1Institute of Biotechnology and Genetic Engineering (IBGE), University of Sindh, Jamshoro-76080, Pakistan.
2Department of Statistics. University of Sindh, Jamshoro-76080, Pakistan.
3Dr. Panjwani Centre for Molecular Medicine and Drug Research, International Centre for Chemical and
Biological Sciences, University of Karachi, Karachi-70802, Pakistan
4Institute of physiotherapy and rehabilitation sciences Liaquat University of Medical & Health Sciences
(LUMHS), Jamshoro-76090, Pakistan.
Corresponding author email: munzihyder@yahoo.com
Article Publishing History
Received: 20/04/2020
Accepted After Revision: 22/06/2020
The α-amylases (EC 3.2.1.1; 1,4-α-D-glucan glucanohydrolase) are very important commercial industrial enzyme produced by various microorganisms. Aim of present study is to analyze the production of crude amylase including other hydrolytic enzymes (xylanases and pectinases) in Bacillus subtilis LC4 nutrient culture supplemented with banana agriculture waste as a fermentation substrate. The B. subtilis culture was grown on TY [trypton (10 g L-1), yeast extract (5 g L-1), NaCl (5 g L-1)] medium as used as a control medium (LB₀). Two levels of extracts (15% and 30%) of different banana parts (leaf, leaf sheath, floral leaflets, banana peel and sucker) were maintained in LB₁ (⅛ LB₀) medium. Maximum cell growth was observed in LB₂ₐ (2.167±0.120) supplemented with leaf blade as a substrate and LB₀ (1.570±0.069) control TY-medium. The cultures were harvested after 12-hrs of incubation. Production of enzymes and complex banana substrate saccharification into simple soluble sugars was observed. Maximum soluble sugars (10.06±0.188 mg ml-1) in LB₅ₐ and reducing sugars (6.217±0.097 mg ml-1) in LB₆ₐ noted than other cultures (p≤0.05), while total proteins in LB₇ₐ and prolines in LB₃ₐ, which remined lower than LB₀ (TY-medium). The hydrolytic enzymes like as α-amylase showed highest activity in LB₂ₐ (leaf blade) and LB₄ₐ (fruit peel), xylanase in LB₂ₐ (leaf blade) and LB₇ₐ (sucker), while pectinase in LB₆ₐ (fruit peel) and in LB₇ₐ. The above outcomes have shown that cheapest agro-banana industry wastes are promising fermentation substrate to produce low cost industrial enzymes as well as secondary metabolites.
Bacillus Subtilis Lc4, Saccharification, Agro-Banana Wastes, Amylases, Sub-Merged Fermentation
Haq I-U, Jatoi I, Gill N. P, Mirza M. R, Sangrasi S. A. Biosynthesis of Alpha-Amylase and Other Crude Enzymes in Bacillus Subtilis Culture Rich with Banana Organ Specific Cultures. Biosc.Biotech.Res.Comm. 2020;13(2).
Haq I-U, Jatoi I, Gill N. P, Mirza M. R, Sangrasi S. A. Biosynthesis of Alpha-Amylase and Other Crude Enzymes in Bacillus Subtilis Culture Rich with Banana Organ Specific Cultures. Biosc.Biotech.Res.Comm. 2020;13(2). Available from: https://bit.ly/2A6Skbj
Copyright © Haq et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Banana (Musa spp.) is grown extensively since earliest human agriculture (Frison and Sharrock, 1998). It produces agriculture wastes in the form of fruit peel, pseudostem, leaves and sucker, which are posing serious environmental problems due to its improper management (Essien et al 2005). Meanwhile, its wastes are rich with fermentable proteins and starch (Mohiuddin et al., 2014) could be useful in production of biogas (Tewari et al., 1986), ethanol (Itelima et al., 1992), methane (Li et al., 2016) and lactic acid (López‐Bac and Gómez, 1992). Banana waste may also be useful for production of low-cost industrial enzymes (Barredo, 2005; Patel et al., 2012 Ahmad, and Danish, 2018).
The amylases are the most important textile industries enzymes it is also useful in other industries including food, paper, pharmacy and detergent (Walker and Dundee, 2009; Demirkan, 2011). Its in-vitro production with synthetic cell nutrient medium is appeared costly that’s why short-cut in cost of amylase production is required. Agriculture wastes are the major low-cost available plant polysaccharide such as starch, lignocelluloses and chitins on earth. Being hydrolyzed with extra-cellular α-amylase or glycogenase into dixtrins than smaller oligomers and finally glucose molecules (Demirkan et al., 2005). These extra-cellular enzymes including amylases are produces by microorganisms for their nutrition. Indeed, the secretion of amylases has been reported as in archae (Kwak et al., 1998), actinomycetes (Kar et al., 2008), bacteria (Agüloğlu et al., 2014: Slavić et al., 2016) and fungi cells (Sahnoun et al., 2012) under aseptic conditions.
