Department of Studies and Research in Microbiology, Sahyadri Science College,
Kuvempu University, Shivamogga-577203, Karnataka, India
Corresponding author email: actino.research@gmail.com
Article Publishing History
Received: 21/10/2020
Accepted After Revision: 17/12/2020
Microbial alkaline proteases are among the salient hydrolytic enzyme and used extensively as biocatalysts, Extracellular alkaline protease is of great importance due to its applications in detergent, food and leather industries by the prokaryotic actinomycetes. The present investigations were focusing on soil actinomycetes. Screening for alkaline protease and characterization of specific strain from rhizosphere soil from Western Ghats of Karnataka, India. The zones of hydrolysis were observed on skim milk agar medium under pH 10.0 at 30±02 0C for 5 to 7 days and produced 96 U/mL of alkaline protease by the fermentation. Morphological and biochemical characterization of the isolate was carried out and found that the isolate belongs to the Streptomyces genus. Further species confirmation of the Streptomyces was done by 16S rRNA gene sequencing. The obtained nucleotide sequences SO-13 were submitted to the GenBank database and the accession number assigned is MW130237. The results reveal the isolate SO-13 was identified as Streptomyces tendae. The present data reveal that the isolate SO-13 represents Streptomyces tendae. The potentiality of the eco-friendly enzyme could be used for various industrial applications. The alkaline protease production from Streptomyces tendae SO-13 is the potent strain for commercial use. Further investigation at the commercial level and novel applications of alkaline proteases to be carried out.
Western Ghats, Rhizosphere soil, Streptomyces tendae, Alkaline protease.
Subbaraju A, Onkarappa R. Isolation and Characterization of Alkaline Protease Producing Streptomyces tendae SO-13 from Rhizosphere Soils of Western Ghats. Biosc.Biotech.Res.Comm. 2020;13(4).
Subbaraju A, Onkarappa R. Isolation and Characterization of Alkaline Protease Producing Streptomyces tendae SO-13 from Rhizosphere Soils of Western Ghats. Biosc.Biotech.Res.Comm. 2020;13(4). Available from: https://bit.ly/2UhZeBe
Copyright © Subbaraju and Onkarappa This is an Open Access Article distributed under the Terms of the Creative Commons Attribution License (CC-BY) https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
The Western Ghats of India has been considered as one of thirty-four biodiversity hot spots in the world, with rich flora and fauna. The Western Ghats of Karnataka popularly known as Sahyadri hills are treasure houses of endangered species but less studied concerning microbial biodiversity. Actinomycetes are widely distributed in soil, and constitute a significant part of soil microflora (Bawazir and Manjula 2018). Soil actinomycetes are prokaryotes with various metabolic activities (Chavan et al., 2013). Actinomycetes are gram-positive, filamentous, bacteria, characterized by the formation of aerial mycelium, and spores on solid media, with DNA high in G+C content of 60-70 mol% (Shirling and Gottlieb 1966; Subbaraju and Onkarappa 2018).
Among the microorganisms, actinomycetes acquire special importance as the power source of antibiotics and, other bioactive primary and secondary metabolites such as enzymes (Chavan et al., 2013). Sources of commercial enzymes cover a wide range from microorganisms then animals and plants sources. Fungi and yeast contribute about 50%, bacteria 25%, animal 8% and plant 4% of the total in commercial enzymes production (Azmi et al., 1999). Proteases of neutrophilic as well as alkaliphilic bacterial and fungal origins are utilized for commercial exploitation (Pathak and Rothad 2018). Several microbial species are reported for the alkaline protease productions. The most copiously studied the alkaline protease producers among bacteria are the genera of Bacillus (Keshavamurthy et al., 2018) and Pseudomonas (Alexander et al., 2012), Streptomyces species (Sarkar and Suthindhiran 2020).
