1Department of Biology, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia.
2Department of Biology, Faculty of Science, Taif University, Saudi Arabia.
3Department of Cell Biology, National Research Center, Dokki, Giza, Egypt.
4Botany Department, Faculty of Science, Kafrelsheikh University, Egypt
Article Publishing History
Received: 14/07/2020
Accepted After Revision: 25/09/2020
Isolating microbes from diverse natural ecological units has led to achieve metabolite structural diversity. Hot springs have been less explored ecological sects for discovery of novel microbial bioactive compounds as compared to other terrestrial samples. The capability of the rmophilic microorganisms to flourish at high temperatures makes their enzyme systems ideal for various biotechnological applications. In this study, a total of 11 bacterial strains were isolated from Al-Lith hot spring which named Oyun Al-Haar, located about 17 km northeast of Gomika, at about 250 km south of Jeddah, Saudi Arabia. All these isolates were characterized and screenedfor some enzymes production. They were able to abundantly grow on starch nitrate agar medium and grew optimally at 45℃ with pH 7.0. According to bioinformatics analysis, the eleven bacterial isolates were encompassing 9 actinomycetes and 2 eubacteria namely, Streptomyces tendae (3 isolates) and only one isolate for each of Streptomyces mutabilis, Streptomyces chitinivorans, Streptomyces barkulensis, Leclerciaadecarboxylata, Streptomyces fradiae, Streptomyces azureus, Streptomyces macrosporus and Enterobacter cloacae. Based on the enzymatic activities, all isolates were positive for keratinase, gelatinase, chitinase, and lipase. Nine out of the eleven isolates showed protease production in good levels, and out of these11 isolates, 6 isolates exhibited remarkable amylase activity. Bacteria, especially actinomycetes isolated from hot springs area have gained commercial significance as source of thermostable enzymes. There arefew reports of enzymes production from the microflora of hot springs, which opens a window for exploring this resource as potential cache of novel strains with bioactive compounds.
Bahamdain L. A, Aba S. E. A, Sabry A, Amasha R. H, Noor S. O, Aly M. M. Molecular Identification and Phylogenetic Analysis of Some Rare Actinomycetes Obtained From Al-Lith Hot Spring Area of Saudi Arabia. Biosc.Biotech.Res.Comm. 2020;13(3).
Bahamdain L. A, Aba S. E. A, Sabry A, Amasha R. H, Noor S. O, Aly M. M. Molecular Identification and Phylogenetic Analysis of Some Rare Actinomycetes Obtained From Al-Lith Hot Spring Area of Saudi Arabia. Biosc.Biotech.Res.Comm. 2020;13(3). Available from: https://bit.ly/2Fz5bp8
Copyright © Bahamdain et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Secondary products play important roles in several sectors of our society because they are valuable for industrial, biotechnological and pharmacological uses in addition to treat human diseases, such as cancer and bacterial infections (Harvey, 2008). The natural products are largely produced from primary and secondary metabolism of plants and microorganisms (Jose and Jebakumar, 2013). Microbial natural products have made an incredible contribution to the diverse industrial applications (Salehghamari and Najafi, 2016). There are more than 22,000 known microbial secondary metabolites, 70% of which are produced by actinomycetes, 20% from fungi, 7% from Bacillus spp. and 1-2% by other bacteria (Subramani and Aalbersberg, 2013). The industrial enzyme market is one of the fastest growing revenue generating sectors in the world. Biocatalysis offers green and clean solutions to chemical processes and is emerging as a challenging and revered alternative to chemical technology. Only 20 enzymes are currently utilized on the industrial level indicating the need for further research and development of low-cost enzymes and their production (Prakash et al., 2013). Enzymes derived from microbial source are generally regarded as safe and they are functional at wide range of temperature, pH, salinity or other extreme conditions (Mukhtar et al., 2017).
Nowadays, the search for new bioactive secondary metabolites has switched to rare genera of actinomycetes from normal habitats or to discovery of strains/species found in unusual habitats. The logic behind these approaches is that such strains may be producers of novel bioactive compounds (Khanna et al., 2011).The unexplored and underexplored environments are promising sources of rare isolates that are believed to be rich sources of interestingly new compounds (Subramani and Aalbersberg, 2013).The extreme habitats are examples of unexplored environments and characterized by chemical or physical conditions that differ significantly from those found in environments that support more abundant and varied life forms (Zhao et al., 2011). Microorganisms have been detected in a variety of extreme environments (Merino et al., 2019).
In recent years, researchers have shown great interest in thermophilic bacteria because of their economic potential, either in useful biological processes such as biodegradation, or in the production of antibiotics and enzymes (Agarwal and Mathur, 2016). Thermophiles including bacterial and archaeal species are found in various geothermally heated regions of the earth such as hot springs (El-Gayar et al., 2017).Hot springs are defined as springs with water at temperatures substantially higher than the air temperature of the surrounding region (Mahajan and Balachandran, 2017). Consistent exposure to high temperatures, high free water content, high moisture content, and a typical chemical composition facilitates the growth of a typical range of hyper thermophilic microorganisms (Mahajan, 2015). Many microbial strains of following major groups/genera have been reported from hotsprings as sporulating and non-sporulating bacilli, actinomycetes and cyanobacteria (Jiang and Xu, 1993). In Saudi Arabia, there are about ten geothermal springs with varying deep temperatures and different flow rates. They are distributed in Gizan and Al-Lith areas (El-Gayar et al., 2017). The aim of the present investigation is the characterization of some thermophilic bacteria, especially actinomycetes isolated from Al-Lith hot spring area, Saudi Arabia in addition to the determination of their potential production of some important enzymes.
MATERIAL AND METHODS
Chemicals and media: Cultivation media and components were obtained from different companies: BDH Limited Poole (England), Biomatic Corporation (Canada), Difco Laboratories (USA), Fluka Chemie (Switzerland), Himedia (India), Holyland (Saudi Arabia), Merck (Germany), and Techno Pharmchem Haryana (India).
Sample collection :Samples from different six locations were obtained from Al-Lith hot spring, Saudi Arabia. The hot spring of Al-Lith which named Oyun Al-Haar (Figures 1) was located about 17 km northeast of Gomika, at about 250 km south of Jeddah at 20˚27’39.4776”N, 40˚28’14.7252”E. The samples (mainly from the area not tampered by human activities) were obtained from: a) soil sediments from surface and at a depth of 1-2 cm, b) water from hot spring, and c) scraping of the inner wall of small channels. The samples were collected aseptically using sterile bottles along with a sterile spatula, transported at 4◦C, and processed directly.
Isolation and purification of bacteria: The soil samples were air dried at room temperature for about 7 daysprior to inoculation onto isolation plates. Then, soil suspension of each sample was made separately by mixing 1g with 10 ml distilled water and vortexed for 2-5 min, then the mixtures could settle (Istianto et al., 2012). For each soil dilution, about 0.5 ml was taken (Al-Dhabi et al., 2016) and 0.1 ml was taking from hot spring water (Chaudhary and Prabhu, 2016).All these samples were separately spread evenly with a sterile glass rod onto starch nitrate agar medium (Shirling and Gottileb, 1966).Samples from the inner walls of hot spring were directly inoculated on starch nitrate agar medium by cotton swap. The inoculated plates were then incubated at 45°C for a week with presence of humidity (Chaudhary and Prabhu, 2016). After the incubation period, morphologically distinct colonies were chosen and sub-cultured to obtain pure isolates. The pure cultures were maintained on starch nitrate agar slants at 4◦C; for long-term storage, cultures were maintained in glycerol broth (50%, v/v) at -20◦C (Uzel et al., 2011). Taxonomic characterization and producing some important enzymes were studied for all isolates.