The α-amylase could be obtained from higher organisms such as plants and animals. Like as, it has been isolated from barley and rice (Oboh, 2005) but commercially not available because of complex downstream purification from the cultures of higher organisms. The α-amylases isolated from various microbial genotypes have shown diverse performances under normal to harsh physical condition (Vengadaramana, 2013). However, microorganisms are playing very important role in efficient production of α-amylases due to their rapid growth rates and being genetically manipulate-able (Dumorné et al., 2017).Microbial enzymes are useful under industrial conditions because of their low cost availability, large productivity and stability under harsh chemical conditions with variable plasticity in their activity (Rana et al., 2013). The B. subtilis is well known soil growing, non-pathogen (Baysal and Yıldız, 2017) and is being horse for numerous extra-cellular heterologous proteins secreting micro-organism (Huang et al., 2018).
A large variety of industrial amylases have been derived from very wide and diversified Bacillus genus (Barros et al., 2013). A number of studies are performed with better α-amylase production either in wild or recombinant form in different B. subtilis strains (Yang et al., 2011). As it has been widely used for α-amylases production with different nutrition rich in-vitro cultures. The agriculture wastes are the available low cost microbial fuel, which could be utilized for the growth of microbial cultures as well as to gain their by-products including extra-cellular enzymes (Haq et al., 2018).
The fate of agriculture wastes may be on choice either allowed to decompose itself with the passage of time or fired-up. Meanwhile, there is not proper management or utilization of banana agro-wastes. Even its all parts especially fruit peel is rich with minerals, carbohydrates and proteins (Subedi and Walsh, 2011; Fahrasmane et al., 2014). It could be utilized as a raw material for biosynthesis of various bio based products. The B. subtilis is able to utilize banana wastes either in fresh or dry mass and could produce extra-cellular hydrolytic enzymes while may be helpful to reach the industrial need (Silpa et al., 2018).
There has not been much work done for the selection of a specific part of banana agro-wastes except fruit peel is useful for the production of α-amylase or other enzymes with B. subtilis.
The aim of the present study is to search out a proper organ of banana plant which plays a significant role in the production of enzymes specially α-amylases. The B. subtilis as being non-pathogenic industrial micro-organism could make it possible to produce low cost α-amylases, which is a prime need of the time to boom the economy of a country in food to garment industries.
MATERIAL AND METHOD
Preparation of fermentation substrate: The banana agriculture wastes were collected from open banana fields. Farmers excises the plants banana fields at the time of plant harvest as well as diseased plants. Different parts of banana plant were targeted such as leaf blade, leaf midrib, pseudostem, floral leaflets, fruit peel and sucker. The collected material was washed with tap-H2O to remove dirt, dried with filter-paper and crushed into fine pieces (looks like powder) with Molineux blender. Its exact 50 g was mixed with 50 ml dH2O and again crushed with blender for 5 min. The crushed mixture was squeezed first with muslin cloth and then filtered with filter-paper. The extracted fluid was stored at 4˚C for next use (Gomes et al., 2008).
Preparation of agriculture nutrient based fermentation medium: The tryptone yeast extract-based TY or Schaeffer [tryptone (10 g L-1), yeast extract (5 g L-1), NaCl (5 g L-1)] medium was used for batch mode fermentation (Sambrook et al., 1989). It was used as a control culture denoted as LB₀. Two levels of concentration (15% and 30%) of each extract of banana parts were maintained in LB₁ (⅛ LB₀) medium (Table 1). Total volume of each fermentation culture maintained 25 ml in 200 mL Erlenmeyer flasks. The cultures were fermented in an orbital shaker (250 rpm) at 37°C after its sterilization through autoclave at 121˚C for 15 min.
Table 1. Composition Of Bacillus Subtilis Lc4 Growth Cultures Supplemented With Agro Banana-Wastes As Fermentation Substrate.