Alkaline proteases are also utilized for therapeutic agent’s development. The oral administration of proteases from Aspergillus oryzae to aid in digestion and rectify lytic enzyme deficiency syndromes is already in practice (Mikawlrang, 2016).More recently, actinomycetes are an auspicious source of a wide range of important enzymes. Streptomyces protease preparations that are profit-oriented used include FRADIASE 7M (S. fradiae) and PRONASE 7M (S. griseus). While alkaline proteases from bacteria are vastly characterized, similar attention has not been paid to actinobacteria. Currently, antibiotics are the major bioactive compounds from the actinobacteria. However, in these prokaryotes, the ability to produce a variety of enzymes may be an attractive phenomenon (Govindharaj et al., 2016). In the present investigation, the new isolate SO-13 capable of producing the alkaline protease. The study includes morphological, biochemical and molecular identifications.
MATERIAL AND METHODS
Isolation Proteolytic actinomycetes from soil samples: The rhizosphere soil samples were collected in a sterile Ziplock plastic cover from a depth of 10-15 cm from the Western Ghats in Karnataka, India (Around the latitude 140 13l 07.3ll N and longitude 740 49l 57.2ll E). The samples were air-dried for 6 to 7 days and ground in a mortar using a pestle (Lingakumar et al., 2014; Subbaraju and Onkarappa, 2018). The samples were serially diluted and 0.1 ml of 10-2 to 10-6 dilutions were plating on Starch Casein agar medium (g/ml: Starch 10; Casein 3; KNO3 2; NaCl 2; K2HPO4 2; MgSO4 0.05; CaCl2 0.02; FeSO4.7H2O, 0.01; agar 20, distilled water 1000ml pH=7.0± 0.2) described by Guravaiah (2016). The plates were incubated at 30±20 0C for 5 to 7 days. The isolates were grown on starch casein nitrate agar medium at 30±20 0C and stored at 40C for short term storage.
Screening for alkaline proteolytic activity: Proteolytic activity from isolated pure cultures was screened by plating on Skim milk agar Supplement with Na2CO3 for detection alkaline protease producing isolate (g/L Skim milk, 100.0; Yeast exact, 5.0: Na2CO3, 10.0; Agar, 20; distilled water, Final pH=10). Incubate for 5 to 7 days at 30±02 0C (Davoudi et al., 2014).
Characterization of potent alkaline producing isolate SO-13:The classifications of actinomycetes were originally based upon the morphological observations. Preferably, the Streptomyces species were identified and recorded using the Bergey’s Manual of Systematic Bacteriology. Primarily, the characterizations of actinomycetes isolates were done by their colony morphology, spore colour, aerial mass colour and substrate mycelium, pigmentation appearance on the medium. The isolate was subjected to grams and acid-fast staining procedures. The isolate was identified to genus level based on their spore chain arrangement by coverslip technique and spore surface ornamentation by SEM analysis (Gautham et al., 2012).
Biochemical tests of isolate:Standard biochemical tests were employed, the Indole test, Methyl red test, Voges – Proskauer test, Citrate utilization test, Urease test, Catalase test, Degradation of Cellulose, hydrolysis of Casein, Gelatin and Starch to determine the potent alkaline protease producing strain% (Shirling and Gottlieb 1966; Gautham et al., 2012; Subbaraju and Onkarappa 2018).
16S rRNA gene sequencing of the isolate:The strain was further subjected for identification by 16S rRNA sequence using universal primers and genomic DNA as a template. The genomic DNA extracted from the isolate by using spin column kit (HiMedia, India). Bacterial 16S rRNA gene (1500 bp), (Clarridge JE, 2004) was amplified using polymerase chain reaction (PCR) in a thermal cycler and were purified using Exonuclease I -Shrimp Alkaline Phosphatase (Exo-SAP) (Darby et al., 2005). Purified amplicons were sequenced by Sanger method in ABI 3500xL genetic analyzer (Life Technologies, USA).