Figure 1. A: A map showing the sampling site at Al-Lith, B, C : The studied hot spring area, Gomika, Saudi Arabia.
Taxonomical studies of the isolates: Cultural characteristics of the isolates: Growth capability (heavy, moderate, scanty, or no growth), color of substrate and aerial mycelia, and presence of soluble pigment were examined using various media. These media were International Streptomyces Project (ISP) including yeast extract-malt extract agar (ISP-2) (Pridham et al., 1956, 1957), inorganic salts-starch agar (ISP-4) (Küster, 1959), tyrosine agar (ISP-7) (Shinobu, 1958), and starch nitrate agar (Shirling and Gottileb, 1966). The production of melanin pigment was examined using ISP-7 and dark brown to black color pigment means positive result (Srinivasan et al., 2016). In all previous media, the plates were incubated at 45℃for a week with presence of humidity. The color of substrate mycelia was described by the colors of RAL code (Vishwanatha et al., 2017).
Micromorphological characteristics of the isolates:Light microscope was used to observe the morphology of the isolates. Chitin agar plate was separately inoculated by the isolates and incubated at 45℃ for 7 days to examine by direct microscopic observation at 400X magnification (Caviedes et al., 2000).
Physiological characteristics of the isolates: To determine the optimal temperature and pH of all isolates, the growth capability was observed using incubated the isolates at different temperatures (45, 50 and 55°C) and at various pH (5, 6, 7, 8 and 9). The pH of the media was adjusted by adding 1N NaOH or 1N HCl solution (Al-Dhabi et al., 2016).
Biochemical characteristics of the isolates: Gram staining was carried out by using standard Gram reaction. Antibiotic susceptibility was examined on Muller-Hinton agar (Himedia, India) plates. The isolates were separately spread over the agar plates with disks containing different antibiotics such as amikacin (AK), ceftazidime (CAZ), aztreonam (ATM), piperacillin (PRL), imipenem (IMI), and ciprofloxacin (CIP). After incubation at 45℃ for 24-72 hr., the plates were examined for the presence of inhibition zones around the disc of antibiotic (Aly et al., 2012; Karp et al., 2017).
Molecular identification of the isolates: DNA extraction and PCR amplification
Genomic DNA extraction was performed at King Fahd Medical Research Centre, KAU according to Kumar et al., (2010) with some modifications. The purified isolates were grown for approximately 5 days on starch nitrate agar plates. In a clean tube, bacterial colonies of each isolate were separately mixed with 500 μl of TE buffer. This was followed by 3 cycles of heating in a water bath at 99◦C and then treating by liquid nitrogen (3 min in each per cycle). The crushed cells were treated with lysozyme (20 mg/ml) and incubated in a water bath at 37℃ for 30 min. After cell lysis with 10% SDS (w/v) and proteinase K at 55◦C for 30 min, the lysate was cooled, extracted for 5 min with a mixture of phenol: chloroform (1:1 v/v) at 10,000 rpm. In the aqueous phase, DNA was precipitated by adding 70-90% ethanol at -20◦C for 30 min. After centrifugation, the formed pellet was washed twice with 90% ethanol and dissolved in TE buffer. To obtain RNA free DNA, 20 μl of RNAase solution (20 μg/ml) was added and then incubated at 37◦C for 1 hr. The sample was once again extracted with equal volume of phenol: chloroform and precipitated as above. The purity and concentration of DNA was checked using NanoDrop 2000 spectrophotometer (Thermo Scientific).
The 16S rRNA was amplified using a thermal cycler (Applied Biosystems Veriti™ 96-Well Thermal Cycler). Amplification was performed using GoTaq® Green Master Mix, 2X (Promega) and two selected primers 27F (5′-AGAGTTTGATCMTGGCTCAG-3′) and 519R (5′- GWATTACCGCGGCKGCTG -3′) (Lane, 1991). PCR conditions consisted of an initial denaturation at 95◦C for 5 min; followed by 35 cycles of denaturation (94◦C for 40 sec), annealing (58◦C for 40 sec), and extension (72◦C for 1 min), and a final extension at 72◦C for 10 min. The amplification products were examined by 1% agarose gel electrophoresis and the gel was visualized by Ultra-Violet Product (UVP BioSpectrum ® Imaging System).
Bioinformatics analysis: The sequences of 11 isolates were subjected to NCBI BLAST search tool to detect non-chance sequence similarity. BLAST search was restricted to 16S ribosomal RNA sequences, where models (XM/XP) as well as uncultured/environmental samples were also filtered out, such that more reliable results would be attained. Each individual sequence was solely blasted, where the BLAST hit with the lowest expect-value (which indicates the number of non-chance alignments) was picked. In order to ensure that BLAST outputs were governed by expected-value (aka e-value), BLAST algorithm parameter was decreased such the expected threshold was set to more stringent value of 1e−6. Alignment of the 11 sequences was carried out using version 2 of Clustalx (Larkin et al., 2007).
Exploratory data and phylogenetic analyses were carried out under R Project for Statistical Computing (R Core Team, 2019) where exploratory data analysis was done using SeqinR package (Charif and Lobry, 2007). Phylogenetic analysis was carried out by ape package (Paradis et al., 2004). Reconstruction of the phylogenetic tree was done using neighbor-joining method (Nei, 1987). DNA Sequence Polymorphism (DnaSP) (Librado and Rozas, 2009) software was used to analyze the haplotype diversity (Hd), average number of nucleotide differences (Tajima, 1983), the nucleotide diversity (π), polymorphic sites (S), singleton variable sites (SP), parsimony-informative sites (PIP) for each gene, and the average number of nucleotide substitutions per site between species (Dxy) (Lynch and Crease, 1990).
Screening of the isolates for their extracellular enzymes production: Production of extracellular enzymes by potential isolates was studied by inoculated individually on different agar media at 45℃ for 7-14 days. Bacterial isolates were subjected to screen for amylase, protease, keratinase, gelatinase, chitinase, and lipase activity.For screening amylolytic activity, all isolates were individually grown on starch agar medium (Mohseni et al., 2013). After growth, the plates were flooded with iodine solution. Amylase production was detected as a zone of clearance around the colony on bluish black background due to starch digestion. Iodine forms a bluish black complex with starch but not with hydrolyzed starch (Singh et al., 2019). Indicator of hydrolysis of casein was assessed by growing the isolates individually on skim milk agar medium (Mohseni et al., 2013). Protease production was observed by appearing a clear zone around the colony in the medium (Das et al., 2010).
Regarding the screening for keratinase, feather meal agar medium (Agrahari and Wadhwa, 2010) was individually inoculated with the bacterial isolates. A clearing zone around the growth indicates that this isolate has keratinase.Gelatinase production by the isolates was tested using nutrient gelatin medium in test tubes (Ekpenyong et al., 2016). Partial or total gelatin liquefaction of the inoculated tubes even after exposure to cold temperature of refrigerator (4°C) was considered that these isolates produced gelatinase (Kole et al., 1998).
The bacterial cultures were further examined for their chitinase activity on plates containing chitin agar medium (Kim et al., 2003). The isolates that produced chitinase can grow on the medium and their growth depends, at least partially, on their ability to solubilize chitin. Also, appearance of a clear zone surrounding the colony on a creamish background was regarded that this isolate positive for chitinase activity (Sukalkar et al., 2017; Lacombe-Harvey et al., 2018). To determine lipase activity, all isolated bacteria were individually streaked on solid agar medium with Tween 80 as substrate (Niyonzima and More, 2013). The lipolysis was detected due to occurrence of a zone of clearance around the growth and subsequent formation of white precipitate of calcium monolaurate (Sierra, 1957; Cardenas et al., 2001) because of the combination of released fatty acids and Ca2+ ions (Bernal et al., 2015).