| S. #s | Medium | Composition of medium |
| 1 | LB₀ | Tryptone 10 g L-1, NaCl 5 g L-1 and yeast extract 5 g L-1 |
| 2 | LB₁ | ⅛ LB₀ + dH2O |
| 3 | LB₂ | LB₁ + 15 % leaf blade extract + dH2O |
| 4 | LB₂ₐ | LB₁ + 30 % leaf blade extract + dH2O |
| 5 | LB₃ | LB₁ + 15 % leaf midrib + dH2O |
| 6 | LB₃ₐ | LB₁ + 30 % leaf midrib + dH2O |
| 7 | LB₄ | LB₁ + 15 % pseudostem + dH2O |
| 8 | LB₄ₐ | LB₁ + 30 % pseudostem + dH2O |
| 9 | LB₅ | LB₁ + 15 % floral leaflets + dH2O |
| 10 | LB₅ₐ | LB₁ + 30 % floral leaflets + dH2O |
| 11 | LB₆ | LB₁ + 15 % fruit peels + dH2O |
| 12 | LB₆ₐ | LB₁ + 30 % fruit peels + dH2O |
| 13 | LB₇ | LB₁ + 15 % sucker + dH2O |
| 14 | LB₇ₐ | LB₁ + 30 % sucker + dH2O |
| Note: LB₀: TY-medium; Volume of each culture was maintained 25 ml in 200 ml conical volumetric flask | ||
Pure Culture of B. subtilis and Preparation of Inoculum:For B. subtilis LC4 cell activation, almost 20 µl of glycerol stock was used to inoculate 5 ml TY-medium. The culture was incubated with 250 rpm shaking at 37°C for overnight in dark. Its 0.5 ml was sub-cultured in 50 ml LB medium and incubated at same conditions for 1 hour. The O.D of culture was checked it’s around OD600 0.5). From this master culture, other cultures were inoculated to raise an initial OD600 equivalent to 0.05. All these cultures were incubated at 37°C with constant 250 rpm shaking for 12 hours Bacillus fermentation period. After O.N., cultures were harvested after taking culture’s OD.
Extraction of Crude Enzyme: For crude enzyme extraction, over-night (O. N.) fermented medium was diluted with 50 ml dH2O. The mixture was agitated for 1 hr flask with 160 rpm shaking. The mixture was filtered with cheesecloth for removal of cells. It was also centrifuged at 5000 rpm for 10 minutes at 4°C. Supernatant was transferred in new tube and used as crude enzyme (Singh et al., 2009).
Determination of Organic Contents in B. subtilis Culture: Total proteins measured by mixing sample (0.5 ml) with 2.5 ml alkaline copper reagent at room temperature than 0.25 ml Follin-Ciocalteau reagent was added after 10 min and absorbance read at 750 nm (Lowry et al., 1951). Reducing sugars were also analyzed in same sample by mixing 1 ml supernatant and 1 ml DNS (3,5-dinitrosalicyclic acid). After heating the mixture in boiling H2O for 5 min, absorbance was read at 540 nm (Miller, 1959), while total sugars were determined according to (Hansen and Moller, 1975). For free proline determination, 0.5 ml sample was mixed with equal volume of glacial acetic acid in a test tube. The 0.5 ml acidic ninhydrin and 1 ml toluene were also added and mixture was incubated for 1 hour in a boiling water-bath. The reaction was cooled down at room temperature and absorbance was read against blank at 540 nm on spectrophotometer (Marin et al., 2010).
Performance of Hydrolytic Enzyme Assays: The α-amylase activity of B. subtilis enzyme crude was measured with DNS assay (Worthington, 1993). In brief, reaction mixture was prepared by mixing 2 ml 20 mM phosphate buffer (pH 7), 0.2 ml crude enzyme, 0.2 ml freshly prepared substrate (5 % starch). The mixture was incubated at 37°C for 15 minutes. The 2 ml of DNS was added to develop a complex of α-amylase product as well as to stop the enzyme reaction. The liberated amount of reducing sugars was estimated by reading OD at 540 nm with UV-VIS spectrophotometer. For xylanase activity, 1 ml of crude enzyme sample was mixed with 1 ml substrate [1 g xylose dissolved in 100 ml citrate buffer (pH 5.3)] in the test tubes. The mixture was incubated at 60°C for 15 minutes. Exactly, the equal volume of DNS was added to stop the reaction. Absorbance was noted at 540 nm against blank (Hansen and Moller, 1975). For pectinase assay, 167 μl crude enzyme was mixed with 1 ml buffer [0.1 M citric acid or phosphate Buffer (pH 5.0)] than add 167μl substrate (0.5% D-galacturonic acid).
The mixture was incubated at 37°C in water bath for 60 min. Exact 0.334μl of reagent A (40 g anhydrous Na2CO3 dissolved in 600 ml ddH2O than stirred after adding 16 g glycine until dissolved. The 0.450 gm CuSO4 5H2O was also added during stirring and volume raised to 1 liter) and B (1.2 g neocuprine-HCl dissolved 1-liter ddH2O and stored in brown bottle at 4°C) were added separately. Mixture was heated in boiling water bath again for 13 min. tubes were cool down at room temperature then 2ml H2O added and mixed by inversion. Absorbance was read at A450 against water blank and activity was determined from D-galacturonic acid standard curve (Holtzhauer, 2006).
