1East Siberia State University of Technology and Management, Ulan-Ude, Russia.
2Khabarovsk State University of Economics and Law, Khabarovsk, Russia.
Corresponding autor email: shchekotova.a.v@mail.ru
Article Publishing History
Received: 08/07/2021
Accepted After Revision: 26/09/2021
The problem of an increase in the level of alimentary-dependent diseases has a global scale, and the fulfilment of tasks to solve it is included in the state programs of most developed countries, including the Russian Federation. Enrichment of food products of mass consumption with essential micronutrients is a modern, most economically profitable, effective and physiological way to improve the health of the population. The research is aimed at development of the technology of probiotic fermented milk product enriched with magnesium-containing whey protein concentrate for the prevention of alimentary-dependent diseases. The experiments were aimed at studying the effect of various doses of WPC-Mg on the main organoleptic, physicochemical and microbiological indicators of the enriched fermented milk product.
As a result of the conducted research, it has been determined that introduction of WPC-Mg to the milk base in the amount of 10% has a stimulating effect on the biochemical processes in the production of fermented milk drinks. It has been found that the structural and mechanical characteristics of WPC-Mg promote the formation of stronger intermolecular bonds in the fermented milk clot, which significantly improves the rheological characteristics of the product and makes the consistency of the drink similar to that of the products with a high mass fraction of fat. Based on the experimental data obtained, the technology of obtaining a probiotic fermented milk product enriched with magnesium and whey proteins has been developed. The obtained results open up broad perspectives for creating probiotic enriched products for functional and therapeutic nutrition.
Fermented Milk Product, Magnesium, Propionic Acid Bacteria, Whey Proteins.
Shchekotova A. V, Atlasova D. V, Khamagaeva I. S, Khazagaeva S. N, Bubeev A. T, Boiarineva I. V. Development of the Technology for Obtaining a Probiotic Fermented Milk Product Enriched with Magnesium and Whey Proteins. Biosc.Biotech.Res.Comm. 2021;14(3).
Shchekotova A. V, Atlasova D. V, Khamagaeva I. S, Khazagaeva S. N, Bubeev A. T, Boiarineva I. V. Development of the Technology for Obtaining a Probiotic Fermented Milk Product Enriched with Magnesium and Whey Proteins. Biosc.Biotech.Res.Comm. 2021;14(3). Available from: <a href=”https://bit.ly/3k3wUio“>https://bit.ly/3k3wUio</a>
Copyright © Shchekotova et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Ince-Coskun & Ozdestan-Ocak, 2020Modern physiologists increasingly attribute magnesium to the priority micronutrients for the human organism (; Trisvetova, 2012; Al Alawi et al., 2018; Glasdam et al., 2016; Guerrero-Romero et al., 2016; Sarrafzadegan et al., 2016; Farsinejad-Marj et al., 2016; Li et al., 2016; Kirkland et al., 2018; Joy et al., 2019). Being a necessary macroelement for the cells and tissues, magnesium is involved in many physiological processes that ensure normal vital activity of the organism: in the synthesis of enzymes (ATP substrate, ADP, creatine kinase, hexokinase, etc.), direct activation of enzymes, regulation of the cell membrane function (stabilization of cell membranes, cell adhesion, the transmembrane flow of electrolytes),
antagonism with calcium (muscle contraction/relaxation, the release of neurotransmitters, the excitability of the specialized cardiac conduction system), and plastic processes (synthesis of protein and catabolism, metabolism of nucleic acids and lipids, and mitochondria) (Al Alawi et al., 2018; Glasdam et al., 2016; Trisvetova, 2012). According to the WHO, magnesium deficiency takes one of the leading places in human pathologies caused by disorders of mineral metabolism (Glasdam et al., 2016; Al Alawi et al 2018; Severino et al., 2019; Hernández-Becerra et al., 2020).
Low magnesium level in the organism is associated with such diseases as osteoporosis, high blood pressure, blockage of arteries, hereditary heart diseases, diabetes, and apoplectic attack (Al Alawi et al., 2018; Glasdam et al., 2016; Guerrero-Romero et al., 2016; Sarrafzadegan et al., 2016; Farsinejad-Marj et al., 2016; Li et al., 2016; Kirkland et al., 2018; Hernández-Becerra et al., 2020; Joy et al., 2019; Trisvetova, 2012, Ince-Coskun & Ozdestan-Ocak, 2020).
Like other macroelements, magnesium is received with food and water. The need for magnesium cannot always be satisfied through nutrition. In this case, mineral supplements and magnesium-containing preparations are prescribed. The effectiveness of drugs containing magnesium depends mainly on two factors: the amount of “elemental” magnesium in the compound and its bioavailability (the ability to be assimilated in the organism). High bioavailability is characteristic of chelated forms of magnesium — compounds of magnesium with amino acids (Glasdam et al., 2016; Al Alawi et al., 2018; Severino et al., 2019; Hernández-Becerra et al., 2020).