RESULTS
Isolation of actinomycetes: A total of 11 morphologically distinct colonies were obtained from Oyun Al-Haar hot spring using starch nitrate agar medium. The temperature of water in the spring ranged from 46°C to 75°C with pH 5.0-6.0. A total of 7 isolates, named from HL1 to HL7 were obtained from air-dried sediments, while isolates named HL8, HL9, and HL10 were obtained from inner wall of channels, and only one isolate HL11 was obtained from spring water.
| Soluble pigment | Color of substrate mycelium | Color of aerial mycelium | *Source of isolation | Growth | Isolate |
| no. | |||||
| – | Terra brown | Pale grey | Sediment 1 | Heavy | HL1 |
| – | Sepia brown | Grey | Sediment 1 | Heavy | HL2 |
| – | Terra brown | Grey | Sediment 1 | Heavy | HL3 |
| – | Signal yellow | White | Sediment 2 | Moderate | HL4 |
| – | Golden yellow | Pale grey | Sediment 2 | Heavy | HL5 |
| – | ND | Grey | Sediment 3 | Heavy | HL6 |
| – | Brown beige | Brown | Sediment 3 | Heavy | HL7 |
| – | Brown beige | Pale brown | Wall 1 | Heavy | HL8 |
| – | Olive brown | Dark green | Wall 2 | Heavy | HL9 |
| – | ND | Dark grey | Wall 1 | Heavy | HL10 |
| – | Terra brown | Grey | Water | Heavy | HL11 |
*Sediment 1 and sediment 3: at a depth of 1-2 cm; Sediment 2: from surface; -: Soluble pigment absent; ND: Not detected
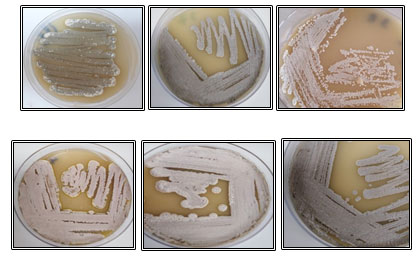
Cultural characterization of the isolates: From the result, it was indicated that cultural characteristics of the isolates on starch nitrate agar medium showed various colonies appearance and no soluble pigments were observed in the medium. Also, it was noticed that most isolates (7 isolates) had grey color in different degrees, 2 isolates were brown, one isolate was white and one isolate was green (Table 1 and Figure 2). Colonies characteristics of the isolates on isolation medium can be transformed into different appearance when the organism is sub-cultured on another growth medium. All isolates showed good growth (heavy or moderate) on starch nitrate agar; however, the growth on ISP-2, ISP-4, and ISP-7 plates was varied.
On ISP-2 medium, one isolate HL8 had heavy growth, one isolate HL6 showed moderate growth, 7 isolates HL1, HL2, HL3, HL7, HL9, HL10, and HL11 had poor growth, and isolates HL4 and HL5 showed no growth. On ISP-4 medium, the isolates were divided into two groups; the isolates HL2, HL3, HL6, HL7, HL8, HL9, HL10, and HL11 showed heavy growth and the isolates HL1, HL4, and HL5 showed moderate growth. The growth and melanin pigment production were recorded on IPS-7 (tyrosine agar). On the previous medium, the growth was heavy for isolate HL6, moderate for isolates HL1, HL7, HL8, and HL9, and scanty for these isolates HL2, HL3, HL4, HL5, HL10, and HL11, while melanin pigment was noticed only for isolate HL6.
Figure 2: Colonies morphology and color of some hot spring isolates grown on starch nitrate agar medium at 45°C for 7 days.
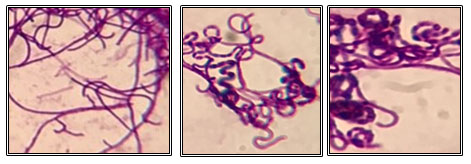
3.3Micromorphological characterization of the isolates: The morphology of the isolates was examined under a light microscope. All isolates, except isolates HL6 and HL10, were Gram-positive with filamentous hyphae (Figure 3). Isolates HL6 and HL10 were Gram-negative with rod-shaped.
Figure 3: Gram staining of some hot spring actinomycetes under a light microscope at 1000X magnification.
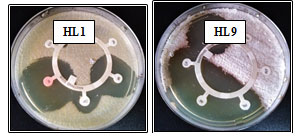
Physiological and biochemical characterization of the isolates: As shown in Table 2, the obtained data from physiological and biochemical examination showed that the optimal growth temperature for all isolates was 45°C. Out of 11 isolates, 6 isolates HL4, HL5, HL7, HL8, HL9, and HL10grew approximately up to 55°C and the isolates HL1, HL2, HL3, HL6, and HL11 could grow up to 50°C only. It was noticed that all isolates, except isolate HL5, grew well at pH between 5.0 and 9.0 and the pH growth range for isolate HL5 was 6.0-9.0. The optimum pH for all isolates was 7.0.Using antibiotic susceptibility pattern, the growth ability of the isolates was evaluated in the presence of six different antibiotics. All isolates except isolates HL4 and HL9recorded a clear sensitivity against amikacin, imipenem, and ciprofloxacin with different degrees. Meanwhile, isolates HL4 and HL9 demonstrated resistance against ceftazidime and aztreonam. Table 3 clarified antibiotic susceptibility profiles of all isolates. Figure 4 illustrated antibiotic susceptibility of the isolates HL1 and HL9.
Table 2. Some morphological and physiological characters of hot spring bacterial isolates.
| Result | Isolate no. | Tested character |
| + ve | Isolate HL6 | |
| Isolates HL1, HL2, HL3, HL4, HL5, HL7, HL8, HL9, HL10, and HL11 | Melanin production pigment on IPS-7 | |
| -ve | ||
| 45-50°C | Isolates HL1, HL2, HL3, HL6, and HL11 | Growth temperature range |
| Isolates HL4, HL5, HL7, HL8, HL9, and HL10 | ||
| 45-55°C | ||
| 6.0-9.0 | Isolate HL5 | |
| 5.0-9.0 | Isolates HL1, HL2, HL3, HL4, HL6, HL7, HL8, HL9, HL10, and HL11 | pH range |
Table 3. Antibiotic susceptibility of hot spring bacterial isolates to some antibiotics.
| *Resistance to antibiotics | ||||||
| Isolate | ||||||
| no. | AK | CAZ | ATM | PRL | IMI | CIP |
| HL1 | S | R | R | R | S | S |
| HL2 | S | R | R | R | S | S |
| HL3 | S | R | R | R | S | S |
| HL4 | S | R | R | S | S | S |
| HL5 | S | R | R | R | S | S |
| HL6 | S | R | R | R | S | S |
| HL7 | S | R | R | R | S | S |
| HL8 | S | R | R | R | S | S |
| HL9 | S | R | R | S | S | S |
| HL10 | S | R | R | R | S | S |
| HL11 | S | R | R | R | S | S |
S: sensitive (20 mm or more)., R: resistance Resistant (16 mm or less), *(AK): Amikacin; (CAZ): Ceftazidime; (ATM):
Aztreonam; (PRL): Piperacillin; (IMI): Imipenem; (CIP): Ciprofloxacin.
Figure 4: Antibiotic resistance patterns of the two isolates HL1 and HL9, AK: Amikacin; CAZ: Ceftazidime; ATM: Aztreonam; PRL: Piperacillin; IMI: Imipenem; CIP: Ciprofloxacin.