Statistical Analysis: Cultures of this experiment were maintained with three replicates. Collected data of different parameters of experiment were subjected for its significance analysis and Duncan’s Multiple Range test at 5 %. It was computed with COSTAT (CoHort software, Berkeley, USA) software, a statistical package.
RESULTS AND DISCUSSION
The increasing human population acquires variant qualities in food to textile products. Both depend on annual production of agriculture crops to their processing ways either chemical and or enzymatic based. Specifically, enzymatic processing is considered as safe, while is being costly when enzymes are produced with the utilization of synthetic nutrient medium. Micro-organisms are efficient for the production of extracellular enzymes, while costs remain high if secreted in commercial synthetic medium. It is quite possible to reduce the production costs of extracellular hydrolytic enzymes with the utilization of cheap agro-wastes as raw materials for the cell growth of microorganisms (Yang et al., 2011). The agriculture wastes like as peels of potato, banana, orange, molasses and cane bagasse including their plants or aqueous extracts are useful carbon sources either used in minimal or synthetic growth medium. Such medium could be more workable, if they are used for Bacillus subtilis for the production of industrial important hydrolytic enzymes (Haq et al., 2018).
Table 2. Adjustment Of Various Biochemical In Bacillus Subtilis Lc4 Growth Cultures Supplemented With Agro Banana-Wastes As Fermentation Substrate.
| #s | Nutrient medium | Culture growth OD₆₀₀ | Soluble sugars
(mg ml⁻ᶦ) |
Reducing sugars
(mg ml⁻ᶦ) |
Total proteins
(mg ml⁻ᶦ) |
Proline contents
(mg ml⁻ᶦ) |
| 1. | LB₀ | 1.570±0.069ᵇᶜ | 12.66±0.276ᵃ | 6.821±0.236ᵇ | 15.63±0.045ᵃ | 2.177±0.045ᵃ |
| 2. | LB₁ | 0.653±0.017ʰ | 4.360±0.552ʰ | 2.644±0.195ᶠ | 12.06±0.023ʲ | 1.540±0.062ᵇᶜ |
| 3. | LB₂ | 1.753±0.068ᵇ | 7.328±0.138ᶠ | 3.924±0.171ᵈᵉ | 11.20±0.158ᵉ | 1.608±0.080ᵃᵇᶜ |
| 4. | LB₂ₐ | 2.167±0.120ᵃ | 9.114±0.227ᶜᵈ | 6.189±0.569ᵇ | 13.53±0.102ᵇᶜ | 0.843±0.057ᵈᵉ |
| 5. | LB₃ | 1.155±0.009ᵉᶠᶢ | 8.200±0.187ᶢ | 5.387±0.284ᵉᶠ | 11.16±0.023ᶢ | 1.441±0.119ᵇᶜᵈ |
| 6. | LB₃ₐ | 1.112±0.039ᶠᶢ | 8.045±0.081ᵉᶠ | 3.249±0.089ᶜ | 13.63±0.208ᵈ | 1.549±0.057ᵇᶜ |
| 7. | LB₄ | 1.339±0.062ᶜᵈᵉ | 7.215±0.284ᵈᵉᶠ | 6.217±0.162ᵈᵉ | 13.06±0.057ʰ | 1.471±0.034ᵇᶜᵈ |
| 8. | LB₄ₐ | 1.412±0.093ᶜᵈ | 9.508±0.162ᵇᶜ | 3.924±0.154ᵇ | 13.29±0.260ᶜ | 0.980±0.023ᶜᵈᵉ |
| 9. | LB₅ | 1.400±0.049ᶜᵈ | 6.793±0.154ᶢ | 5.331±0.073ᵈᵉ | 12.77±0.045ᶠ | 1.549±0.091ᵇᶜ |
| 10. | LB₅ₐ | 1.506±0.117ᶜ | 10.08±0.138ᵇ | 3.924±0.365ᶜ | 13.96±0.442ᵇ | 0.618±0.040ᵉ |
| 11. | LB₆ | 1.233±0.035ᵈᵉᶠ | 8.200±0.171ᵉᶠ | 4.402±0.008ᵈ | 12.77±0.113ᵈ | 1.461±0.063ᵇᶜᵈ |
| 12. | LB₆ₐ | 1.530±0.015ᶜ | 10.06±0.188ᵇ | 6.217±0.097ᵃ | 12.55±0.260ᵇᶜ | 0.892±0.096ᵈᵉ |
| 13. | LB₇ | 0.983±0.070ᶢ | 5.513±0.179ᶢ | 2.921±0.219ᵈ | 12.45±0.226ⁱ | 0.667±0.125ᵉ |
| 14. | LB₇ₐ | 1.482±0.110ᶜ | 6.751±0.114ᶜᵈᵉ | 3.260±0.049ᵇ | 13.75±0.340ᵇᶜ | 1.382±0.006ᵃᵇ |
| p – significance | 24.844*** | 40.132*** | 43.513*** | 225.87*** | 5.962*** | |
The B. subtilis is the subject for production of α-amylases including other hydrolytic enzymes and its efficiency of saccharification for banana agro-industrial wastes used as fermentation carbon source. For this experiment, a 12-hrs B. subtilis culture on seven organs of banana agro-waste was maintained in LB₁ (⅛LB₀) medium including LB₀ (control) TY-medium. After 12 hours of culture incubation, maximum cell growth (OD₆₀₀) was observed in LB₂ₐ (2.167±0.120) supplemented with leaf blade as a substrate and LB₀ (1.570±0.069) with standard TY-medium. The distinct B. subtilis growth phases observed among the organ specific cultures, which depends on the type or part of banana agro-waste materials supplemented as nutrient source (Sharma et al., 2017).