Whey proteins are advised for consumption by the authors as a source of amino acids for obtaining chelate complexes. By their amino acid composition, whey proteins are among the most valuable proteins of animal origin (they are sources of essential amino acids, exhibit immunomodulatory, antagonistic, and anticarcinogenic activity; they are responsible for transporting fat-soluble vitamins and microelements in the organism) (Gordienko et al., 2015; Nechaev et al., 2007; Khramtsov, 2011). Whey proteins contain significant amounts of branched-chain amino acids and are physiologically beneficial: for example, consuming whey proteins in combination with power training accelerates fat loss in humans (Wang et al., 2020; Lockwood et al., 2017).
Besides, whey proteins are widely used for technological purposes, such as forming gels (Egan et al., 2014; Oztop, 2014), changing viscosity (Patocka et al., 2006), and fat substitution (Akalın et al., 2008). Chelated complexes of magnesium with amino acids from whey proteins are obtained through thermal denaturation with the use of magnesium salt as a coagulant, followed by fermentation of the protein mass by probiotic cultures (Shchekotova & Khamagaeva, 2017; Ince-Coskun & Ozdestan-Ocak, 2020).
The studies performed in recent years have clearly shown that probiotics have a beneficial effect on gut microbiota and mineral metabolism (Skrypnik & Suliburska, 2018). Microflora is involved in the metabolism of many micro- and macroelements, including magnesium (Skrypnik & Suliburska, 2018). Biotechnological processing of whey proteins with probiotic cultures will improve their functional properties after thermomagnesium precipitation. Normalization of the intestinal microflora will cause acidification of the medium in the large intestine and ensure better magnesium absorption (Glasdam et al., 2016; Skrypnik & Suliburska, 2018; Al Alawi et al., 2018; Hernández-Becerra et al., 2020).
The presented literature data show that joint enrichment of dairy products with magnesium, WPC, and probiotic cultures will allow obtaining functional food for various purposes, including the prevention of nutrition-related diseases. The work is aimed at developing a technology for obtaining a probiotic fermented milk product enriched with the magnesium-containing WPC.
MATERIAL AND METHODS
Experimental studies were performed at the Technology of Dairy Products. Merchandising and Examination of Goods Department of the HE FSBEI East Siberia State University of Technology and Management (ESSUTM) in Ulan-Ude, Russia, during the period from May to December 2019.
The objects of research at different stages were whole milk, fermented WPC-Mg, probiotic fermented milk product. Pure cultures of Propionibacterium freundenreichii subsp. freundenreichii AC-2585 obtained from the All-Russian Collection of Industrial Microorganisms of Federal Institution “State Research Institute of Genetics and Selection of Industrial Microorganisms of the National Research Center” Kurchatov Institute” (Russia) were used to obtain probiotic yeast.
The fermented WPC-Mg was used for enriching the fermented milk drink. Unclarified curd whey was used as the raw material for the production of the fermented WPC that was obtained by thermal coagulation with the addition of magnesium salt as a coagulant, followed by the fermentation of proteins with propionic acid bacteria of the P. freundenreichii subsp. freundenreichii species and drying. Before the introduction, WPC-Mg was preliminarily dissolved in a small amount of pasteurized milk cooled to (60 – 65) 0C. The content of magnesium in the fermented WPC was 236 ± 0.7 mg/100g, the mass fraction of moisture was 70 – 80%, and the mass fraction of protein was 13 ± 0.6%.
Cow’s raw milk was used for the production of fermented milk product. The technological process for the production of the enriched fermented milk product included milk acceptance, purification, heating, normalization to mass fraction of fat of 2.5%, homogenization, pasteurization at (93 ± 2) 0C for (15-20) sec, cooling of normalized mixture up to (30 ± 2) 0C, introduction of WPC-Mg, fermentation of the mixture with 5% starter culture until the acidity reached (70-90) 0 Т, cooling and bottling. In order to exclude undesirable relationships among microorganisms, fermentation of the normalized mixture was carried out with an active ferment based on the same probiotic cultures that were used for fermentation of WPC-Mg – P. freundenreichii subsp. freundenreichii AC-2585.
The research scheme included study of the effect of various doses of fermented WPC-Mg in a milk base on the fermentation process of fermented milk product (assessment of titratable acidity and the number of viable cells of propionic acid bacteria); assessment of the organoleptic properties of fermented milk product with different content of WPC-Mg (taste, smell, color, consistency); study of the effect of WPC-Mg on the structural, mechanical and rheological characteristics of fermented milk clots (assessment of dynamic viscosity, clot density, degree of seneresis); establishment of shelf life and quality indicators of the enriched fermented milk product; development of technology for a probiotic fermented milk product enriched with whey proteins and magnesium.