Sequencing files (.ab1) edited using CHROMASLITE (version 1.5) and further analyzed by Basic Local Alignment Search Tool (BLAST) with closest culture sequence retrieved from the National Centre for Biotechnology Information (NCBI) database that finds regions of local similarity between sequences (Altschul et al., 1990). A duly annotated partial nucleotide sequence of the strain was deposited with NCBI Genbank (https://www.ncbi.nlm.nih.gov). Molecular Evolutionary Genetics Analysis (MEGA) 6.0 software to construct the Phylogenetic tree using the neighbour-joining method (Tamura et al., 2011).
Alkaline protease production and enzyme assay
Alkaline Protease production by the selected isolate was carried out by One ml of fresh isolate inoculum were added 100 ml of production medium (Glucose,10.0g/L; casein, 5.0 g/L; yeast extract, 5.0 g/L; K2H PO4, 2.0 g/L; KH2 PO4, 2.0 g/L; MgSO4. 7H2O, 1.0 g/L and at pH 9.0-9.5) into 250ml Erlenmeyer flask. The flask was placed in a rotary shaker incubator at 150 rpm at 30 °C for 5 to 7 days. The fermentation broth was centrifuged at 8,000rpm for 20 min at 4 0C to obtain the crude culture filtrate (Hosseini et al., 2016). The alkaline protease activity of the crude enzyme was done by taking 0.5 ml of culture filtrate was added to 0.5 ml of 1% casein (a substrate) in 0.1 M Phosphate buffer (pH 7.0) and then incubated for 10 min at room temperature.
To stop reaction 3ml of 10% (w/v) trichloroacetic acid and the mixture was centrifuged at 5000 rpm for 10 min. The supernatant, 5 ml 0.5M Na2CO3 solution and 0.5ml of two Folin Ciocalteau reagent was added and mixed thoroughly, incubated for 30 min at room temperature in dark condition. The optical density of blue colouration was measured using the UV-VIS spectrophotometer at 660 nm and the blank (Keshavamurthy et al., 2018). The amount of the released amino acids was calculated using the tyrosine standard. One unit of enzyme activity represents the amount of the enzyme required to release 1μg of tyrosine per ml per min (Pant et al., 2015).
RESULTS AND DISCUSSION
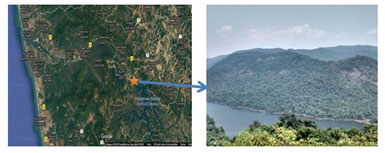
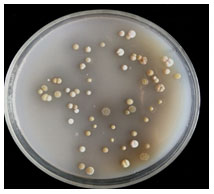
Fifteen rhizosphere soil samples were collected in sterile Zip lock plastic cover from at different Latitude and longitude of the Western Ghats in Karnataka, India (Figure 1) and air-dried soil samples were subject to isolation and based on the zone of hydrolysis on agar plates, SO-01 to SO-16 visible colonies were obtained on 10-2 to 10-6 dilution Petri plates (Figure 2).
Figure 1: The map showing sampling stop of Western Ghats, Karnataka.
Figure 2: Visible colonies on 10-4dilution petriplate
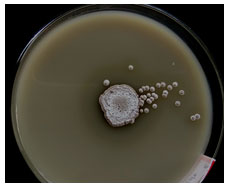
Isolate SO-13 was found to have alkaline protease activity depend on diameter zone of hydrolysis of proteolytic on the plate (Figure 3). Similar result found in Streptomyces griseorubens E44G (Rashadb et al., 2015) and Streptomyces Indus (Guravaiah 2016) for proteolytic activity. Hence the isolate SO-06 was selected for further studied.
