1Department of Biological Sciences, Faculty of Science,
2Princess Dr. Najla Bint Saud Al-Saud Center for Excellence Research in Biotechnology, King Abdulaziz University, Jeddah, Saudi Arabia.
Corresponding author email: saalghamdi17@gmail.com
Article Publishing History
Received: 07/10/2019
Accepted After Revision: 29/11/2019
Gestational diabetes mellitus (GDM) is defined as diabetes that only progresses during pregnancy and resolves after delivery. Family history (FH) of type 2 diabetes mellitus T2DM is one of the major risk factors for GDM. The aim of this study was to compare GDM between women with and without FH of T2DM. In this study, 137 GDM;150 non-GDM women were involved. Anthropometric and biochemical data were recorded. Calculation analysis confirmed the negative impact with the FH of T2DM between GDM (58.4%) and non-GDM (56%) women (p=0.97). Negative association was observed when the GDM cases with family history of T2DM was compared with GDM without family history of T2DM (p=1.01). However, age, FBG, and PPBG levels were p<0.05; BMI was non-significant (p=0.22). In conclusion, this study disclosed FH of T2DM did not affect GDM in women in an Indian population.
Gestational diabetes mellitus, Family history, Type 2 diabetes mellitus, Obesity, Indian population
Al-Ghamdi S. A. Influence on Diabetic Pregnant Women with a Family History of Type 2 Diabetes. Biosc.Biotech.Res.Comm. 2019;12(4).
Al-Ghamdi S. A. Influence on Diabetic Pregnant Women with a Family History of Type 2 Diabetes. Biosc.Biotech.Res.Comm. 2019;12(4). Available from: https://bit.ly/34z7ccF
Copyright © Al-Ghamdi This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
In biochemical terms, Gestational diabetes mellitus (GDM) is well-defined as glucose intolerance identified during pregnancy (Ravnsborg et al., 2019). In traditional terms, GDM is defined as the initial detection of hyperglycemia during pregnancy (Agha-Jaffar et al., 2019, Xie et al., 2019). Later in life, GDM women will increase the risk of obesity, type 2 diabetes mellitus (T2DM) or metabolic disorders (Khan et al., 2014). GDM-risk is eight times advanced in obesity pregnant women (Ravnsborg et al., 2019). Advance maternal age, family histories of GDM and T2DM, and western lifestyle have also been documented as risk factors for GDM (Khan et al., 2014). Till now controversies raise for diagnostic criteria which is further needs for treatment and monitoring of GDM; however, omics studies documented regarding the maternal metabolic profile in GDM and non-GDM women, which can be helpful in predicting the GDM disease (Souza et al., 2019). Obesity, hypertension, and physical activity are modifiable-risk-factors while age, ethnicity, and family history (FH) are non-modifiable risk factors of GDM (Khan et al., 2019, Rodrigues et al., 2019).
During pregnancy, a woman’s body undergoes numerous metabolic changes; for example, placental hormones stop proper functioning of insulin, leading to insulin resistance, and this results in accumulation of glucose in the blood (Ghassibe-Sabbagh et al., 2019). Presently, no prevention strategy or treatment is available for GDM. However, some lifestyle interventions, such as diet, exercise, and occasionally, insulin therapy, are available for abnormal glucose values during pregnancy. Currently, long-term oral antidiabetics (glyburide and metformin) are promising candidates for long-term treatment of both mother and children (Plows et al., 2018). GDM is detected by GCT/OGTT. World Health Organization presently recommends that OGTT tests be performed during fasting (92 mg/dL), 1h (180 mg/dL), and 2h (153 mg/dL). GDM is diagnosed when the results of any of the tests cross the normal or prescribed values (Kautzky-Willer et al., 2018). In our previous study, we performed OGTT tests during fasting and 1, 2, and 3 h after meals (Khan et al., 2014). Women with GDM can develop T2DM later in life. Their children can be macrosomic at birth or have glucose intolerance in adulthood (Rahman et al., 2019). Additionally, they are prone to obesity from childhood. The risks of GDM, T2DM, and obesity are inter-connected (Lowe et al., 2019). Women should be more careful about these diseases. Genetic predisposition is a risk factor for metabolic disorders; family history plays an important role in medical history. Lifestyle, non-nutritious diet, and environmental factors are other factors that makes the genetic predisposition as an important factor apart from the family histories (Alharithy et al., 2018). Numerous studies have evaluated GDM in Indian populations, but no studies have evaluated the role of family history in GDM development. Thus, this study aimed to compare women with GDM who did or did not have family history of T2DM in an Indian population.