When performing the experimental part of the work, standard and generally accepted methods of physicochemical, organoleptic, microbiological analysis were used. Organoleptic indicators were determined visually, as well as by smelling and tasting the product. The titratable acidity was determined by titration: the method was based on the neutralization of the acids contained in the product with sodium hydroxide solution in the presence of phenolphthalein indicator.
The rheological characteristics of acid clots were determined on a Brookfield RVDV-II + Pro rotational viscometer (United States, Brookfield Engineering Labs. Inc., 2009). The clot density was determined by measuring the immersion depth of a plate with certain weight and area, exerting pressure on the clot for (30-60) sec (Krekker et al., 2016). The plate with the weight of 12.4 g and the base area of 1.6 cm3 was used in the experiment. The clot density was calculated by the equation: D = (0.5 • q • hc) / (d • hn), where D was the clot density, g/cm3, q was the load created by the plate (weight of the plate, g), hc was the clot height in the glass (mm), d was the plate base area (cm2), hn was the plate immersion depth (mm).
The syneresis was determined by the filtration method through measuring the amount of whey released during filtering 100 cm3 of the decomposed clot through a paper filter for eight hours at room temperature.The mass fraction of magnesium was determined by the method of capillary electrophoresis on a Kapel-105M device (Russia, St. Petersburg, Lumex-Marketing LLC, 2012). The method was based on sample dilution, further separation, identification and quantitative determination of the mass concentration of magnesium (mg/L) by capillary electrophoresis (Lumex, 2013).
The mass fraction of fat in the enriched product was determined by the acid-butyrometric method based on the separation of fat from the fermented milk product under the action of concentrated sulfuric acid and isoamyl alcohol, followed by centrifugation and measurement of the volume of released fat in the graduated part of the butyrometer (Gosstandart of the USSR, 1990).
The mass fraction of protein was determined by the Kjeldahl method. The method was based on the mineralization of the analyzed product sample with concentrated sulfuric acid in the presence of a catalyst with the formation of ammonium sulfate, its conversion into ammonia, distillation of the latter into a boric acid solution, quantitative accounting of ammonia by the titrimetric method and calculation of the mass fraction of protein in the analyzed sample (Rosstandart, 2018).
The number of cells of propionic acid microorganisms was determined by the method of limiting dilutions (Rosstandart, 2014b). The method was based on sowing propionic acid bacteria in certain dilutions in (on) selective nutrient media for submerged cultivation, their cultivation at a temperature of (30 – 1) 0C for 48 hours with limited oxygen access and subsequent quantitative calculation of the content of propionic acid bacteria in the product.Bacteria of the E. coli group were determined by the signs of growth in liquid Kessler medium (Rosstandart, 2014a). Yeast and molds were determined by sowing the product on a solid nutrient medium (Sabouraud’s agar) (Rosstandart, 2015).
All experiments were carried out 3-5 times. The data obtained were processed using personal computer in Microsoft Excel 14 with the calculation of arithmetic mean values and corresponding errors (M ± m). The significance of differences between the compared indicators in the groups was assessed by Student’s t-test. Differences were considered statistically significant at р<0,05. Graphical dependencies in the figures were presented after the experimental data processing. Calculations, plotting and specification of diagrams were performed using Microsoft Office 14 and Excel 14 applications on Windows 10.
RESULTS AND DISCUSSION
At the first stage of the studies, the authors studied the effect of fermented WPC-Mg in a milk base on the fermentation process of the fermented milk product. WPC-Mg was introduced into normalized milk after heat treatment in the amounts of 5, 10, and 15%. The ripening process was monitored by the increase in acidity Fig. (1a) and the growth of propionic acid bacteria Fig. (1b) in the product. During ripening, the authors tracked the time required for the acidity of the tested samples of the product to reach (70 – 90) 0T. The reference was normalized milk with the fat mass fraction of 2.5%.
Figure 1: The effect of the mass fraction of fermented WPC-Mg on the dynamics of acid formation in the normalized mixture and the growth of propionic acid bacteria in the fermented milk product
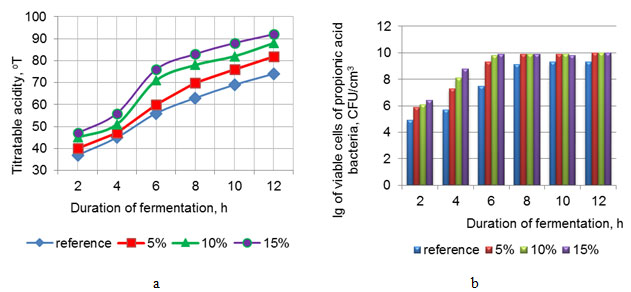
Fig. (1) shows that adding WPC-Mg accelerated the fermentation process in the normalized mixture of the fermented milk product. The required acidity (70 – 90 °T) of the fermented milk product clot was reached after six hours of cultivation with the addition of 10 and 15% of WPC-Mg Fig. (1a). An increase in acid formation with increasing the dose of WPC-Mg in the tested samples was explained by the change of the buffer capacity of the mixture and its enrichment with additional sources of nitrogenous nutrition and the macroelement for propionic acid bacteria (Skrypnik & Suliburska, 2018; Cousin et al., 2011; Begunova et al., 2019; Vorobjeva et al., 2008). An intensive increase in the titratable acidity with the introduction of WPC-Mg at a dosage of 10 and 15% allowed shortening the process by (2 – 4) hours, compared to the reference Fig. (1a).