Bioinformatics analysis of the data: NCBI BLAST query: Table 4 showed results of NCBI BLAST query for the 11 sequenced isolates. The criteria used for query sequence aimed to narrow down the search space (database), such that a smaller the database has more likely to contain the sequence of interest. Therefore, search query was restricted to 16S ribosomal RNA sequences. For all the 11 queries, zero e-values were attained indicated that all alignments were non-chance alignments. The percentages of query coverage ranged from 27 to 96%, where identities % were also high which ranged from 95 to 100%.
The results of BLAST query indicated that 11 strains of bacteria encompassing 9 actinomycetes and 2 eubacteria namely, Streptomyces tendae (3 isolates) and only one isolate for each of Streptomyces mutabilis, Streptomyces chitinivorans, Streptomyces barkulensis, Leclerciaa decarboxylata, Streptomyces fradiae, Streptomyces azureus, Streptomyces macrosporus and Enterobacter cloacae.
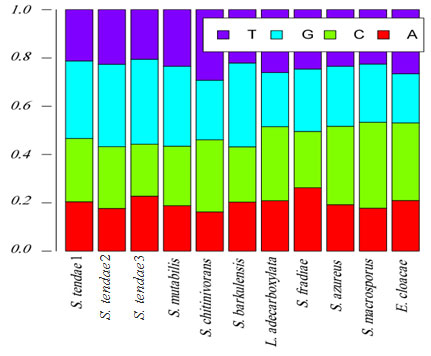
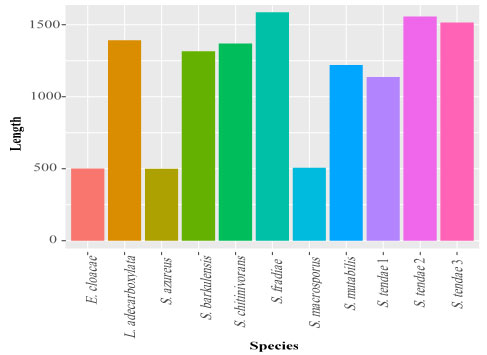
Exploratory data analysis: The base frequencies of the 11 isolates were presented in Figure 5. Base frequencies varied greatly among the 11 isolates, this may be result of great fluctuation in sequence lengths (Figure 6). Sequence length ranged from 499 for S. azureus to 1587 base pair (bp) for S. fradiae (Table 5). Similarly, GC percentage (%) also varied greatly, where GC% ranged from 49% for S. fradiae to 60% for S. tendae 2.
Table 4.Results of NCBI BLAST query for the 11 sequences isolated from hot spring.
| Isolate no. | Species | Query | Identity (%) |
| coverage (%) | |||
| HL1 | Streptomyces tendae1 | 40 | 99 |
| HL2 | Streptomyces tendae2 | 30 | 99 |
| HL3 | Streptomyces mutabilis | 36 | 100 |
| HL4 | Streptomyces chitinivorans | 34 | 97 |
| HL5 | Streptomyces barkulensis | 34 | 98 |
| HL6 | Leclerciaadecarboxylata | 34 | 97 |
| HL7 | Streptomyces fradiae | 27 | 100 |
| HL8 | Streptomyces azureus | 90 | 96 |
| HL9 | Streptomyces macrosporus | 85 | 100 |
| HL10 | Enterobacter cloacae | 96 | 95 |
| HL11 | Streptomyces tendae3 | 30 | 99 |
Figure 5: Base frequencies of hot spring bacterial isolates.
Figure 6: Sequence lengths of hot spring bacterial isolates.
Table 5. Sequence lengths and GC contents of hot spring bacterial isolates.
| *GC content | Sequence length | Species
|
| 0.58 | 1138 | S. tendae1 |
| 0.60 | 1556 | S. tendae2 |
| 0.57 | 1515 | S. tendae3 |
| 0.58 | 1219 | S. mutabilis |
| 0.54 | 1370 | S. chitinivorans |
| 0.58 | 1315 | S. barkulensis |
| 0.53 | 1392 | L. adecarboxylata |
| 0.49 | 1587 | S. fradiae |
| 0.57 | 499 | S. azureus |
| 0.60 | 506 | S. macrosporus |
| 0.52 | 501 | E. cloacae |
*GC content: Guanine +
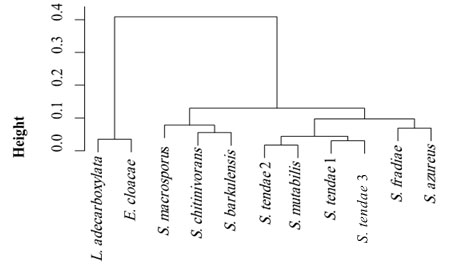
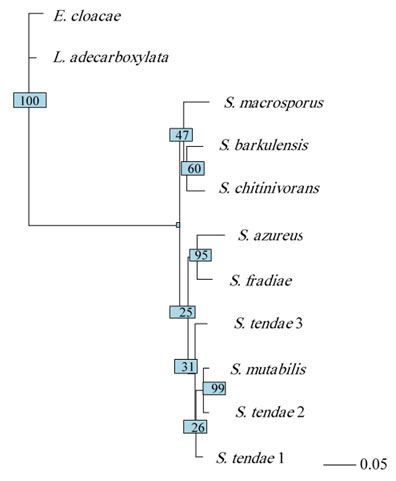
Cluster and phylogenetic analysis: Cluster analysis was carried out as pre-processing step to glean an insight into the data distribution. Results of cluster analysis were shown graphically in Figure 7 and Table 6. The resulted dendrogram comprised of 2 large clusters, where Leclercia and Enterobacter species clustered in one cluster and Streptomyces species clustered in the second cluster. The evolutionary history of the 11 isolates was inferred based on neighbor-joining method (Figure 8). The optimal tree with the sum of branch length = 2.5 is shown. The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances were computed using the Maximum Composite Likelihood method (Tamura et al., 2004) and were expressed as the number of base substitutions per site. This analysis involved 11 nucleotide sequences. All ambiguous positions were removed for each sequence pair (pairwise deletion option). There were a total of 1650 positions in the final dataset.
Polymorphism and genetic diversity among species: General information about the polymorphisms on the 11 isolates was found in Table 7. The number of sites was 1650, the number of monomorphic informative sites was 296 sites, and the number of polymorphic sites was 175. Of polymorphic sites, 151 sites were parsimony-informative sites (i.e. sites that have a minimum of two nucleotides that are present at least twice) and 24 sites were singletons. The 11 sequences were also analyzed to characterize the sequence diversity. The results of the analysis were presented in Table 8, the haplotype diversity was 1±0.04 and nucleotide diversity was only 0.15, where the average number of nucleotide differences was 68.6. Only one conserved region was found among the 11 isolates in the region between 18 to 552. Measurements of conservation (C)=0.59, homozygosity =0.82 and P-value <0.001. Conservation (C) was calculated as the proportion of conserved site in the alignment region, where homozygosity was measured as 1- heterozygosity.
Figure 7: Cluster analysis of hot spring bacterial isolates.
Table 6. Distribution of hot spring bacterial isolates in 3 clusters.
| Clusters |
Species |
||
| 3 | 2 | 1 | |
| 1 | 0 | 0 | E. cloacae |
| 1 | 0 | 0 | L. adecarboxylata |
| 0 | 0 | 1 | S. azureus |
| 0 | 1 | 0 | S. barkulensis |
| 0 | 1 | 0 | S. chitinivorans |
| 0 | 0 | 1 | S. fradiae |
| 0 | 1 | 0 | S. macrosporus |
| 0 | 0 | 1 | S. mutabilis |
| 0 | 0 | 3 | S. tendae |
Figure 8: Neighbor-joining phylogenetic tree of hot spring bacterial isolates.