MATERIALS AND METHODS
Recruitment of pregnant women
Ethical approval was obtained to include GDM and non-GDM women in this study. Enrolled 137 pregnant women were diagnosed with GDM and 150 pregnant women as non-GDM. All pregnant women were initially screened by performing glucose challenge tests (GCTs) and OGTTs. Diabetes during pregnancy was confirmed based on abnormal glucose values either in GCT or OGTT. The details of GCTs and OGTTs and their normal and abnormal values were documented in the previous publication (Khan et al., 2014). The criteria for inclusion and exclusion of women with and without GDM were given in recent publication (Khan et al., 2019).
Questionnaire
The questionnaire used in this study collected of anthropometric, biochemical, and clinical details as well as family history of T2DM. Age (years), height (cm), weight (kg), mean gestational age of women with GDM, and BMI were calculated. For determining FBG and post-prandial blood glucose (PPBG) levels and for performing GCT and OGTT (fasting hour and 1, 2, and 3 h after meals), fasting and non-fasting coagulant blood was collected and the serum was separated; this serum was used for biochemical analysis. Clinical data consisted of information about whether the women with GDM were receiving insulin treatment or dietary interventions.
Statistical analysis
The data of GDM cases and controls was analyzed by sample t-tests using R-software system.
RESULTS AND DISCUSSION
Analysis results of clinical factors of women with and without GDM are shown in Table 1. GDM women had ages ranging from 22 years to 38 years, with the mean age of 26.7±5.1 years. Women without GDM had ages ranging from 17-34 (24.6±3.5) years. Mean age was p=0.0001. Weight (69.3±10.1 vs 66.2±9.2 kg) and BMI (26.8±3.9 vs 24.1±3.5 kg/m2) were high in GDM women (p=0.26-0.22). Mean gestational age (24.4±5.0 years) and medication-related factors, such as diet (40.9%) and insulin levels (59.1%) were measured only in women with GDM. Serum glucose factors, such as FBS (110.6±3.9 mg/dL vs. 99.2±11.3 mg/dL) and PPBG (158.8±47.7 vs 112.0±39.7) levels were significantly different between women with and without GDM (p<0.05). FH of T2DM was more frequent in women with GDM (58.4%) than in women without GDM (56%); however, this difference was not significant (p=0.97). In the GDM group, 80 women had FH of T2DM whereas 57 did not have FH of T2DM or any other diabetes. There was no deceptive differences in either GDM with FH of T2DM (n=80) or GDM without FH of T2DM (n=57; Table 2). In the GDM group, mean age was found be higher in women with FH than in women without FH of T2DM (27.3±4.9 vs 25.9±4.4 years; p=0.39). In the GDM group, 51 (63.7%) and 29 (36.3%) women with FH of T2DM were receiving insulin therapy and dietary interventions, respectively; of the women without FH of T2DM, 30 (52.6%) and 27 (47.4%) GDM were receiving insulin therapy and dietary interventions, respectively.