Quantitative accounting of the probiotic cultures showed more intensive growth of microorganisms in the milk with the addition of WPC-Mg Fig. (1b). The number of viable cells of propionic acid bacteria in the experimental samples with WPC-Mg after six hours of cultivation was (2·109 – 9·109) CFU/cm3, which was two orders of magnitude greater than in the reference Fig. (1b). The high biological value of whey proteins probably created favorable conditions for the development of propionic bacteria. The presence of lactose, peptides, and free amino acids in WPC-Mg provided the possibility of the propionic bacteria’s faster growth during cultivation.
Thus, summarizing the results obtained, a conclusion can be drawn that adding WPC-Mg to normalized milk intensifies the fermentation process and increases the number of probiotic microorganisms in the fermented milk product. This fact indicates the prebiotic properties of the fermented WPCs enriched with magnesium. It was noted that a significant increase in the titratable acidity and the number of viable cells of propionic acid bacteria had been observed after adding 10 – 15% of WPC-Mg to the normalized mixture. The joint presence of whey proteins and the macro element in WPC-Mg contributes to a synergistic effect, enhancing their positive effect on the activity of propionic acid bacteria in the fermented milk product. This fact opens up wide opportunities for using WPC-Mg in the fermented milk product technology, providing symbiotic properties and functional orientation to it.
According to the literature (Al Alawi et al., 2018; Glasdam et al., 2016; Guerrero-Romero et al., 2016; Sarrafzadegan et al., 2016), the process of product enrichment with various magnesium-containing salts and additives may affect the organoleptic properties of the product. In this regard, in further experiments, the organoleptic properties of the test samples with various contents of WPC-Mg were assessed Table (1).
Table 1. Organoleptic assessment of the milk base after the introduction of various amounts of WPS-Mg
| The studied property | The dose of WPC introduced | |||
| reference | 5% | 10% | 15% | |
| taste and odor | the taste and odor are clean with sour milk aftertaste | the taste and smell are clean, with a sour milk aftertaste and a subtly bitter flavor | the taste and smell are those of fermented milk with a pronounced bitter aftertaste | |
| color | cream | |||
| consistency | Liquid, homogeneous, glossy, and delicate | homogeneous, glossy, and thickish | homogeneous, glossy, and thick | |
The analysis of the data in Table (1) showed that the introduction of fermented WPC-Mg to the fermented milk drink affected the taste of the product: a bitter aftertaste appeared. After the introduction of (5 – 10) % of WPC-Mg, this change in the taste was barely noticeable and was not a defect; with increasing the dose, the bitter taste increased, which significantly reduced the consumer properties of the fermented milk drink. This defect may be explained by the bitter taste of magnesium salts. It should be noted that in all studied samples, the introduction of fermented WPC-Mg improved the consistency of the products. This fact is especially important for producing fermented milk drinks with a low mass fraction of fat. The introduction of WPC-Mg ensured the consistency of the drink similar to that of the products with a high mass fraction of fat, even without the use of stabilizing systems.
An objective assessment of the consistency of fermented milk products was provided by the rheological properties that were determined by the type of structure and mechanical properties of the product. These properties were sensitive to the changes in the chemical composition of the product, physical parameters, and processing conditions (Ababkova et al., 2016). In this regard, the next series of experiments was devoted to studying the effect of WPC-Mg on the structural, mechanical, and rheological properties of fermented milk clots Table (2).
Table 2. The effect of WPC-Mg on the structural, mechanical, and rheological properties of fermented milk clots
| The studied property | The dose of WPC introduced | |||
| reference | 5% | 10% | 15% | |
| Dynamic viscosity, Pa•s | 33 ± 0.3 | 38 ± 0.5 | 42 ± 0.2 | 47 ± 0.3 |
| Clot density, 102 g/cm3 | 1.20 ± 0.01 | 1.36 ± 0.03 | 1.78 ± 0.02 | 1.96 ± 0.02 |
| Syneresis degree, % | 67 ± 0.5 | 59 ± 0.2 | 52 ± 0.4 | 40 ± 0.3 |
The analysis of the data in Table (2) showed that the presence of WPC-Mg in the product contributed to forming stronger bonds between the structural elements of the fermented milk clot. This was confirmed by the 0.8 – 1.4 times increased viscosity and 1.1 – 1.6 times increased strength of the resulting lumps. With increasing the dose of introduced WPC-Mg, a decrease in the acid clots syneresis ability was observed, compared to the reference (Table (2). In the studied samples, the degree of syneresis decreased from 67% to 40%.