Lowest growth was noted in LB₃ₐ (1.112±0.039) and LB₄ₐ (1.412±0.093) cultures, when cells were growing on medium supplemented with leaf midrib and pseudostem respectively (Table 1 and 2). Variable amounts of total soluble sugars and reducing sugars observed among the cultures. This fluctuation among these parameters might be due to the structural variation in substrate type of the nutrient medium (Table 1). In this study, maximum total soluble sugars were noted as 10.06±0.188 mg ml-1 and reducing sugars 6.217±0.097 mg ml-1 in LB₆ₐ and LB₄ respectively (Table 2). The total proteins observed higher in LB₇ₐ and prolines in LB₃ₐ (p≤0.05), while remained lower than LB₀ (TY-medium) B. subtilis cultures.
The production of lower to higher concentrations of the soluble sugars or reducing sugars basically depends on the added value of nutrient substrates. Type of a substrate controls the growth rate of an organism with production of desired secondary products. It could be led to stimulate the production of supplemented substrate relevant enzymes by B. subtilis LC4, which results into good conversion of complex to small molecules simply like as reducing sugars (Bell et al., 2005; Shahrim et al., 2008). Meanwhile, prolonged fermentation time may not be a key for significant increase in reducing sugar concentration depends on the secretion of its associated enzymes. The B. subtilis growth is coupled with the production level of amylases in the culture medium. This suggests that growth of Bacillus is crucial for the secretion of extra-cellular proteins (Table 2).
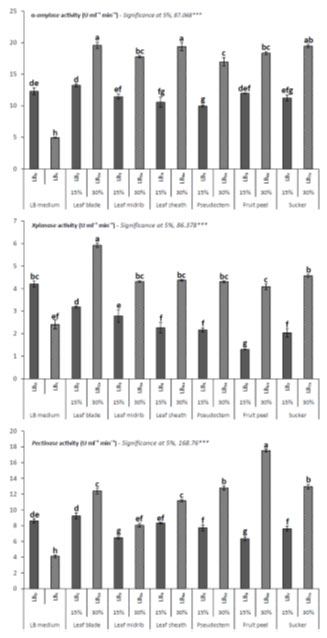
The concentrations and type of plant organ based raw materials are taken as major fermentation substrate for estimation of hydrolytic enzyme productions. The α-amylase yield was maximum at 30 % concentration of leaf blade extract (LB₂ₐ) and sucker (LB₇ₐ) with 12.91 U ml⁻¹ min⁻¹ and 12.91 U ml⁻¹ min⁻¹ activities (Fig 1). The α-amylase production was variable among the cultures which depend on type of substrate. In LB₅ and LB₅ₐ cultures, yield was lower as due to targeted floral leaflets used as substrate. Similar trend in the production of xylanase was observed among the cultures as shown by α-amylase (Fig 2). However, production of pectinase has shown variation in yields among the maintained cultures from both α-amylase and xylanase (Fig 3). It means that the required energy source of microbes for high extracellular protein secretion is retained in the type of substrate source and its available concentrations. Since it could be belonging to category of agro-lignocellulosic raw materials with capability of providing many essential nutrients for growth of microbes for useful productions.