Table 7. Estimated parameters of the polymorphic sites of hot spring bacterial isolates.
| Singleton
variable sites |
Parsimony–
informative sites |
No. of polymorphic
sites |
No. of monomorphic
informative sites |
No. of sites |
| 24 | 151 | 175 | 296 | 1650 |
Table 8. Estimated parameters of DNA polymorphism of hot spring bacterial isolates.
| Average number of
nucleotide differences |
Nucleotide
diversityπ |
Haplotype
diversity±sd |
No. of
Haplotypes |
| 68.6 | 0.15 | 1±0.04 | 11 |
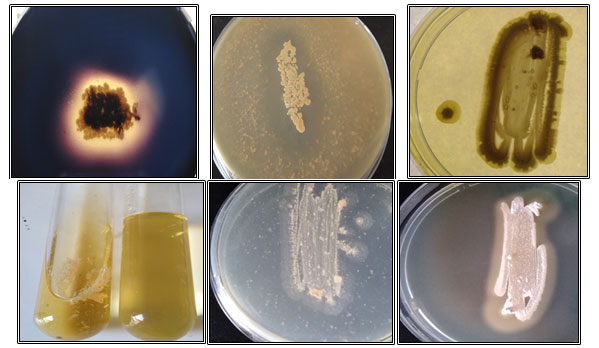
Screening for enzymes production : From the results (Table 9), it was noticed that all isolates produced keratinase, gelatinase, chitinase, and lipase. Out of 11 isolates, 6 isolates HL1, HL6, HL8, HL9, HL10, and HL11 produced amylase, while isolates HL2, HL3, HL4, HL5, and HL7 showed weak amylase production. On skim milk agar medium, all isolates showed protease production, but the isolates HL1 and HL2 exhibited protease activity in very low levels. Some results for enzymes production were depicted in Figure 9.
Table 9. Enzymes production by bacterialisolates.
| Enzyme
production |
Isolate no. | Enzyme activities |
| +ve | Isolates HL1, HL6, HL8, HL9, HL10, and HL11 | Amylase |
| -ve | Isolates HL2, HL3, HL4, HL5, and HL7 | |
| +ve | Isolates HL3, HL4, HL5, HL6, HL7, HL8, HL9, HL10, and HL11 | Protease |
| -ve | Isolates HL1and HL2 | |
| +ve | Isolates HL1, HL2, HL3, HL4, HL5, HL6, HL7, HL8, HL9, HL10, and HL11 | keratinase
Gelatinase Chitinase Lipase |
+ve: Enzyme production; -ve: Very weak production.
Figure 9: Enzymes production by hot spring isolates on different agar media, (A): Starch hydrolysis produced by isolate HL9, (B): A clear zone of skim milk hydrolysis by isolate HL4, (C): Keratinase degradation by isolate HL8, (D):Gelatin liquefaction by isolate HL3, (E): Chitinase production by isolate HL8and (F): Lipolytic activity of isolate HL2.
DISCUSSION
Factors such as pH, temperature, pressure, salt and nutrient concentrations, water availability, radiation, harmful heavy metals, and toxic compounds are used in defining extreme habitats (Satyanarayana et al., 2005; Procopio et al., 2016). Microorganisms, including actinomycetes, from extreme environments have attracted a great deal of attention, due to their special mechanisms of adapting to the extreme conditions and because they can produce unusual compounds (Meklatet al., 2011). It is postulated that extremotolerants may have larger genetic and metabolic plasticity (Mohammadipanah and Wink, 2016).
Studies on the microbial potential of extreme environments can be utilized to produce novel enzymes that can become future harbingers of green biotechnology (Mukhtar et al., 2017).It is possible to consider thermophiles as sources of industrially relevant thermostable enzymes. Many industrial processes require enzymes such as amylase, cellulase, xylanase, pectinase, protease, and lipase that are operationally stable at high temperatures (Haki and Rakshit, 2003; Bouzaset al., 2006).Thermophilic bacteria and their hydrolytic enzymes have become a chief research subject due to their valuable biotechnological applications(El-Gayaret al., 2017).Thermophilic actinomycetes play an important role in habitats where decomposition of organic matter takes place at elevated temperatures and under aerobic conditions. Using these isolates for direct hydrolysis agro-industrial waste and in biofuel generation also seems a promising strategy (Chaudhary and Prabhu, 2016).
This research aimed to find novel bacteria, especially actinomycetes that produced some useful enzymes from poorly studied extreme habitat, hot spring ecosystem in Saudi Arabia. In this study, a total of 11 bacterial strainswere successfully isolated from sediments, walls, and water samples collected from Al-Lithhot spring named Oyun Al-Haar. This spring is located about 17 km northeast of Gomika, at about 250 km south of Jeddah, Saudi Arabia. From the results, it was noticed that numbers of bacteria obtained from hot spring walls or water were lower than sediment samples. Some previous studies reported similar findings. Khiyamiet al. (2012) and El-Gayar et al. (2017) recorded that the total count of bacteria in hot spring water was low. Also, Chaudhary and Prabhu (2016) reported that the sediment sample from hot spring showed a good diversity of actinomycete colonies, while the water sample had only two types. Akmaret al. (2011) found that isolation rate of actinomycetes from hot spring was higher from sediment and biomat than from water sample.
In the current study, taxonomic characterization of the isolates by morphological, physiological, and biochemical characters were studied. Sequencing of the ribosomal RNA gene of bacteria followed by bioinformatics studies were used to identify the bacteria as reported in previous studies (Caverly et al., 2015; Alsanie et al., 2018). In this study, bioinformatics analysis was performed for 11 hot spring isolates to determine the evolutionary and phylogenetic relationships among the isolates. The following strains were identified from the isolates: Streptomyces tendae (3 isolates) and only one isolate for each of Streptomyces mutabilis, Streptomyces chitinivorans, Streptomyces barkulensis, Leclerciaadecarboxylata, Streptomyces fradiae, Streptomyces azureus, Streptomyces macrosporus, and Enterobacter cloacae.
The genus Streptomyces is classified in the family Streptomycetaceae and has received attention for three main reasons. First, streptomycetes are abundant and important in the soil, where they play major roles in the cycling of carbon trapped in insoluble organic debris, particularly from plants and fungi. This action is enabled by the production of diverse hydrolytic exoenzymes (Barka et al., 2016). Second, the genus exhibits a wide phylogenetic spread (Aderem, 2005).Thirdly, streptomycetes produce a stunning diversity of bioactive secondary metabolites; consequently, they are of great interest in medicine and industry (Hopwood, 2007).
Leclercia adecarboxylata is a Gram-negative bacillus belonging to the family Enterobacteriaceae. It is mainly isolated from environmental or animal specimens but has been recognized as an emerging opportunistic pathogen, with the potential to cause severe infections in immunocompromised patients (Spiegelhauer et al., 2019). L. adecarboxylata is a member of the normal gut flora in animals (Hess et al., 2008). Enterobacter cloacae is a Gram-negative, facultative anaerobic, rod-shaped, non-spore-forming bacteria belonging to the family Enterobacteriaceae. Species of the E. cloacae complex are widely encountered in nature, but they are also pathogens. E. cloacae has taken on clinical significance as opportunistic bacterium and has emerged as nosocomial pathogens from intensive care patients pathogenic, especially to those who are on mechanical ventilation (Mezzatesta et al., 2012).