Figure 3: Zone of hydrolysis by the isolate Streptomyces tendae SO-13
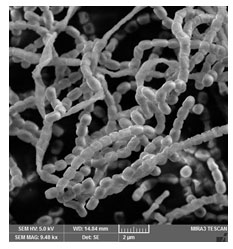
Characterization of the potent alkaline protease producing isolate SO-13: The result showed a diverse morphological characteristic with spore colour-grey, colony morphology-radial, substrate-white and aerial mycelium-grey (Figure 3). The spore-bearing hyphae and spore chains show straight-flexuous with open and spore surface Ornamentation-smooth (Figure 4). Isolate shown the gram-positive, filamentous, rod structure, identified by gram staining and Nonacid fast. The biochemical characteristics as potent alkaline protease isolate SO-13 exhibit positive for methyl red, voges-proskauer, citrate, urease, catalase, hydrolysis of casein, starch and gelatin (Table 1). Based on the spore chain arrangements the isolate was assigned to the genus Streptomyces sp. Similar results were observed in Streptomyces sp. (Takeuchi et al., 1996) and Streptomyces tendae AR1 (Laidi et al.,2006).
Figure 4: SEM of the isolate Streptomyces tendae SO-13
Table 1. Morphological and Biochemical Characterization of isolate Streptomyces tendae SO-13
| Morphological identification | |
| Media | Starch Casein Nitrate |
| Growth | Abundant |
| Colony Morphology | Radiating |
| Aerial Mycelium | Grey |
| Substrate Mycelium | White |
| Diffusible Pigment | No |
| Spore Arrangement | Straight-flexuous |
| Spore surface Ornamentation | Smooth |
| Biochemical tests | |
| Indole | – |
| Methyl Red | + |
| Voges – Proskauer | + |
| Citrate | + |
| Urease | + |
| Catalase | + |
| Casein | +++ |
| Starch | ++ |
| Cellulose | – |
| Gelatin | + + |
| pH | 7.0-10.0 |
| NaCl | 5% |
| Tentative genera | Streptomyces sp. |
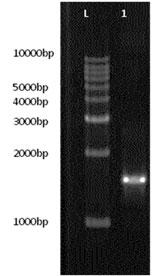
The 16S rRNA gene was amplified through PCR which showed 1500 kb band on 2% agarose gel (Figure 5). Subsequently, 16S rRNA gene sequence analyses were carried out to elucidate the taxonomic relationships among closely related Streptomyces species. The strain SO-13 has been matriculated into a cluster containing Strepomyces tendae. Based on the phylogenetic analysis the strain was closely related to Streptomyces tendae strain ATCC 19812 exhibiting high similarity (99.88 %) (Takeuchi et al., 1996). So it is assigned as Streptonyces tendae SO-13 (Figure 6). Similar results were noticed in Streptonyces tendae AR1 (Laidi et al., 2006) and Streptonyces tendae1&2 (Bahamdain et al., 2020). The obtained nucleotide sequences SO-13 were submitted to the GenBank database and the accession number assigned is MW130237. The results reveal the isolate SO-13 was identified as Streptomyces tendae.
Figure 5: Agarose gel electrophoresis of Streptomyces tendae SO-13 L-Step-up 1 Kb DNA Ladder Lane 1- 16S rRNA amplicon
Figure 6: Phylogram obtained based on phylogenetic analysis of 16S rRNA gene sequence data showing the phylogenetic positions of isolate Streptomyces tendae SO-13
Streptomyces tendae SO-13 was subjected to secondary screening for quantitative protease production. The protease produces 96 U/mL during 5 to 7days at pH 9.0. Similar results were observed in Saccharomonospora viridis SJ-21 (Hosseini et al., 2016) and Streptomyces flavogriseus HS1 (Sofiane et al., 2014).Streptomyces were industrially important organisms for commercial production of Protease. Alkaline proteases have been produced from numerous Streptomyces like thermoalkaline proteases by marine Streptomyces sp. D1 (Madanrao et al., 2013). Streptomyces.sp LL-DAP (Karthik et al., 2010) and Streptomyces flavogriseus HS1 (Sofiane et al., 2014) are also reported for protease production.
CONCLUSION
The Western Ghats are the treasure house for novel actinomycetes species associated with the rhizosphere soil. The characterizations of alkaline protease Streptomyces tendae SO-13 was confirmed by morphology, biochemical and 16S rRNA sequencing. The alkaline protease producing from Streptomyces tendae SO-13 is the potent strain for commercial production. Further investigation at the commercial level and novel applications of alkaline proteases to be carried out.