In general, banana waste is comprised on 50-60% cellulose, 25-30% hemicelluloses, 12-18% lignin, 3-5% pectin, 3-5% fat and wax, 2-3% water soluble sugars and proteins (Mukhopadhyay et al., 2008). The rate of saccharification and enzymes production depends on the available type of substrate in the culture medium. Apparently, enzyme production could be repressed due to surplus saccharification and availability of catabolite. One enzyme could be reached to maximum production limit while others entered to decline phase. It results into lowering the levels of total soluble sugars and reducing sugars. In this regard, catabolite base repression of amylases (Vinuselvi et al., 2012), xylanases (Coleri et al., 2009; Shulami et al., 2014) and pectinase (Kumar et al., 2009; Sharma et al., 2012) have been reported in various fermentation organisms including bacteria and fungi.
The rate of enzyme production is limited with growth phases like as activities of xylanase and pectinase slightly decline after log phase, while amylases increases. It might be possible that these enzymes are degraded by proteases, which are released from autolyzing cells during or after stationary growth phase (Berg and Pettersson, 1977). At early fermentation culture hours (log phase), increasing phase of reducing sugars is observed, which is due to the hydrolysis of xylan and pectin by xylanases and pectinases (Gaewchingduang and Pengthemkeerati, 2010). At the stationary phase, B. subtilis secreted numerous heterologous proteins in the culture medium, while dying cells and deficiency of nutrition in medium triggers the production of proteases in the medium. Due to this reason, fluctuations in the amount of total proteins in cell cultures are observable.
Figure 1: Production Of Different Hydrolytic Industrial Enzymes In Bacillus Subtilis Lc4 Growth Cultures Supplemented With Agro Banana-Wastes As Fermentation Substrate.
CONCLUSION
Production costs of hydrolytic enzymes could be reduced with the specific selection of free energy rich organs from agriculture waste materials. Banana plant is also one with 100% wasted vegetative materials including fruit peels. Its leaf blade, pseudostem and fruit peels are energy rich source and could be useful to establish an optimum growth of the fermentation microorganism. Like as B. subtilis is able to grow with saccharification of complex to simple soluble sugars and reducing sugars as well as produces various enzymes including amylase, xylanase and pectinase. Optimization of cell culture growth depends on the mode of substrate saccharification, which in final results into efficient enzyme production. The banana can provide enough nutrients for microorganism growth without additional supplement of carbon or nitrogen sources. The above system could be useful in future to reduce the overall production costs of industrial enzymes including other secondary metabolites.
ACKNOWLEDGEMENTS
This research work was supported financially for undergraduate thesis research by University of Sindh, Jamshoro, Pakistan.
Conflict of Interest: No conflict of interest.
REFERENCES
Agüloğlu, F. S., Enez, B., Özdemir, S., and Matpan, B. F. (2014). Purification and characterization of thermostable α-amylase from thermophilic Anoxybacillus flavithermus. Carbohydrate Polymers, 102, pp 144-150.
Ahmad, T., and Danish, M. (2018). Prospects of banana waste utilization in wastewater treatment: A review. Journal of Environmental Management, 206, pp 330-348.
Barredo, J. L. (2005). Microbial enzymes and biotransformation. In: Methods in Biotechnology, Humana Press; Totowa, NJ, USA, 17, pp 1-27.
Barros, F. F. C., Simiqueli, A. P. R., de Andrade, C. J., and Pastore, G. M. (2013). Production of enzymes from agroindustrial wastes by biosurfactant-producing strains of Bacillus subtilis. Biotechnology Research International, 2013, pp 103960.
Baysal, Ö., and Yıldız, A. (2017). Bacillus subtilis: An Industrially Important Microbe for Enzymes Production. EC Microbiology, 4(2017), pp 148-156.
Bell, T., Newman, J. A., Silverman, B. W., Turner, S. L., and Lilley, A. K. (2005). The contribution of species richness and composition to bacterial services. Nature, 440, pp 165-173.
Berg, B., and Pettersson, G. (1077). Location and Formation of Cellulases in Trichoderma viride. Journal of Applied Bacteriology, 42(1), pp 65-75.
Coleri, A., Cokmus, C., Ozcan, B., Akkoc, N., and Akcelik, M. (2009). Isolation of alpha-glucosidase producing thermophilic bacilli from hot springs of Turkey. Mikrobiologiia, 78(1), pp 68-78.
Demirkan, E. (2011). Production, purification, and characterization of α-amylase by Bacillus subtilis and its mutant derivates. Turkish Journal of Biology, 35, pp 705-712.
Demirkan, E. S., Mikami, B., Adachi, M., Higasa, T., and Utsumi, S. (2005). α-Amylase from B. amyloliquefaciens: Purification, characterization, raw starch degradation and expression in E. coli. Journal of Process Biochemistry, 40, pp 2629-2636.
Dumorné, K., Córdova, D. C., Astorga-Eló, M., and Renganathan, P. (2017). Extremozymes: A potential source for industrial applications. Journal of Microbiology and Biotechnology, 27(4), pp 649-659.