The bioinformatics analysis results in this study revealed that there was a high variability of sequence lengths between and within species, some species contained long sequences, whereas other species contained short sequences. Furthermore, GC contents were highly variable among the target species. Phylogenetic tree construction of isolated hot spring bacterial strains was done using neighbor-joining method (Nei, 1987). Sequence variations and evolutionary distances were calculated using the Maximum Composite Likelihood method (Tamura et al., 2004). Similarly, same phylogenetic analysis methods were applied by Alsanie et al. (2018) for identification of isolated multidrug-resistant (MDR) bacterial strains.
Molecular identificationof the isolates demonstrated low diversity of thermophilic bacteria. Out of 11 isolates, 9 strains were belonging to the genus Streptomyces, one isolate was identified as Leclerciaa decarboxylata, and another isolate was identified as Enterobacter cloacae. Similarly, Aannizet al. (2015) recorded a very low number of thermophilic bacteria in four hot springs in Morocco. In 2012, Khiyami and coauthorshave identified about 15 thermo-aerobic bacteria from Jazan and Al-Lith geothermal springs where Bacillus cereus, Bacillus licheniformis, Bacillus thermoamylovorans, Pseudomonas aeruginosa and Enterobacter sp. were the dominant strains. The low density of the bacterial populations in hot springs could be attributed to the adverse conditions of these environments or due to the application of culture-dependent identification approaches that identify only a small portion of the total microbial communities (Ranjard et al., 2000). In contrast to the results obtained from this study, 73 strains of bacteria encompassing 8 actinomycetes and 65 eubacteria were isolated and purified from the four sampling points of Vajreshwari-Ganeshpurihot springs, India. The isolates reflected the diversity with respect to macroscopic and microscopic characteristics. Twenty-four were Gram-positive and 49 were of Gram-negative bacteria. About 11% of the isolates were actinomycetes (Pednekaret al., 2011).
Valverde et al. (2012) studied actinobacteria in the hot springs in Zambia, China, New Zealand, and Kenya and observed around 28 major operational taxonomic units (OTUs). They opined that the actinobacterial diversity and endemism were very high in hot spring ecosystems. In addition, several strains of actinomycetes have been previously reported from hot springs (Thawai, 2012; Comanet al., 2015; Kamburaet al., 2016). Duanet al. (2014) isolated a novel species Streptomyces calidiresistens from a sediment sample collected from Hehua hot spring in China. Regarding enzymes production in this study, all hot spring bacteria showed positive for keratinase, gelatinase, chitinase, and lipase activity. However, out of the total isolates, only 6 isolates exhibited highly amylase activity and 9 isolates showed good protease production.
From hot springs in Gazan, Saudi Arabia, two thermophilic bacteria Brevibacterium linens and Bacillus subtilis were isolated and showed the capability to produce amylolytic and proteolytic enzymes (El-Gayaret al., 2017).
Another study was conducted with Khalil (2011) who isolated 13 thermophilic bacteria from hot springs in Saudi Arabia. Based on the biochemical characterization, all the isolates were lipase positive, 11 isolates showed amylase activity, while only 3 of them showed cellulase activity. Bacilli like Thermus aquaticus andThermus brockianus, from hotsprings have gained commercial significance as source of thermostable enzymes(Brock and Freeze, 1969; Breithaupt, 2001).Thermophilic actinobacteria are biotechnologically important producers of several enzymes such as DNA polymerases, pullulanases, amylases, xylanases, lipases, and proteases (Panda et al., 2017). Chaudhary and Prabhu (2016) isolated species of Streptomyces from hot spring water, and these strains produced remarkable amount of thermostable amylase and cellulase with temperature optimum at 55℃. The thermophilic Streptomyces Al-Dhabi-2 strain isolated from hot spring produced various enzymes such as amylase, gelatinase, and deoxyribonuclease (DNase) (Al-Dhabi et al., 2019).
CONCLUSION
In conclusion, hot spring environments are underexplored microbiologically and should not be overlooked for the search and discovery of novel bacteria and their chemical diversity of useful compounds. Studies on unique ecological environments could yield enzymes that could be of great commercial importance in near future. The results of this investigation revealed that Al-Lith hot spring named Oyun Al-Haar was potent source of thermophilic bacteria with hydrolytic enzymes production that can catalyze various reactions at high temperatures. Hence, these isolates can be further evaluated and studied in detail for commercial scale production of enzymes to use in various pharmaceutical and industrial applications. Evidently, more studies on optimal cultivation techniques for thermophiles are required, not only for basic research but also for profiling of their unique microbial products.
REFERENCES
Aanniz, T., Ouadghiri, M., Melloul, M., Swings, J., Elfahime, E., Ibijbijen, J. and Amar, M. (2015). Thermophilic bacteria in Moroccan hot springs, salt marshes and desert soils. Braz J Microbiol, 46(2):443-453.
Aderem, A. (2005). Systems biology: its practice and challenges. Cell, 121:511-513.
Agarwal, A. and Mathur, N. (2016). Thermophilic actinomycetes are potential source of novel bioactive compounds: a review. Ejpmr, 3(2):130-138.
Agrahari, S. and Wadhwa, N. (2010). Degradation of chicken feather a poultry waste product by keratinolytic bacteria isolate from dumping site at Ghazipur poultry processing plant. Int J Poult Sci, 9:482-489.
Akmar, H.N., Asma, I., Venugopal, B., Latha, L.Y. and Sasidharan, S. (2011). Identification of appropriate sample and culture method for isolation of new thermophilic bacteria from hot spring. Afr J Microbiol Res, 5:217-21.
Al-Dhabi, N.A., Esmail, G.A., Duraipandiyan, V., Arasu, M.V. and Salem-Bekhit, M.M. (2016) Isolation, identification and screening of antimicrobial thermophilic Streptomyces sp. Al-Dhabi-1 isolated from Tharban hot spring, Saudi Arabia. Extremophiles, 20(1):79-90.
Al-Dhabi, N.A., Esmail, G.A., Duraipandiyan, V., Valan, M. and Arasu, M.V. (2019). Chemical profiling of Streptomyces sp. Al-Dhabi-2 recovered from an extreme environment in Saudi Arabia as a novel drug source for medical and industrial applications. Saudi J Biol Sci, 26(4):758-766; doi:10.1016/j.sjbs.2019.03.009.
Alsanie, W.F., Felemban, E.M., Farid, M.A., Hassan, M.M., Sabry, A. and Gaber, A. (2018). Molecular identification and phylogenetic analysis of multidrug-resistant bacteria using 16S rDNA sequencing. Pure Appl Microbiol, 12(2):489-496.
Aly, M.M., Tork, S., Al-Garni, S. and Nawar, L. (2012). Production of lipase from genetically improved Streptomyces exfoliates LP10 isolated from oil contaminated soil. Afr J Microbiol Res, 6(6):1125-1137.
Barka, E.A., Vatsa, P., Sanchez, L., Gaveau-Vaillant, N., Jacquard, C., Meier-Kolthoff, J.P., Klenk, H.P., Clément, C., Ouhdouch,Y. and van Wezel, G.P. (2016). Taxonomy, physiology, and natural products of actinobacteria. Microbiol Mol Biol Rev, 80(1):1-43; doi: 10.1128/MMBR.00019-15.
Bernal, M.G., Campa-Córdova, Á.I., Saucedo, P.E., González, M.C., Marrero, R.M. and Mazón-Suástegui, J.M. (2015). Isolation and in vitro selection of actinomycetes strains as potential probiotics for aquaculture. Vet world, 8(2), 170-176; doi:10.14202/vetworld.2015.170-176.
Bouzas, T.M., Barros-Velázquez, J. and Villa, T.G. (2006). Industrial applications of hyperthermophilic enzymes: a review. Protein Pept Lett,13:645-651.