ACKNOWLEDGEMENTS
The authors acknowledge the Department of Studies and Research in Microbiology, Sahyadri Science College (Autonomous), Kuvempu University, Shivamogga for providing adequate lab facilities and first author sincere thanks to UGC-RGNF for providing fellowship for research work.
Conflict of interest: Authors declare that they have no conflict of interest in the publication.
REFERENCES
Alexander J Laarman, Bart W Bardoel, Maartje Ruyken, Job Fernie, Fin J Milder, Jos A G, Van Strijp and Suzan H M. (2012). Rooijakkers. Pseudomonas aeruginosa Alkaline Protease Blocks Complement Activation via the Classical and Lectin Pathways. Journal of Immunology, 188:386-393.
Ali Mohammed Abdullah Bawazir and Manjula Shantaram. (2018). Ecology and distribution of actinomycetes in nature –A review International Journal of Current Research, 10(7): 71664-71668.
Altschul SF, Gish W, Miller W, Myers EW and Lipman DJ. (1990). Basic local alignment search tool. Journal Molecular Biology. 215(3):403-10.
Anupama P Pathak and Mukundraj G Rathod. (2018). A review on alkaline protease producers and their biotechnological perspectives. Indian Journal of Geo Marine Sciences, 47 (06): 1113-1119.
Astalakshmi A, Thangapandian V and K Lingakumar. (2014). Isolation and Characterization of actinomycetes from the Soil of Devathanam – A Foot-hill of Western Ghats International Journal of Pharma Research & Review, 3(1):15-20.
Azmi W, Banerjee U C, Sani R K and Soni R. (1999). Thermostable alkaline protease from Bacillus brevis and its characterization as a laundry detergent additive. Proc. Biochem, 35: 213-219.
Chavan Dilip V, Mulaje S S and Mohalkar RY. (2013). A review on actinomycetes and their biotechnological application. International Journal of Pharmaceutical Sciences and Research, 4(5): 1730-1742.
Clarridge JE. (2004). Impact of 16S rRNA gene sequence analysis for identification of bacteria on clinical microbiology and infectious diseases. Clinical Microbiology Review, 17(4):840-62.
Darby AC, Chandler SM, Welburn, SC, and Douglas AE. (2005). Aphid-symbiotic bacteria cultured in insect cell lines. Applied Environmental Microbiology, 71(8): 4833-4839.
Elham Davoudi, Keivan Beheshti Maal and Hashem Nayeri. (2014). Isolation and identification of Streptomyces pseudogriseolus as a nominate for producing alkaline protease Advances in Environmental Biology, 8(10):1072-1077.
Gargi Sarkar and Suthindhiran K. (2020). Extraction and characterization of alkaline protease from Streptomyces sp. GS – 1 and its application as dehairing agent Biocatalysis and Agricultural Biotechnology, 25:101590.
Gautham SA, Shobha KS, Onkarappa R and Prashith KTR. (2012). Isolation, characterization and antimicrobial potential of Streptomyces sps from Western Ghats of Karnataka, India. Research Journal of Pharmacy and Technology, 5(2): 233-238.
Govindharaj Vaijayanthi, Ramasamy Vijayakumar and Dharmadurai Dhanasekaran. (2016). Actinobacteria — A Biofactory of Novel Enzymes, 331-35.
Guravaiah. M. (2016). Production of alkaline protease by streptomyces indicus. Darshan publishers, India.1-186.
Jayasree D, Sandhya Kumari T D, Kavi Kishor P.B, Vijaya Lakshmi M and Lakshmi Narasu M. (2010). Optimization of Production Protocol of Alkaline Protease by Streptomyces pulvereceus. Inter JRI Science and Technology, 1(2):79-82.