Essien, J. P., Akpan, E. J., and Essien E. P. (2005). Studies on mould growth and biomass production using waste banana peel. Bioresources Technology, 96(13), pp 1451-1456.
Fahrasmane, L., Parfait, B., and Aurore, G. (2014). Banana: A source of compounds with health properties. In: Acta Horticulturae, 1040, pp 75-82.
Frison, E., and Sharrock, S. (1998). The economic, social and nutritional importance of banana in the world. In: Bananas and Food Security International Symposium, Douala, Cameroon, pp 10-14.
Gaewchingduang, S., and Pengthemkeerati, P. (2010). Enhancing efficiency for reducing sugar from cassava bagasse by pretreatment. World Academy of Science, Engineering and Technology, 4(10), pp 477-480.
Gomes, I., Shaheen, M., Rahman, S. R., and Gomes, D. J. (2008). Comparative Studies on Production of Cell Wall-Degrading Hydrolases by Trichoderma reesei and T. viride in Submerged and Solid-State Cultivations. Bangladesh Journal of Microbiology, 14(1), pp 1-16.
Hansen, J., and Moller, I. B. (1957). Analysis of starch and soluble sugars with Anthrone reagent. Anals of Biochemistry, 68, pp 87-94.
Haq, I.-U., Maher, A., Gill, N. P., Fatima, U., Qadir, G., and Ali, K. (2018). Growth of Bacillus subtilis and production of acetic acid with rotten potato: Used as substrate. International Journal of Pharmaceutical Sciences and Research, 9(10), pp 4229-4235.
Holtzhauer, M. (2006). Basic Methods for the Biochemical Lab. Publisher Springer-Verlag Berlin and Heidelberg GmbH & Co.
Huang, G. L., Gosschalk, J. E., Kim, Y. S., Loo, O. R. R., and Clubb, R. T. (2018). Stabilizing displayed proteins on vegetative Bacillus subtilis cells. Applied Microbiology and Biotechnology, 102(15), pp 6547-6565.
Iizuka, M., Uenakai, K., Svendsby, O., and Yamamoto, T. (1985). Alcohol fermentation of green banana. Journal of Fermentation Technology, 63(5), pp 475-477.
Itelima, J., Onwuliri, F., Onwuliri, E., Onyimba, I., and Oforji, S. (2013). Bio-ethanol production from banana, plantain and pineapple peels by simultaneous saccharification and fermentation process. International Journal of Environmental Sciences and Development, 4(2), pp 213-216.
Kar, S., Ray, R.C., and Mohapatra, U. B. (2008). α-Amylase production by Streptomyces erumpens MTCC 7317 in solid state fermentation using response surface methodology (RSM). Polish Journal of Microbiology, 57, pp 289-296.
Kumar, S., Sharma, H. K., and Sarkar, B. C. (2011). Effect of substrate and fermentation conditions on pectinase and cellulase production by Aspergillus niger NCIM 548 in submerged (SmF) and solid state fermentation (SSF). Food Science and Biotechnology, 20, pp 1289.
Kwak, Y. S., Akiba, T., and Kudo, T. (1998). Purification and characterization of α-amylase from hyperthermophilic archaeon Thermococcus profundus, which hydrolyzes both α-1,4 and α-1,6 glucosidic linkages. Journal of Fermentation and Bioengineering, 86, pp 363-367.
Li, C., Liu, G., Nges, I. A., Deng, L., Nistor, M., and Liu, J. (2016). Fresh banana pseudo-stems as a tropical lignocellulosic feedstock for methane production. Energy Sustain in Society, 6, pp 27.
López‐Baca, A., and Gómez, J. (1992). Fermentation patterns of whole banana waste liquor with four inocula. Journal of Science Food and Agriculture, 60(1), pp 85-89.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry, 193(1), pp 265-275.
Marin, J. A., Andreu, P., Carrasco, A., and Arbeloa, A. (2010). Determination of proline concentration , an abiotic stress marker in root exudates of excised root cultures of fruit tree rootstocks under salt stress. In: Actes Du 3ème Meeting International ‘ridoculture et Cultures Oasisennes.
Miller, G. L. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anals Chemistry, 31(3), pp 426-428.
Mohiuddin, A., Saha, M., Hossian, M., and Ferdoushi, A. (2014). Usefulness of banana (Musa paradisiaca) wastes in manufacturing of bio-products: a review. The Agriculturists, 12(1), pp 148-158.
Mukhopadhyay, S., Ph, D., Fangueiro, R., Arpaç, Y., and Ulkü, Ş. (2008). Banana Fibers – Variability and Fracture Behaviour. Journal of Engineered Fibers and Fabrics, 3, pp 39-45.
