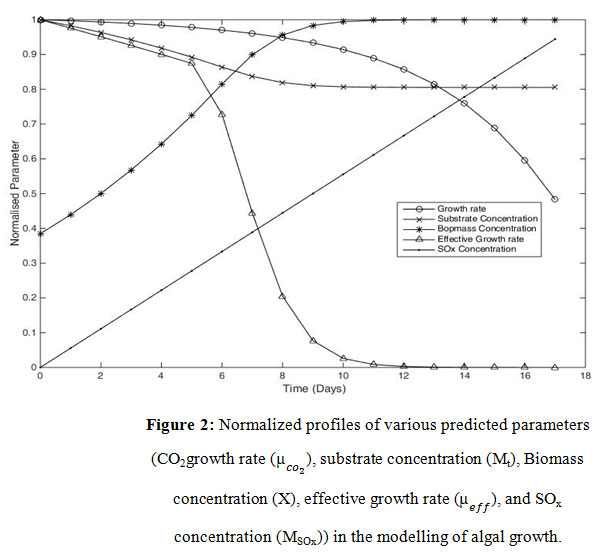
In figure 2, CO2growth rate ( ), substrate concentration (Mt), Biomass concentration (X), effective growth rate ( ), and SOx concentration (MSOx) were taken as normalized parameters against time. Growth kinetics like effective growth rate and CO2growth rate were plotted with their maximum inputs against time. The is stable until reaching critical SOx concentration (Mt) and then declined drastically after reaching, affecting the , according to Eq. (11). Biomass concentration increases with time until it reaches the phase of decline and then remains constant. Substrate (HCO–3) concentration depletes as it is consumed during algal growth and becomes constant when the algal growth stops. Since the SOx concentration is not utilized during the algal growth, it keeps on increasing as flue gas is supplied to the algal culture.
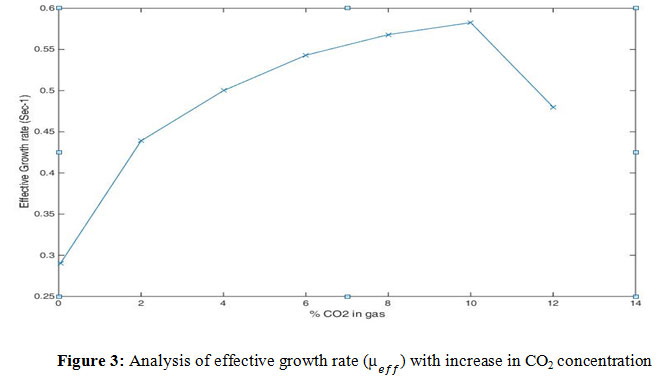 |
Figure 3: Analysis of effective growth rate ( ) with increase in CO2 concentration |
Air contains 0.04% of carbon dioxide and the toxins like SOx and NOx are negligible and therefore, there is no inhibition of SOx to the algal growth. The algal increases as CO2percentage in the flue gas increases. The increase in the is seen when carbon dioxide ranges from 0.0004% to 10%. Continuous supplying of CO2 to the algal culture, beyond 10% of CO2 leads to the decline in the effective growth rate and eventually, to the death phase.
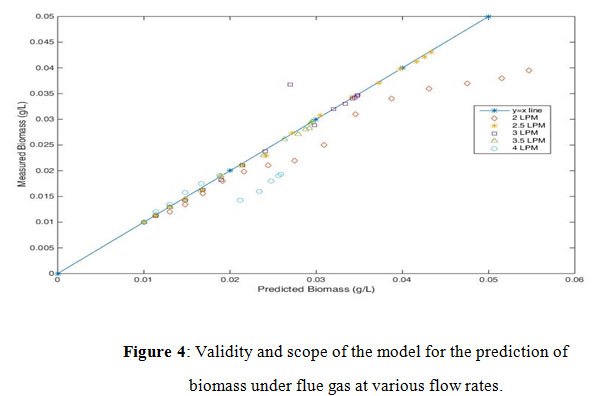 |
Figure 4: Validity and scope of the model for the prediction of biomass under flue gas at various flow rates. |
Flow rate is one of the operating parameters and it shows a direct impact on biomass production. Six different flow rates were used to check the validation of the model (Figure 4) by using predicted and experimented biomass concentrations. From the profile shown in figure 4, for the validity of the model for the prediction of biomass, higher deviations are observed at high flow rates of >3.5LPM and at low flow rates of <2.5LPM. The model is valid at the region of flow rates ranging 2.5LPM to 3.5LPM.