Keshavamurthy M, Vishwanatha T, Suresh Kumar M and Subhaschandra M Gaddad. (2018). Enhanced production of alkaline protease from novel bacterium Bacillus cereus GVK21 under submerged fermentation. Bioscience Biotechnology Research Communication, 11(3): 416-425.
Lina A Bahamdain, Salah E Abo Aba, Ayman Sabry, Reda H Amasha, Samah O Noor and Magda M Aly. (2020). Molecular Identification and Phylogenetic Analysis of Some Rare Actinomycetes Obtained from Al-Lith Hot Spring Area of Saudi Arabia Bioscience Biotechnology Research Communication, 13 (3):1037-1049.
Loganathan Karthik, Gaurav Kumar and Kokati Venkata Bhaskara Rao. (2010). Mutational effects on the protease producing marine actinomycetes isolated from scylla serrata. Pharmacology online, 1: 221-227.
Madanrao Mane, Kakasaheb Mahadik and Chandrakant Kokare. (2013). Purification, characterization and applications of Thermostable alkaline protease from marine Streptomyces sp. D1. International Journal Pharmacy and Biological Science, 4(1): 572-582.
Mikawlrang, K. (2016). Aspergillus in Biomedical Research. In: Gupta, V.K. (Ed.), New and future developments in microbial biotechnology and bioengineering: Aspergillus system properties and applications. Elsevier, 228-238.
Pant G Prakash, A Pavani, J V P Bera, S Deviram, G.V.N.S, Kumar A, Panchpuri M and Prasuna R G. (2015). Production, optimization and partial purification of protease from Bacillus subtilis. Journal of Taibah University for Science, 9(1): 50-55.
Rabah Forar Laidi, Amany L Kansoh, Ali, M Elshafei and Bengraa Cheikh. (2006). Taxonomy, identification and biological activities of a novel isolate of Streptomyces tendae. Arab Journal of Biotechnology, 9(3): 427-436.
Seyed Vesal Hosseini, Zahra Saffari, Ali Farhanghi, Seyed Mohammad Atyabi, Dariush Norouzian. (2016). Kinetics of alkaline protease production by Streptomyces griseoflavus PTCC1130. Iran Journal Microbiology, 8(1): 8-13.
Shilpa A Jani, Chaitanya J Chudasama, Deval B Patel, Parul S Bhatt and Harshad N Patel. (2012). Optimization of Extracellular Protease Production from Alkali Thermo Tolerant Actinomycetes: Saccharomonospora viridis SJ-21 Bull. Environ. Pharmacological Life Science, 1(6): 84-92.
Shirling EG and Gottlieb D. (1966). Methods for characterization of Streptomyces species. International Journal Systemic Bacteriology, 16: 313-340.
Sofiane Ghorbel, Maher Kammoun, Hala Soltana, Moncef Nasri, and Noomen Hmidet. (2014). Streptomyces flavogriseus HS1: Isolation and Characterization of Extracellular Proteases and Their Compatibility with Laundry Detergents. Bio Med Research International, 1-8.
Subbaraju. A and R. Onkarappa. (2018). Antibacterial potential of biologically reduced silver nanoparticles from Streptomyces sp. SO-01. International Journal of Pharmacy and Biological Science, 8 (2): 445-451.
Rashadb YM, Elsayed Elsayed Hafezc, Waleed Mohamed Abdulkhaird, Zakaria Awad Bakae and Khalid Mohamed Ghoneem. (2015). Characterization of alkaline protease produced by Streptomyces griseorubens E44G and its possibility for controlling Rhizoctonia root rot disease of corn, Biotechnology & Biotechnological Equipment, 29(3); 457-462.
Takeuchi T, Sawada H, Tanaka F and Matsuda I. (1996). Phylogenetic analysis of Streptomyces spp. causing potato scab based on 16S rRNA sequences International Journal of Systemic Bacteriology, 46 (2): 476-479.
Tamura K, Peterson D, Peterson N, Stecher G, Nei M and Kumar S. (2011). MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molecular biology and evolution, 28(10): 2731-2739.