The high water-binding capacity of WPC-Mg was explained by the presence of amino acids that adsorbed water from hydrophilic elements. Usually, hydration of the native whey proteins is weak, however, the thermal denaturation during WPC-Mg production might have significantly increased this ability, which had a positive effect on the water-binding ability of the fermented milk clots of the product.
It should be noted that samples of fermented milk products enriched with WPC-Mg retained uniformity of consistency and a high number of viable cells (108 – 109 CFU/cm3) during storage (for 10 – 12 days), in contrast to the reference sample. The homogeneity of the consistency of the samples with the WPC, compared to the reference, was explained by the stabilizing properties of whey proteins, which had a water-holding ability and improved the quality of the products and their storage life.
The obtained results allowed concluding that fermented WPC-Mg not only enriched the fermented milk drink with protein, easily digestible chelated magnesium, and probiotic cultures, but also intensified the production process, prolonged the shelf life of the product, and improved its structural and mechanical properties, which fact was especially important in the production of fermented milk drinks with low fat content. A comprehensive study of the organoleptic, physicochemical, and rheological parameters of the fermented milk drink made it possible to conclude that a sample with the 10% content of WPC-Mg had the best consumer properties.
Within the study, a technology for the production of a probiotic fermented milk product enriched with magnesium and whey proteins was developed. The process envisaged the milk acceptance, purification, normalization, homogenization, heat treatment and introduction of WPC-Mg into normalized milk in the amount of 10%. This method of introduction was explained by the fact that in the case of using the fermented WPC, subsequent pasteurization of the mixture was not advisable (due to the death of probiotic cultures),
as well as homogenization, which could affect the structure of the whey concentrates after mixing the components. This was followed by fermentation of a mixture of 5% starter culture, fermentation, cooling and bottling. The ripening time according to the developed technology was only 6-7 hours. The enriched fermented milk product was characterized by good organoleptic properties and contained a high number of viable cells of propionic acid bacteria (109 CFU/g).
The qualitative characteristics of the developed fermented milk product enriched with WPC-Mg are shown in Table (3).
Table 3. The qualitative characteristics of the enriched fermented milk drink
| Indicators | Characteristic | |
| Appearance and consistency | Thick, stretchy, glossy, and homogeneous consistency | |
| Taste and odor | The taste and odor are clean, of fermented milk, with a light, barely noticeable, bitter aftertaste | |
| Color | Milky-white, homogeneous, with a creamy shade | |
| Mass fraction of fat, % not less than | 2.5 ± 0.01 | |
| Mass fraction of protein, % not less than | 3.4 ± 0.03 | |
| Mass fraction of nonfat milk solids, % not less than | 7.8 ± 0.04 | |
| Acidity, oT, not more than | 89 ± 0.2 | |
| Mass fraction of magnesium, mg/l, not less than | 256 ± 0.5 | |
| The number of propionic acid bacteria cells, CFU/cm3 | (7 – 9) ·109 | |
| The volume (cm3) in which are not allowed: | Coliform bacteria | 0.1 |
| pathogenic (including salmonella) | 25 | |
| S. aureus staphylococci | 1 | |
| L. monocytogenes listeria | – | |
| Yeast, mold, CFU/cm3 (g), not more than | D-50, P-50 | |
The data in Table 3 show that the obtained fermented milk product had good organoleptic properties, contained a prophylactic dose of magnesium in an easily digestible form, and had high protein content and a great number of viable cells of propionic acid bacteria. The consumption of 0.25 liters of the developed product will satisfy the daily need of an adult in macro elements by (16 – 18) %, and consumption of 0.5 liters — by (32 – 36) %, respectively. These values are within the safe levels of product enrichment for magnesium (10 – 40%), as recommended by the leading nutritionists and physicians.
At present, a number of effective dairy products enriched with WPC are available in our country and abroad (Khramtsov & Nesterenko, 2004; Lawrence, 1993; Lelievre, 1990; Cozzolino, 2003; Patocka, 2006; Smirnova et al., 2014; Lagrange et al., 2015; Henriques et al., 2012, 2017; Nastaj et al., 2020).
Protein concentrates, isolated by various expensive membrane methods, prevail among the used WPCs. These concentrates, with all their advantages, have one significant drawback – high allergenic activity (Kattan et al., 2011; Botteman & Detzel, 2016; Vonk, 2017; Abbring, 2020).
In this work, to enrich a fermented milk product, the authors propose to use WPC obtained by thermal coagulation with the addition of magnesium salt as a coagulant, followed by fermentation of protein clots with probiotic cultures. Biotechnological processing of WPC-Mg using propionic acid bacteria allows increasing the functional properties of the protein concentrate obtained and reducing the allergenic effect of whey proteins.