Conclusion
In this model, certain amount of carbon dioxide can be reduced from the atmosphere as flue gas from industries or vehicles is used for algal growth. The cell growth was directly affected by SOx concentration which also influenced the pH dependent kinetics. The validity of this model was observed as ±0.5Lmin-1. Little variation (±10%) of the base value of constants used in GIM was observed in sensitivity analysis. It was also found out that up to 10% (v/v) of CO2 is required for this model and beyond this CO2 concentration, effective growth rate leads to decline phase.
References
Çelekli, A., Balcı, M. and Bozkurt, H. (2008). Modelling of Scenedesmus obliquus; function of nutrients with modified Gompertz model. Bioresource Technology, 99(18), pp.8742-8747.
Demirbas, A. and Demirbas, M. (2013). Algae Energy. Guildford, Surrey: Springer London.
Difusa, A., Talukdar, J., Kalita, M., Mohanty, K. and Goud, V. (2015). Effect of light intensity and pH condition on the growth, biomass and lipid content of microalgae Scenedesmus species. Biofuels, 6(1-2), pp.37-44.
Duan, Y. and Shi, F. (2014). Bioreactor design for algal growth as a sustainable energy source. Reactor and Process Design in Sustainable Energy Technology, pp.27-60.
Islam, M., Magnusson, M., Brown, R., Ayoko, G., Nabi, M. and Heimann, K. (2013). Microalgal Species Selection for Biodiesel Production Based on Fuel Properties Derived from Fatty Acid Profiles. Energies, 6(11), pp.5676-5702.
Juneja, A., Ceballos, R. and Murthy, G. (2013). Effects of Environmental Factors and Nutrient Availability on the Biochemical Composition of Algae for Biofuels Production: A Review. Energies, 6(9), pp.4607-4638.
MacDougall, K., McNichol, J., McGinn, P., O’Leary, S. and Melanson, J. (2011). Triacylglycerol profiling of microalgae strains for biofuel feedstock by liquid chromatography–high-resolution mass spectrometry. Analytical and Bioanalytical Chemistry, 401(8), pp.2609-2616.
Majid, M., Shafqat, S., Inam, H., Hashmi, U. and Kazi, A. (2014). Production of Algal Biomass. Biomass and Bioenergy, pp.207-224.
Moisescu, C., Ardelean, I. and Benning, L. (2014). The effect and role of environmental conditions on magnetosome synthesis. Frontiers in Microbiology, 5.
Mondal, M., Goswami, S., Ghosh, A., Oinam, G., Tiwari, O., Das, P., Gayen, K., Mandal, M. and Halder, G. (2017). Production of biodiesel from microalgae through biological carbon capture: a review. 3 Biotech, 7(2).
Natural Gas Production. (2012). Oil and Energy Trends, 37(9), pp.22-24.
Shalaby, E., El-Moneim M. R. Afify, A. and Shanab, S. (2010). Enhancement of biodiesel production from different species of algae. Grasas y Aceites, 61(4), pp.416-422.
Shao, X., Mugler, A., Kim, J., Jeong, H., Levin, B. and Nemenman, I. (2017). Growth of bacteria in 3-d colonies. PLOS Computational Biology, 13(7), p.e1005679.
Sharma, K., Schuhmann, H. and Schenk, P. (2012). High Lipid Induction in Microalgae for Biodiesel Production. Energies, 5(5), pp.1532-1553.
Sharma, N., Aggarwal, A., Eastwood, P., Gupta, T. and Singh, A. (2019). Air Pollution and Control. Springer.
Sharma, N., Aggarwal, A., Eastwood, P., Gupta, T. and Singh, A. (2019). Air Pollution and Control. Springer.
Singh, S. and Singh, P. (2019). Effect of CO2 concentration on algal growth: A review.
Tjørve, K. and Tjørve, E. (2017). The use of Gompertz models in growth analyses, and new Gompertz-model approach: An addition to the Unified-Richards family. PLOS ONE, 12(6), p.e0178691.