Breithaupt, H. (2001). The hunt for living gold: The search for organisms in extreme environments yields useful enzymes for industry. EMBO Rep,2:968-71.
Brock, T.D. and Freeze H. (1969). Thermus aquaticus gen. n. and sp. n., a non-sporulating extreme thermophile. Bacteriol,98:289-97.
Cardenas, J., Alvarez, E., de Castro Alvarez, M.S., Sanchez-Montero, J.M., Valmaseda, M., Elson, S.W. and Sinisterra, J.V. (2001). Screening and catalytic activity in organic synthesis of novel fungal and yeast lipase. Mol Catal B: Enz, 14:111-123.
Caverly, L.J., Zhao, J. and LiPuma, J.J. (2015). Cystic fibrosis lung microbiome: opportunities to reconsider management of airway infection. Pediatr Pulmonol, 50 (Suppl 40): S31-8.
Caviedes, L., Lee, T.S., Gilman, R.H. and Sheen, P. (2000). Rapid, efficient detection and drug susceptibility testing of Mycobacterium tuberculosis in sputum by microscopic observation of broth cultures. Clinic Microbiol, 38(3):1203-8.
Charif, D. and Lobry, J.R. (2007). SeqinR 1.0-2: a contributed package to the R project for statistical computing devoted to biological sequences retrieval and analysis. in: Bastolla, U., Porto, M., Roman, H.E. and Vendruscolo, M. (eds) Structural Approaches to Sequence Evolution. Biological and Medical Physics, Biomedical Engineering,p 207-232, Berlin, Heidelberg: Springer.
Chaudhary, N. and Prabhu, S. (2016). Thermophilic actinomycetes from hot water spring capable of producing enzymes of industrial importance. Int J Res Stud Biosci (IJRSB), 4(6):29-35.
Coman, C., Chiriac, C.M., Robeson, M.S., Ionescu, C., Dragos, N., Barbu-Tudoran, L., et al. (2015). Structure, mineralogy, and microbial diversity of geothermal spring metabolites associated with deep oil drilling in Romania, Front Microbiol, 6:253.
Das, S., Ward, L.R. and Burke, C. (2010). Screening of marine Streptomyces spp. for potential use as probiotics in aquaculture. Aquaculture, 305(1):32-41.
Duan, Y.Y., Ming, H., Dong, L., Yin, Y.R., Zhang, Y., Zhou, E.M., Liu, L., Nie, G.X. and Li, W.J. (2014). Streptomyces calidiresistens sp. nov., isolated from a hot spring sediment. Antonie van Leeuwenhoek, 106(2):189-196.
Ekpenyong, M., Asitok, A., Odey, A. and Antai, S. (2016). Production and activity kinetics ofgelatinase by Serratia sp. SLO3. Nig J Biopestic, 1:70-82.
El-Gayar, K.E., Al-Abboud, M.A. and Essa, A.M.M. (2017). Characterization of thermophilic bacteria Isolated from two hot springs in Jazan, Saudi Arabia. Pure Appl Microbiol, 11:743-752.
Haki, G.D. and Rakshit S.K. (2003). Developments in industrially important thermostable enzymes: a review. Bioresour Technol, 89:17-34.
Harvey, A.L. (2008). Natural products in drug discovery. Drug DiscovToday, 13:894-901;doi:10.1016/j.drudis.2008.07.004.
Hess, B., Burchett, A. and Huntington, M.K. (2008). Leclerciaa decarboxylata in an immunocompetent patient. Med Microbiol,57:896-898.
Hopwood, D.A. (2007). Streptomyces in nature and medicine: the antibiotic makers. New York: Oxford University Press.
Istianto, Y., Koesoemowidodo, R.S.A., Saputra, H., Watanabe, Y., Pranamuda, H. and Marwoto, B. (2012). Application of phenol pretreatment for the isolation of rare actinomycetes from Indonesian soil. Microbiol (Indonesia), 6(1):42-47.
Jiang, C. and Xu, L. (1993). Actinomycete diversity in unusual habitats. Actinomycetes,4:47-57.
Jose, P.A. and Jebakumar, S.R.D. (2013). Non-streptomycete actinomycetes nourish the current microbial antibiotic drug discovery. Front Microbiol, 4:240; 10.3389/fmicb. 2013.00240.
Kambura, A.K., Mwirichia, R.K., Kasili, R.W., Karanja, E.N., Makonde, H.M. and Boga, H.I. (2016). Bacteria and archaea diversity within hot springs of Lake Magadi and Little Magadi in Kenya, BMC Microbiol, 16(1):136.
Karp, B.E., Tate, H., Plumblee, J.R., Dessai, U., Whichard, J.M., Thacker, E.L., Hale, K.R., Wilson, W., Friedman, C.R., Griffin, P.M. and McDermott, P.F. (2017). National antimicrobial resistance monitoring system: two decades of advancing public health through integrated surveillance of antimicrobial resistance. Foodborne Pathog Dis,14(10):545-557.
Khalil, A. (2011). Screening and characterization of thermophilic bacteria (lipase, cellulase and amylase producers) from hot springs in Saudi. Arabia. Food Agric Environ, 9(2): 672-675.
Khanna, M., Solanki, R. and Lal, R. (2011). Selective isolation of rare actinomycetes producing novel antimicrobial compounds. Int J Adv Biotech Res, 2(3):357-375.
Khiyami, M.A., Serour, E.A., Shehata, M.M. and Bahklia, A.H. (2012). Thermo-aerobic bacteria from geothermal springs in Saudi Arabia. Afr J Biotechnol, 11(17):4053-4062.
Kim, K.J., Yang, Y.J. and Kim, J.G. (2003). Purification and characterization of chitinase from Streptomyces sp. M-20. Biochem Mol Biol, 36 (2):185-189.
Kole, M.M., Draper, I. and Gerson, D.F. (1998). Production of Protease by Bacillus subtilis using simultaneous control of glucose and ammonium concentrations. Chem Technol Biotechnol, 41:197-206.
Kumar, V., Bisht, G.S. and Institu, S.B.S.P.G. (2010). An improved method for isolation of genomic DNA from filamentous actinomycetes. Sci Eng Technol Mgt, 2:10-13.
Küster, E. (1959). Outline of a comparative study of criteria used in characterization of the actinomycetes. Int Bull Bact Nomen Taxon, 9:98-104.
Lacombe-Harvey, M.È., Brzezinski, R. and Beaulieu, C. (2018). Chitinolytic functions in actinobacteria: ecology, enzymes, and evolution. Appl Microbiol Biotechnol, 102:7219-30.
Lane, D.J. (1991). 16S/23S rRNA sequencing, in: Stackebrandt, E. and Goodfellow, M. (eds) Nucleic Acid Techniques in Bacterial Systematics, p. 115-175, New York: John Wiley & Sons.
Larkin, M.A., Blackshields, G., Brown, N.P., Chenna, R., McGettigan, P.A., McWilliam, H., Valentin, F., Wallace, I.M., Wilm, A., Lopez, R., Thompson, J.D., Gibson, T.J. and Higgins, D.G. (2007). Clustal W and Clustal X version 2.0. Bioinform, 23(21):2947-2948.
Librado, P. and Rozas, J. (2009). DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinform, 25(11):1451-1452.
Lynch, M. and Crease, T.J. (1990). The analysis of population survey data on DNA sequence variation. Mol Biol Evol, 7(4):377-394.
Mahajan, G.B. (2015). Antibiotics from micro-organisms from hot springs/geysers,in:Sánchez, S. and Demain, A.L. (eds)Antibiotics: current Innovations and Future Trends, p. 205-212, USA: Caister Academic Press.