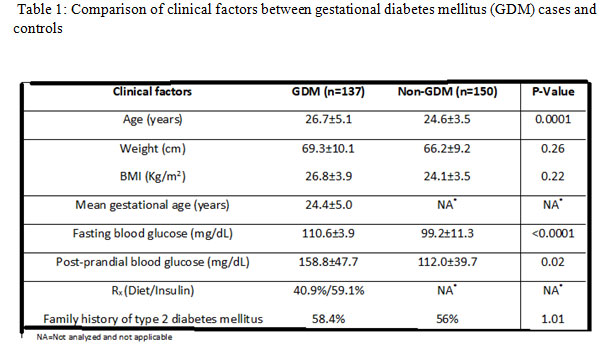 |
Table 1: Comparison of clinical factors between gestational diabetes mellitus (GDM) cases and controls |
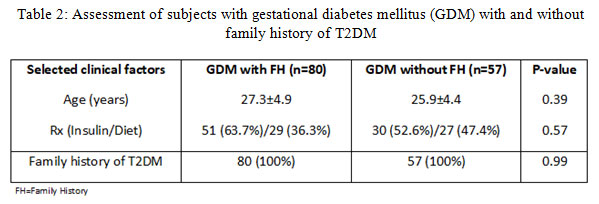 |
Table 2: Assessment of subjects with gestational diabetes mellitus (GDM) with and without family history of T2DM |
This is the first study to compare the risk of GDM according to family history of T2DM in women in an Indian population. Study results indicate the negative association when compared the risks with age and medications (Rx) with (n=80) and without (n=57) family history of T2DM. History of specific disease(s) in the family or family pedigree can affect the presence of genetic diseases in any individual. For example, genetics or family pedigree can affect diabetes. Among complex diseases, T2DM is mainly influenced by genetic and environmental factors; a similar influence is observed on obesity, which is also known as an important predictor of development of T2DM and cardiovascular diseases (CVDs). FH of T2DM is a well-known risk factor for GDM, is known to be a medical condition equally appears in T2DM. Both T2DM and GDM have similar pathophysiologies that involve β-cell dysfunction and insulin resistance (Khan et al., 2014). FH has been known to be a risk factor for CVD and T2DM, and specific diseases reflect the consequences of genetic susceptibility, shared environment, and common behaviors. In the field of clinical medicine, FH is recognized as an important but not yet non-modifiable risk factor diseases prevents the influences of suspected diagnosis. The major advantage of FH over genomic tools includes the low cost of determination of genetic and environmental risk factors; however, this utility has been poorly explored in public health (Das et al., 2012). FH of T2DM is a strong metabolic risk factor of T2DM. The major risk factors of T2DM are waist circumference, BMI, physical inactivity, and family history (Diabetologia, 2013). FH of diabetes in first-cousin consanguinity is an initial risk factor for GDM; this further emphasizes the role of genetics in diabetes susceptibility (Kiani et al., 2017).
GDM is associated with maternal obesity, which confers a 4-7-fold risk of T2DM occurrence along with a predisposition toward development of MetS in middle age. FH of GDM in women without diabetes is characterized by increases in FBG and insulin levels, dyslipidemia, and high inflammation in the absence of MetS. Self-histories of GDM women is having the future risks with MetS and 66% of CVD such as stroke, myocardial infarction and coronary artery disease. However, no global studies are available that support the women diagnosed with GDM in the first delivery has increased with MetS and CVD (Gunderson et al., 2014). Moreover, T2DM negates the protected effect in females, but the CVD risk is comparable between men and women with T2DM. MetS and CVD alters the increases T2DM risks which is characterized by adiposity, dyslipidemia, hypertension and elevated fasting glucose levels. Several studies have reported evidence of the MetS in women with a history of GDM (Carr et al., 2006). Limited risk factors were documented in the GDM women. However, the importance of other factors, such as short stature, lower birth weight, ethnicity, smoking status, multiparity, gestational weight gain, physical inactivity, and socioeconomic factors, is still controversial (Dode and Santos, 2009).