Oboh, G. (2005). Isolation and characterization of amylase from fermented cassava (Manihot esculenta Crantz) waste- water. African Journal of Biotechnology, 4, pp 1117-1123.
Patel, H., Patel, A., Surati, T., and Shah, G. (2012). Potential use of banana peels for the production of fermented products. International Journal of Environment and Sustainable Development, 9(1), pp 27-43.
Rana, N., Walia, A. and Gaur, A. (2013). α-amylases from microbial sources and its potential applications in various industries. The National Academy Science Letters, 36, pp 9-17.
Sahnoun, M., Bejar, S., Sayari, A., Triki, M. A., Kriaa, M., and Kammoun, R. (2012). Production, purification and characterization of two α-amylase isoforms from a newly isolated Aspergillus oryzae strain S2. Journal of Process Biochemistry, 47, pp 18-25.
Sambrook, J., Fritsch, E. F., and Maniatis, T. (1989). Molecular Cloning: A Laboratory Manual. In: Cold Spring Harbor Laboratory Press, pp 931-957.
Shahrim, Z., Sabaratnam, V., Rahman, N. A. A., Abd-Aziz, S., Hassan, M. A., and Karim, M. I. (2008). Production of reducing sugars by Trichoderma sp. KUPM0001 during solid substrate fermentation of sago starch processing waste hampas. Research Journal of Microbiology, 3, pp 569-579
Sharma, D. C., and Satyanarayana, T. (2012). Biotechnological potential of agro residues for economical production of thermoalkali-stable pectinase by Bacillus pumilus dcsr1 by solid-state fermentation and its efficacy in the treatment of ramie fibres. Enzyme Research, 2012, pp 281384.
Sharma, K. M., Kumar, R., Panwar, S., and Kumar, A. (2017). Microbial alkaline proteases: Optimization of production parameters and their properties. Journal of Genetic Engineering and Biotechnology, 15, pp 115-126.
Shulami, S., Shenker, O., Langut, Y., Lavid, N., Gat, O., Zaide, G., Zehavi, A., Sonenshein, A. L., and Shoham, Y. (2014). Multiple regulatory mechanisms control the expression of the geobacillus stearothermophilus gene for extracellular xylanase. Journal of Biological Chemistry, 289(37), pp 25957-25975.
Silpa, D., Brahmaji, P., Rao, G. K. K., and Ram, M. R. (2018). Screening of Amylase Producing Bacillus spp. Isolated from Banana Rhizosphere. International journal of pharmaceutical sciences and research, 22(2), pp 133-142.
Singh, S., Tyagi, C. H., Dutt, D., and Upadhyaya, J. S. (2009). Production of high level of cellulase-poor xylanases by wild strains of white-rot fungus Coprinellus disseminatus in solid-state fermentation. New Biotechnology Journal, 26(3-4), pp 165-170.
Slavić, S. M., Pešić, M., Vujčić, Z., and Božić, N. (2016). Overcoming hydrolysis of raw corn starch under industrial conditions with Bacillus licheniformis ATCC 9945a α-amylase. Applied Microbiology and Biotechnology, 100(6), pp 2709-2719.
Subedi, P. P., and Walsh, K. B. (2011). Assessment of sugar and starch in intact banana and mango fruit by SWNIR spectroscopy. Post-harvest Biology and Technology, 62, pp 238-245.
Tewari, H. K., Marwaha, S. S., and Rupal, K. (1986). Ethanol from banana peels. Agricultural Wastes, 16(2), pp 135-146.
Vengadaramana, A. (2013). Industrial Important Microbial alpha-Amylase on Starch-Converting Process. Scholars Academic Journal of Pharmacy, 2(3), pp 209-221.
Vinuselvi, P., Kim, M. K., Lee, S. K., and Ghim, C. M. (2012). Rewiring carbon catabolite repression for microbial cell factory. BMB Reports, 45(2), pp 59-70.
Walker, G. M., and Dundee, A. (2009). Amylases. In: Encyclopedia of Microbiology, 3rd Edition, pp 159-173.
Worthington, V. (1993). Alpha-Amylase, in V. Worthington (Ed.), Worthington Enzyme Manual Freehold, Worthington Biochemical Corp, NJ,. In: Worthington Enzyme Manual Freehold, Worthington Biochemical Corp, NJ.
Yang, H., Liu, L., Li, J., Du, G., and Chen, J. (2011). Heterologous expression, biochemical characterization, and overproduction of alkaline α-amylase from Bacillus alcalophilus in Bacillus subtilis. Microbial Cell Factories, 10, pp 77-85.