Mahajan, G.B. and Balachandran, L. (2012). Antibacterial agents from actinomycetes – a review. Front Biosci (Elite Ed), 1:240-253; doi:10.2741/ e373.
Meklat, A., Sabaou, N., Zitouni, A., Mathieu, F. and Lebrihi, A. (2011). Isolation, taxonomy, and antagonistic properties of halophilic actinomycetes in Saharan soils of Algeria. Appl Environ Microbiol, 77:6710-6714; doi: 10.1128/AEM.00326-11.
Merino, N., Aronson, H.S., Bojanova, D.P., Feyhl-Buska, J., Wong, M.L., Zhang, S. and Giovannelli, D. (2019). Living at the extremes: extremophiles and the limits of life in a planetary context. Front Microbiol, 10:1785.
Mezzatesta, M.L., Gona, F. and Stefani, S. (2012). Enterobacter cloacae complex: clinical impact and emerging antibiotic resistance. Future Microbiol, 7:887-902.
Mohammadipanah, F. and Wink, J. (2016). Actinobacteria from arid and desert habitats: diversity and biological activity. Front Microbiol, 6:Article 1541; 10.3389/fmicb.2015.01541.
Mohseni, M., Norouzi, H., Hamedi, J. and Roohi, A. (2013). Screening of antibacterial producing actinomycetes from sediments of the caspian sea. Int J Mol Cell Med, 2(2):64-71.
Mukhtar, S., Zaheer, A., Aiysha, D., Malik, K.A. and Mehnaz, S. (2017). Actinomycetes: a source of industrially important enzymes. Proteomics Bioinform, 10:12.
Nei, M. (1987). Molecular Evolutionary Genetics. New York: Columbia University Press.
Niyonzima, F.N. and More, S.S. (2013). Screening and identification of a novel alkaline lipase producing bacterium. Int J Pharma Bio Sci, 4(2):1037-1045.
Panda, A.K., Bisht, S.S., Kaushal, B.R., Mandal, S.D., Kumar, N.S. and Basistha, B.C. (2017). Bacterial diversity analysis of Yumthang hot spring, North Sikkim, India by Illumina sequencing. Big Data Anal, 2:7; doi.org/10.1186/s41044-017-0022-8.
Pednekar, P., Jain, R. and Mahajan, G. (2011). Anti-infective potential of hot-spring bacteria. Global Infect Dis, 3(3):241-245; doi:10.4103/0974-777X.83529.
Paradis, E., Claude, J. and Strimmer, K. (2004). APE: analyses of phylogenetics and evolution in R language. Bioinform, 20(2):289-290.
Prakash, D., Nawani, N., Prakash, M., Bodas, M., Mandal, A., Khetmalas, M. and Kapadnis, B. (2013). Actinomycetes: a repertory of green catalysts with a potential revenue resource. Bio Med Res Int, 2013:1-8.
Pridham, T.G., Anderson, P., Foley, C., Lindenfelser, L.A., Hesseltine, C.W. and Benedict, R.G. (1956-57). A selection of media for maintenance and taxonomic study of streptomycetes. Antibiot Ann, 947-953.
Procopio, R.E., Silva, I.R., Martins, M.K., Azevedo, J.L. and Ara, J.M. (2016). Antibiotics produced by Streptomyces. Braz J Infect Dis, 16:466-471.
Ranjard, L., Poly, F. and Nazaret, S. (2000). Monitoring complex bacterial community using culture-independent molecular techniques: application to soil environment. Res Microbiol, 151:167-177.
R Core Team (2019). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Salehghamari, E. and Najafi, M. (2016). Isolation of biologically active actinomycetes from untouched soils: a case study from Karaj district, Iran. Prog Biol Sci, 6(1): 65-74; doi:10.22059/pbs.2016.59009.
Satyanarayana, T., Raghukumar, C. and Shivaji, S. (2005). Extremophilic microbes: diversity and perspectives. Curr Sci, 89(1):78-90.
Shinobu, R. (1958). Physiological and cultural study for the identification of soil actinomycetes species. Mem Osaka Univ Arts Educ B. Natur Sci, 7:l-76.
Shirling, E.B. and Gottlieb, D. (1966). Methods for characterization of Streptomyces species. Int J Syst Bacteriol, 16:313-340.
Sierra, G. (1957). A simple method for the detection of lipolytic activity of microorganisms and some observations on the influence of the contact between cells and fatty substrates. Antonie van Leeuwenhoek, 23:15-22.
Singh, L., Sharma, H. and Sahoo, D. (2019). Actinomycetes from soil of Lachung, a pristine high altitude region of Sikkim Himalaya, their antimicrobial potentiality and production of industrially important enzymes. Adv Microbiol, 9:750-773;doi:10.4236/aim.2019.98046.
Spiegelhauer, M.R., Andersen, P.F., Frandsen, T.H., Nordestgaard, R.L.M. and Andersen, L.P. (2019). Leclerciaa decarboxylata: a case report and literature review of 74 cases demonstrating its pathogenicity in immunocompromised patients. Infect Dis,51:179-188.
Srinivasan, R., Mohan, V., Amaravathy, K., Devi, K. S. and Ramprasath, C. (2016). Molecular characterization of melanin pigment producing actinomycetes.Ind J Appl Microbiol, 19(1):9-20.
Subramani, R. and Aalbersberg, W. (2013). Culturable rare actinomycetes: diversity, isolation and marine natural product discovery. Appl Microbiol Biotechnol, 97: 9291-9321;10.1007/s00253-013-5229-7.
Sukalkar, S.R., Kadam, T.A. and Bhosale, H.J. (2017). Isolation of chitinase producing Streptomyces sp. M1 for recycling of fungal biomass. Int J Sci Res Sci Technol, (3)8:399-404.
Tajima, F. (1983). Evolutionary relationship of DNA sequences in finite populations. Genetics, 105:437-460.
Tamura, K., Nei, M. and Kumar, S. (2004). Prospects for inferring very large phylogenies by using the neighbor-joining method. Proceedings of the National Academy of Sciences of the United States of America, 101(30):11030-11035.
Thawai, C. (2012). Isolation and characterization of antibiotic-producing actinomycetes from hot spring sediment of Thailand, p.215-219. International Conference on BioScience: Biotechnology and Biodiversity, Novi Sad, Serbia.
Uzel, A., Kocabas, E.H. and Bedir, E. (2011). Prevalence of Thermoactinomyces thalpophilus and T. sacchari strains with biotechnologicalpotential at hot springs and soils from West Anatolia in Turkey. Turk J Biol, 35:195-202.
Valverde, A., Tuffin, M. and Cowan, D.A. (2012). Biogeography of bacterial communities in hot springs: a focus on the actinobacteria. Extremophiles, 16(4):669-679.
Vishwanatha, B.T., Babu, K.G., Padmashri, P., Malagi, S.B., Dandin, C.J. and Nayaka, S. (2017). Isolation, identification and characterization of Streptomyces sp. SN-2. Biosci Biotech Res Asia,14(4).
Zhao, L.X., Huang, S.X., Tang, S.K., Jiang, C.L., Duan, Y., Beutler, J.A., Henrich, C.J., McMahon, J.B., Schmid, T., Blees, J.S., Colburn, N.H., Rajski, S.R. and Shen, B. (2011). Actinopolysporins A–C and tubercidin as a Pdcd4 stabilizer from the halophilic actinomycete Actinopolyspora erythraea YIM 90600. Nat Prod, 74(9):1990-1995; doi: 10.1021/np 200603g.