Carr et al (Carr et al., 2006) concluded that CVD was significantly common in women with history of GDM. Dode et al (Dode and Santos, 2009) performed a meta-analysis study by considering age, FH of T2DM, and obesity as well-identified risk factors in GDM women and concluded that publication bias cannot be ruled out with small sample sizes. Kiani et al (Kiani et al., 2017) performed a systematic review and meta-analysis on women with GDM and confirmed that age, obesity, and family history of T2DM were risk factors for GDM in Iranian women. In subjects without metabolic disorders, physical activity has been reported to reduce the risk of diseases and unnecessary fat accumulation, weight gain, insulin resistance, and specifically diabetes. The relation between physical activity and GDM during pregnancy has been stated as lower levels of glucose intolerance (Bung et al., 1991). Aune et al (Aune et al., 2016)performed a meta-analysis on GDM women performing physical activity and confirmed the significant inverse association. Previous studies involving American and Australian women with GDM have confirmed that FH of T2DM is a risk factor, along with other features (Santos-Ayarzagoitia et al., 2006). Other global studies have also confirmed that FH of T2DM is one of the risk factors in women with GDM (Herath et al., 2017, Kuti et al., 2011, Lin et al., 2016, Moosazadeh et al., 2017, Ogonowski et al., 2014, Rhee et al., 2010, Van Leeuwen et al., 2010, Wahabi, 2018, Yang et al., 2009). However, Mahalakshmi et al (2014) performed a study in India and concluded that 70% of the enrolled subjects with GDM had FH of T2DM. Current study results are consistent with those of Riaz et al (Riaz et al., 2018) who did not find FH of T2DM to be a risk factor in women with GDM. Schleger et al (Schleger et al., 2018) concluded that FH of T2DM in the mother affected fetal post-prandial brain function. This study concludes that FH of T2DM is not a risk factor in women with GDM in an Indian population, when compared with either women with GDM who had no FH of T2DM or women without GDM. This could be due to the limited sample size in the capital city of diabetes mellitus.
CONFLICT of INTEREST
There is no conflict of Interest towards this article.
REFERENCES
Agha-Jaffar, R., Oliver, N. S., Kostoula, M., Godsland, I. F., Yu, C., Terry, J., Johnston, D., Gable, D., Robinson, S. J. T. J. O. M.-F. & Medicine, N. 2019. Hyperglycemia Recognised In Early Pregnancy Is Phenotypically Type 2 Diabetes Mellitus Not Gestational Diabetes Mellitus: A Case Control Study. 1-7.
Alharithy, M. K., Alobaylan, M. M., Alsugair, Z. O. & Alswat, K. A. J. E. P. 2018. Impact Of Family History Of Diabetes On Diabetes Control And Complications. 24, 773-779.
Aune, D., Sen, A., Henriksen, T., Saugstad, O. D. & Tonstad, S. 2016. Physical Activity And The Risk Of Gestational Diabetes Mellitus: A Systematic Review And Dose–Response Meta-Analysis Of Epidemiological Studies. Springer.
Bung, P., Artal, R., Khodiguian, N. & Kjos, S. J. D. 1991. Exercise In Gestational Diabetes: An Optional Therapeutic Approach? 40, 182-185.
Carr, D. B., Utzschneider, K. M., Hull, R. L., Tong, J., Wallace, T. M., Kodama, K., Shofer, J. B., Heckbert, S. R., Boyko, E. J. & Fujimoto, W. Y. J. D. C. 2006. Gestational Diabetes Mellitus Increases The Risk Of Cardiovascular Disease In Women With A Family History Of Type 2 Diabetes. 29, 2078-2083.
Das, M., Pal, S. & Ghosh, A. J. J. O. C. D. R. 2012. Family History Of Type 2 Diabetes And Prevalence Of Metabolic Syndrome In Adult Asian Indians. 3, 104-108.
Diabetologia, I. C. J. 2013. The Link Between Family History And Risk Of Type 2 Diabetes Is Not Explained By Anthropometric, Lifestyle Or Genetic Risk Factors: The Epic-Interact Study. 56, 60-69.
Dode, M. A. S. D. O. & Santos, I. S. D. J. C. D. S. P. 2009. Non Classical Risk Factors For Gestational Diabetes Mellitus: A Systematic Review Of The Literature. 25, S341-S359.