It should be noted that in the literature, the authors did not find data on the production of fermented WPC simultaneously enriched with probiotic cultures and any essential elements. Therefore, the use of fermented concentrates for the enrichment of dairy products, obtained by the method of thermal coagulation with the addition of magnesium salt as a coagulant, is a relevant and cost-effective solution (Minj & Anand, 2020)
The proposed biotechnological methods for obtaining a fermented milk product can shorten the production process and significantly improve the quality indicators of the product. The authors have proved the stimulating effect of fermented WPC-Mg on biochemical processes in the production of a fermented milk product: acid formation during fermentation, improvement of structural, mechanical, rheological characteristics and shelf life. The enriched product, developed according to the proposed technology, is of greater importance in dietary nutrition.
The introduction of probiotic fermented milk drinks enriched with magnesium and whey proteins into production and their promotion on the market will significantly expand the range of products for the prevention and correction of alimentary-dependent diseases, as well as allow implementing the principle of waste-free production at dairy enterprises and reducing environmental pollution as a result of utilization of whey protein.
CONCLUSION
As a result of the studies, a new technology for producing the probiotic fermented milk drink has been developed, which has made it possible to obtain an enriched dairy product with functional properties. It has been found that the use of WPC-Mg in the production of the fermented milk drink not only enriches it with an easily digestible macro element and whey proteins but also intensifies the fermentation process and increases the number of probiotic microorganisms in the fermented milk product. This fact is the evidence of prebiotic properties of the fermented WPCs enriched with magnesium. This fact opens up wide opportunities for using WPC-Mg in the fermented milk product technology, providing symbiotic properties and functional orientation to it.
The introduction of WPC-Mg into the milk base improves the structural and mechanical properties of the finished product: the density and viscosity of the fermented milk clots increase, and the syneresis slows down. This circumstance is of particular importance in the production of fermented milk drinks with low fat content since it allows excluding or significantly reducing the number of stabilizers and/or thickeners used in such cases.
The use of the fermented WPC in the production of the fermented milk drink has allowed increasing the shelf life by up to 10 – 12 days without significant changes in the organoleptic, microbiological, and structural and mechanical properties, which increases the economic efficiency of the developed product.
ACKNOWLEDGEMENTS
The study has been performed with the financial support from the Fund for Assistance to the Development of Small Enterprises in Science and Technology (Fund for Innovation Assistance) within the framework of a scientific project under the Umnik program, 2019 (Russia).
REFERENCES
Ababkova, A.A., Neronova, E.Y., & Novokshanova, A.L. (2016). Studying the rheological characteristics of fermented milk clots enriched with hydrolyzate of whey proteins. Dairy Bulletin. 3(23), 37-45 (in Russian).
Abbring, S. (2020). Loss of allergy-protective capacity of raw cow’s milk after heat treatment coincides with loss of immunologically active whey proteins. Food & Function, 11(6), 4982-4993.
Akalın, A.S., Karagözlü, C., & Ünal, G. (2008). Rheological properties of reduced-fat and low-fat ice cream containing whey protein isolate and inulin. European Food Research and Technology, 227(3), 889-895.
Akhmetova, S.V. (2015). Peculiarities of the nutrition of the persons with the risk of developing non-infectious nutrition-related diseases. Nutrition issues, 84(55), 14–14 (in Russian).
Al Alawi, A.M., Majoni, S.W., & Falhammar, H. (2018). Magnesium and human health: perspectives and research directions. International Journal of Endocrinology, 2018, 1-17. https://doi.org/10.1155/2018/9041694
Begunova, A.V., Rozhkova, I.V., Zvereva, E.A., Glazunova, O.A., & Fedorova, T.V. (2019). Lactic and Propionic Acid Bacteria: The Formation of a Community for the Production of Functional Products with Bifidogenic and Hypotensitive Properties. Applied Biochemistry and Microbiology, 55(6), 660-669.
Botteman, M., & Detzel, P. (2016). Cost-effectiveness of partially hydrolyzed whey protein formula in the primary prevention of atopic dermatitis in high-risk urban infants in Southeast Asia. Annals of Nutrition and Metabolism, 66(Suppl. 1), 26-32.
Cousin, F.J., Mater, D.D.G., Foligné, B., & Jan, G. (2011). Dairy propionibacteria as human probiotics: a review of recent evidence. Dairy Science & Technology, 91(1), 1-26.
Cozzolino, A. (2003). Incorporation of whey proteins into cheese curd by using transglutaminase. Biotechnology and applied biochemistry, 38(3), 289-295.
Egan, T., Jacquier, J.C., Rosenberg, Y., & Rosenberg, M. (2014). Cold-set whey protein microgels containing immobilised lipid phases to modulate matrix digestion and release of a water-soluble bioactive. Journal of microencapsulation, 31(2), 184-192.
Farsinejad-Marj, M., Saneei, P., & Esmaillzadeh, A. (2016). Dietary magnesium intake, bone mineral density and risk of fracture: a systematic review and meta-analysis. Osteoporosis International, 27, 1389–1399.