Ghassibe-Sabbagh, M., Mehanna, Z., Farraj, L. A., Salloum, A. K., Zalloua, P. A. J. J. O. C. & Endocrinology, T. 2019. Gestational Diabetes Mellitus And Macrosomia Predispose To Diabetes In The Lebanese Population. 16, 100185.
Gunderson, E. P., Chiang, V., Pletcher, M. J., Jacobs Jr, D. R., Quesenberry Jr, C. P., Sidney, S. & Lewis, C. E. J. J. O. T. A. H. A. 2014. History Of Gestational Diabetes Mellitus And Future Risk Of Atherosclerosis In Mid‐Life: The Coronary Artery Risk Development In Young Adults Study. 3, E000490.
Herath, H., Herath, R. & Wickremasinghe, R. J. P. O. 2017. Gestational Diabetes Mellitus And Risk Of Type 2 Diabetes 10 Years After The Index Pregnancy In Sri Lankan Women—A Community Based Retrospective Cohort Study. 12, E0179647.
Kautzky-Willer, A., Harreiter, J., Winhofer-Stöckl, Y., Bancher-Todesca, D., Berger, A., Repa, A., Lechleitner, M. & Weitgasser, R. J. W. K. W. 2018. Gestationsdiabetes (Gdm)(Update 2019). 1-12.
Khan, I. A., Jahan, P., Hasan, Q., Rao, P. J. D., Research, M. S. C. & Reviews 2019. Genetic Confirmation Of T2dm Meta-Analysis Variants Studied In Gestational Diabetes Mellitus In An Indian Population.
Khan, I. A., Movva, S., Shaik, N. A., Chava, S., Jahan, P., Mukkavali, K. K., Kamineni, V., Hasan, Q. & Rao, P. J. M. G. 2014. Investigation Of Calpain 10 (Rs2975760) Gene Polymorphism In Asian Indians With Gestational Diabetes Mellitus. 2, 299-306.
Kiani, F., Naz, M. S. G., Sayehmiri, F., Sayehmiri, K. & Zali, H. J. D. 2017. The Risk Factors Of Gestational Diabetes Mellitus: A Systematic Review And Meta-Analysis Study. 10, 17.
Kuti, M. A., Abbiyesuku, F. M., Akinlade, K. S., Akinosun, O. M., Adedapo, K. S., Adeleye, J. O. & Adesina, O. A. J. J. O. C. P. 2011. Oral Glucose Tolerance Testing Outcomes Among Women At High Risk For Gestational Diabetes Mellitus. 64, 718-721.
Lin, P.-C., Hung, C.-H., Chan, T.-F., Lin, K.-C., Hsu, Y.-Y. & Tzeng, Y.-L. J. M. 2016. The Risk Factors For Gestational Diabetes Mellitus: A Retrospective Study. 42, 16-20.
Lowe, W. L., Scholtens, D. M., Kuang, A., Linder, B., Lawrence, J. M., Lebenthal, Y., Mccance, D., Hamilton, J., Nodzenski, M. & Talbot, O. J. D. C. 2019. Hyperglycemia And Adverse Pregnancy Outcome Follow-Up Study (Hapo Fus): Maternal Gestational Diabetes Mellitus And Childhood Glucose Metabolism. 42, 372-380.
Moosazadeh, M., Asemi, Z., Lankarani, K. B., Tabrizi, R., Maharlouei, N., Naghibzadeh-Tahami, A., Yousefzadeh, G., Sadeghi, R., Khatibi, S. R., Afshari, M. J. D., Research, M. S. C. & Reviews 2017. Family History Of Diabetes And The Risk Of Gestational Diabetes Mellitus In Iran: A Systematic Review And Meta-Analysis. 11, S99-S104.
Ogonowski, J., Miazgowski, T., Engel, K. & Celewicz, Z. 2014. Birth Weight Predicts The Risk Of Gestational Diabetes Mellitus And Pregravid Obesity. Nutrition, 30, 39-43.
Plows, J., Stanley, J., Baker, P., Reynolds, C. & Vickers, M. J. I. J. O. M. S. 2018. The Pathophysiology Of Gestational Diabetes Mellitus. 19, 3342.
Rahman, M. L., Zhang, C., Smarr, M. M., Lee, S., Honda, M., Kannan, K., Tekola-Ayele, F. & Louis, G. M. B. J. E. I. 2019. Persistent Organic Pollutants And Gestational Diabetes: A Multi-Center Prospective Cohort Study Of Healthy Us Women. 124, 249-258.
Ravnsborg, T., Svaneklink, S., Andersen, L. L. T., Larsen, M. R., Jensen, D. M. & Overgaard, M. J. P. O. 2019. First-Trimester Proteomic Profiling Identifies Novel Predictors Of Gestational Diabetes Mellitus. 14, E0214457.
Rhee, S. Y., Kim, J. Y., Woo, J.-T., Kim, Y. S. & Kim, S.-H. J. T. K. J. O. I. M. 2010. Familial Clustering Of Type 2 Diabetes In Korean Women With Gestational Diabetes Mellitus. 25, 269.
Riaz, M., Nawaz, A., Masood, S. N., Fawwad, A., Basit, A., Shera, A. J. C. E. & Health, G. 2018. Frequency Of Gestational Diabetes Mellitus Using Dipsi Criteria, A Study From Pakistan.
Rodrigues, M. R. K., Lima, S. A. M., Da Silvia Mazeto, G. M. F., Calderon, I. M. P., Magalhães, C. G., Ferraz, G. A. R., Molina, A. C., De Araújo Costa, R. A., Nogueira, V. D. S. N. & Rudge, M. V. C. J. P. O. 2019. Efficacy Of Vitamin D Supplementation In Gestational Diabetes Mellitus: Systematic Review And Meta-Analysis Of Randomized Trials. 14, E0213006.
Santos-Ayarzagoitia, M., Salinas-Martínez, A. M., Villarreal-Pérez, J. Z. J. D. R. & Practice, C. 2006. Gestational Diabetes: Validity Of Ada And Who Diagnostic Criteria Using Nddg As The Reference Test. 74, 322-328.
Schleger, F., Linder, K., Walter, L., Heni, M., Brändle, J., Brucker, S. Y., Pauluschke-Fröhlich, J., Weiss, M., Häring, H.-U. & Preissl, H. J. F. I. E. 2018. Family History Of Diabetes Is Associated With Delayed Fetal Postprandial Brain Activity. 9, 673.
Souza, R. T., Mayrink, J., Leite, D. F., Costa, M. L., Calderon, I. M., Rocha Filho, E. A., Vettorazzi, J., Feitosa, F. E. & Cecatti, J. G. J. C. 2019. Metabolomics Applied To Maternal And Perinatal Health: A Review Of New Frontiers With A Translation Potential. 74.
Van Leeuwen, M., Opmeer, B., Zweers, E., Van Ballegooie, E., Ter Brugge, H., De Valk, H., Visser, G., Mol, B. J. B. A. I. J. O. O. & Gynaecology 2010. Estimating The Risk Of Gestational Diabetes Mellitus: A Clinical Prediction Model Based On Patient Characteristics And Medical History. 117, 69-75.
Wahabi, H. J. J. O. D. R. 2018. Prevalence And Risk Factors For Glucose Intolerance Among Saudi Women With Gestational Diabetes. 2018.
Xie, K., Zhang, Y., Wen, J., Chen, T., Kong, J., Zhang, J., Wu, X., Hu, C., Xu, B. & Ji, C. J. J. O. D. 2019. Genetic Predisposition To Gestational Glucose Metabolism And Gestational Diabetes Mellitus Risk In A Chinese Population.
Yang, H., Wei, Y., Gao, X., Xu, X., Fan, L., He, J., Hu, Y., Liu, X., Chen, X. & Yang, Z. J. D. M. 2009. Risk Factors For Gestational Diabetes Mellitus In Chinese Women—A Prospective Study Of 16 286 Pregnant Women In China. 26, 1099-1104.