Glasdam, S.M., Glasdam, S., & Peters, G.H. (2016). The Importance of Magnesium in the Human Body: A Systematic Literature Review. Advances in Clinical Chemistry, 73, 169-93.
Gordienko, L.A., Evdokimov, I.A., & Gorlacheva, S.V. (2015). The use of whey proteins in the production of fermented milk drinks. Dairy industry, 3, 72–73 (in Russian).
Gosstandart of the USSR. (1990). GOST 5867-90 Milk and dairy products. Methods of determination of fat. Standartinform, Moscow (in Russian). http://docs.cntd.ru/document/1200021592
Guerrero-Romero, F., Flores-García, A., Saldaña-Guerrero, S., Simental-Mendía, L.E., & Rodríguez-Morán, M. (2016). Obesity and hypomagnesemia. European journal of internal medicine, 34, 29-33.
Henriques, M., Gomes, D., & Pereira, C. (2012). Effects of liquid whey protein concentrate on functional and sensorial properties of set yoghurts and fresh cheese. Food and Bioprocess Technology, 6(4), 952-963
Henriques, M., Gomes, D., Pereira, C. (2017). Liquid whey protein concentrates produced by ultrafiltration as primary raw materials for thermal dairy gels. Food Technology and Biotechnology, 55(4), 454-463.
Hernández-Becerra, E., Jímenez-Mendoza, D., Mutis-Gonzalez, N., Pineda-Gomez, P., Rojas-Molina, I., & Rodríguez-García, M.E. (2020). Calcium Deficiency in Diet Decreases the Magnesium Content in Bone and Affects Femur Physicochemical Properties in Growing Rats. Biological Trace Element Research, 197, 224–232.
Ince-Coskun, A.E., & Ozdestan-Ocak, O. (2020). Effects of salt ions and heating on the behaviour of whey protein particle dispersions. Food Hydrocolloids, 101, 105433.
Joy, S.S., George, T.P., & Siddiqui, K. (2019). Low magnesium level as an indicator of poor glycemic control in type 2 diabetic patients with complications. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 13(2), 1303-1307.
Kattan, J.D., Cocco, R.R., & Järvinen, K.M. (2011). Milk and soy allergy. Pediatric Clinics, 58(2), 407-426.
Khramtsov, A.G. (2011). The whey phenomenon. Profession, St. Petersburg (in Russian).
Khramtsov, A.G., & Nesterenko, P.G. (2004). Technology of products from milk whey: study guide. DeLi print, Moscow.
Kim, M.N. (2016). Trends in the development of nutrition-related diseases and the role of functional products in disease prevention. Eurasian Union of Scientists, 1-2(22), 65-68 (in Russian).
Kirkland, A.E., Sarlo, G.L., & Holton, K.F. (2018). The role of magnesium in neurological disorders. Nutrients, 10(6), 730.
Krekker, L.G., Gomboyeva, Zh.B., & Muruyev, I.E. (2016). Investigation of the effect of magnesium salts on the rheological properties of rennet clots. In: Innovative technologies of food products and assessment of their quality: science, education, production. Materials of the I International Scientific and Technical Conference, June 15, 2016, Ulan-Ude, Russia (pp. 72-77). VSGUTU Publishing House (in Russian).
Lagrange, V., Whitsett, D., & Burris, C. (2015). Global market for dairy proteins. Journal of food science, 80(S1), A16-A22.
Lawrence, R.J. (1993). Incorporations of whey proteins in cheese. In: Factors affecting the yield of cheese, IDF Special Issue 9301 (pp. 79-87). Int. Dairy Fed., Brussels.
Lelievre, J. (1990). Incorporation of size-reduced dispersions of whey proteins into Mozzarella cheese. In: Brief Communications and Abstracts of Posters, XXIII Int. Dairy Congress, Montreal, Canada, October 8-12, 1990 (p. 517). Int. Dairy Fed., Brussels.
Li, Y., Yue, J., & Yang, C. (2016). Unraveling the role of Mg++ in osteoarthritis. Life sciences, 147, 24-29.
Lockwood, C.M., Roberts, M.D., Dalbo, V.J., Smith-Ryan, A.E., Kendall, K.L., Moon, J.R., & Stout, J.R. (2017). Effects of hydrolyzed whey versus other whey protein supplements on the physiological response to 8 weeks of resistance exercise in college-aged males. Journal of the American College of Nutrition, 36(1), 16-27.
Lumex. (2013). Method M 04-52-2008. Determination of inorganic cations in beverages (in Russiam). https://www.lumex.ru/metodics/20ARU03.01.04-1.pdf
Minj, S., & Anand, S. (2020). Whey Proteins and Its Derivatives: Bioactivity, Functionality, and Current Applications. Dairy, 1(3), 233-258.
Nastaj, M., Terpiłowski, K., & Sołowiej, B.G. (2020). The effect of native and polymerised whey protein isolate addition on surface and microstructural properties of processed cheeses and their meltability determined by Turbiscan. International Journal of Food Science & Technology, 55(5), 2179-2187.
Nechaev, P.A., Traubenberg, S.E., Kochetkova, A.A., Kolpakova, V.V., Vitol, I.S., & Kobeleva, I.B. (2007). Pishchevaya khimiya [Food chemistry]. GIORD, St. Petersburg (in Russian).
Oztop, M.H. (2014). Monitoring the effects of divalent ions (Mn+ 2 and Ca+ 2) in heat-set whey protein gels. LWT-Food Science and Technology, 56(1), 93-100.
Patocka, G. (2006). Rheological behaviour of dairy products as affected by soluble whey protein isolate. International dairy journal, 16(5), 399-405.
Patocka, G., Cervenkova, R., Narine, S., & Jelen, P. (2006). Rheological behaviour of dairy products as affected by soluble whey protein isolate. International dairy journal, 16(5), 399-405.
Rosstandart. (2014a). GOST 32901-2014 Milk and milk products. Methods of microbiological analysis. Standartinform, Moscow (in Russian). http://docs.cntd.ru/document/1200115745
Rosstandart. (2014b). GOST R 56139-2014 Functional foods. Methods for detection and enumeration of probiotic microorganisms. Standartinform, Moscow (in Russian). http://docs.cntd.ru/document/1200115455
Rosstandart. (2015). GOST 33566-2015 Milk and dairy products. Determination of yeast and molds. Standartinform, Moscow (in Russian). http://docs.cntd.ru/document/1200127751
Rosstandart. (2018). GOST 34454-2018 Dairy products. Determination of protein content by the Kjeldahl method. Standartinform, Moscow (in Russian). http://docs.cntd.ru/document/1200160723
Samodurova, N.Y., Mamchik, N.P., Istomin, A.V., Klepikov, O.V., & Sokolenko, G.G. (2018). Determination of the risk areas by the level of nutrition-related diseases given the regional characteristics of the nutritional structure of the population. Bulletin of the Russian State Medical University, 5, 42–47.
Sarrafzadegan, N., Khosravi-Boroujeni, H., Lotfizadeh, M., Pourmogaddas, A., & Salehi-Abargouei, A. (2016). Magnesium status and the metabolic syndrome: A systematic review and meta-analysis. Nutrition, 32(4), 409-417.
Severino, P., Netti, L., Mariani, M.V., Maraone, A., D’Amato, A., Scarpati, R., Infusino, F., Pucci, M., Lavalle, C., Maestrini, V., Mancone, M., & Fedele, F. (2019). Prevention of Cardiovascular Disease: Screening for Magnesium Deficiency. Cardiology Research and Practice, 2019, 4874921.
Shchekotova, A.V., & Khamagaeva, I.S. (2017). Protein concentrates enriched with probiotic cultures and essential micro- and macroelements. monograph. East Siberia State University of Technology and Management Publishing House, Ulan-Ude (in Russian).
Skrypnik, K., & Suliburska, J. (2018). Association between the gut microbiota and mineral metabolism. Journal of the Science of Food and Agriculture, 98(7), 2449-2460.
Smirnova, I.A., Lobacheva, Ye.M, & Gulbani, A.D. (2014). Use of whey protein microparticulate in dairy products. Molochnaya promyshlennost, 6, 12-14.
Starodubov, V.I., Marczak, L.B., Varavikova, E., Bikbov, B., Ermakov, S.P., Gall, J., Glenn, S.D., Griswold, M., Idrisov, B., Kravchenko, M., Lioznov, D., Loyola, E., Rakovac, I., Vladimirov, S.K., Vlassov, V., Murray, C.J.L., & Naghavi, M. (2018). The burden of disease in Russia from 1980 to 2016: a systematic analysis for the Global Burden of Disease Study 2016. The Lancet, 392(10153), 1138-1146. http://dx.doi.org/10.1016/S0140-6736(18)31485-5
Trisvetova, E.L. (2012). Magnesium in clinical practice. Rational Pharmacotherapy in Cardiology, 8(4), 545-552 (in Russian).
Vonk, M.M. (2017). The efficacy of oral and subcutaneous antigen-specific immunotherapy in murine cow’s milk-and peanut allergy models. Clinical and Translational Allergy, 7(1), 1-13.
Vorobjeva, L.I., Khodjaev, E.Yu., & Vorobjeva, N.V. (2008). Propionic Acid Bacteria as Probiotics. Microbial Ecology in Health and Disease, 20(2), 109-112.
Wang, J., Furtado, G.F., Monthean, N., Dupont, D., Pédrono, F., & Madadlou, A. (2020). CaCl2 supplementation of hydrophobised whey proteins: Assessment of protein particles and consequent emulsions. International Dairy Journal, 110, 104815.


