1,2,3,4Post Graduate and Research Department of Microbiology, PSG College of Arts and Science, Coimbatore Tamilnadu India
Corresponding author Email: geetha.cybrids@gmail.com
Article Publishing History
Received: 10/07/2019
Accepted After Revision: 15/09/2019
Wastewater from the textile industries contains a variety of pollutants, particularly dyes. Azo dyes are the largest group of synthetic aromatic dye used in the textile industry for dyeing purpose and are highly water soluble in nature. The removal of azo dyes from industry effluents is desirable not only for aesthetic reasons but also because azo dyes and their breakdown products are toxic to aquatic life and mutagenic to humans. The treatment of these dyes have been carried out by many methods such as physical, chemical and biological as individual and in combination to reduce the effect of pollution. The biodegradation of azo dyes is difficult due to the complex structure, synthetic nature and some are highly resistant to microbial attack. To overcome this problem, this study aims in developing a consortium to degrade complex azo dyes present in the effluent. The current work aims in screening and identification of potent azo dye degrading bacteria, studying azo dye degradation efficiency of the isolates, preparing the microbial consortia and to determine the efficiency in decolourization and degradation in both chosen dye (Amido black 10 B) and effluent. The degradation efficiency of the effluent using mixed dyes were significantly higher than the individual strain which was evident from reduction in colour and COD which was 76 % and 79 % respectively. The degradation of the effluent using consortium was studied using FT-IR which revealed change in the peak characteristics.
Amido black, Azo dyes, consortium, decolourization,FT-IR.
Geethadevi C, Pavithra C. K, Dhivya S, Rajendran R. Biodegradation of Textile Effluent Containing Azo Dye Using Individual and Mixed Adapted Bacterial Strains. Biosc.Biotech.Res.Comm. 2018;11(4).
Geethadevi C, Pavithra C. K, Dhivya S, Rajendran R. Biodegradation of Textile Effluent Containing Azo Dye Using Individual and Mixed Adapted Bacterial Strains. Biosc.Biotech.Res.Comm. 2018;11(4). Available from: https://bit.ly/2lYzID7
Copyright © Geethadevi et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
Introduction
Textile industry is considered as one of the chief export earning and employment providing sector of the nation. Though it helps developing the economy of the country it also burdens it by polluting the environment through the processes involved in its production especially the dyes and chemicals used. This industry also consumes a generous amount of water during its manufacturing processes especially in the dyeing and finishing operations of the plants. The textile wastewater is considered the most polluter among all the industrial sectors based on the volume generated and the composition of the effluent (Mansour et al 2012). Since, there is an increase demand for textile products the production also increases relatively and this has led to the use of synthetic dyes which in turn has led to be a source of severe pollution (Ogugbue and Sawidis 2011 Yin et al., 2019).
A wide range of dyes are being used in the textile industry among which synthetic dyes play an important role due to their cost effectiveness and dyeability. Synthetic dyes are polyaromatic molecules that give a permanent coloring to materials like textile fabrics. A variety of synthetic dyestuffs released by the textile industry pose a threat to environmental safety. The degree of coloring process continues even after the dyeing process, leading to effluents containing azo dye (Lu & Liu 2010). The chemical structure of the dyes makes it resistant to most types of physical, chemical and biological treatments (Mansour et al. 2011b). The removal of azo dyes from industry effluents is desirable not only for aesthetic reasons but also because azo dyes and their breakdown products are toxic to aquatic life and mutagenic to humans. Therefore, treatment of textile effluent is necessary before discharging in the environment. The treatment of textile effluents is also essential to protect the ecosystem and allow the subsequent recycling of the treated effluent for irrigation or reuse within the procedures of the textile plant (Yaseen and Scholz, 2019).
The treatment of these dyes have been carried out by many methods such as physical, chemical and biological as individual and in combination to reduce the effect of pollution. Physical or chemical methods for textile wastewater pretreatment are of high cost, extremely energy consuming, and environmentally low efficient and generate toxic sludge (Sakar et al ., 2017).These dyes are generally recalcitrant to biodegradation due to their xenobiotic nature. However microorganisms, being highly versatile, have developed enzyme systems for the decolorization and mineralization of azo dyes under certain environmental conditions. The complete mineralizations of organic pollutants by biological methods are cost effective and eco-friendly (Nachiyar et al., 2016)
The biodegradation of azo dyes is difficult due to the complex structure, synthetic nature and some are highly resistant to microbial attack. To overcome this problem, several studies using microbial consortia were attempted to achieve not only dye decolourization, but also degradation of the aromatic amines due to the efficiency of the microorganisms (Chan et al., 2011). Thus, this work aims in screening, identification of potent azo dye degrading bacteria, studying azo dye degradation efficiency of the isolates, preparing the microbial consortia and to determine the efficiency in decolourization and degradation in both chosen dye and effluent.
Methods and Materials
Sample Collection
The textile effluent was collected from Chinnakkari Common Effluent Treatment Plant (CETP), Tiruppur and was used for the study.
Physiochemical analysis
The physiochemical parameters such as color, pH, BOD (Biological Oxygen Demand), COD (Chemical Oxygen Demand), TSS (Total Suspended Solids), TDS (Total Dissolved Solids) were determined immediately after being brought to the laboratory as per the standard APHA methods (Rice, 2012)
Isolation of autochthonous bacterial strains from effluent sample
The spread plate assay method was used for the enumeration of aerobic bacteria from the collected sample. This was done by serial dilution of the sample (10-1 to 10-8), and placing 0.1ml of the diluted samples (10-6, 10-7) in an minimal media (64 g Na2HPO4.7H2O, 15 g KH2PO4, 2.5 g NaCl and 5.0 g NH4Cl, 1M MgSO4 and 1M CaCl2) agar plate containing dyes. The dyes used for the study were Alizarine red, Amido black 10 B, Reactive red and Brilliant violet (SIGMA, India). The diluted samples were spread on the surface of agar plate using L- rod and was incubated at 37˚C as for 48 hours. The cultures which showed a zone of clearance around their colonies were isolated and used for further screening (HeFang et al., 2004).
Screening of microorganisms capable of decolorizing and degrading the azo dye
The minimal broth containing four different dyes (100 mg/L) were prepared; the isolated organisms were inoculated and incubated at 37°C for 7 days. The degradation patterns were observed for each organism in different dyes using visible spectrophotometer in the wavelength ranges between 400 – 650 nm (Omar, 2008).The strains showing decolourization efficiency in chosen all four dyes, where studied for their potency in Amido Black 10 B dye and their percentage decolourization was recorded using the formulae below at 610 nm:
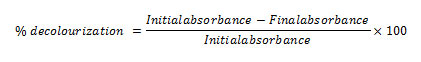
Identification of Selected Microorganisms
The screened bacterial strains were identified by using standard biochemical (Cappucino and Sherman, 2014) and microscopic techniques according to Bergey’s Manual of Systemic Bacteriology.
Strain Identification – Molecular characterization
The three most active isolates were identified by 16S rRNA sequencing after extracting the genomic DNA. The 16S region of ribosomal rRNA gene was and amplified using the universal primers 27F and1492R primer. The PCR amplification was done by initial denaturation at 94°C for 3 min followed by 30 cycles of 94°C for 30seconds, 60°C for 30 sec, 72°C for 60 sec and final extension at 72°C for 10 min. PCR purification was done by SolGent PCR Clean up kit (Millipore).The PCR product was sequenced using the 27F/1492R primers. Using ABI PRISM® BigDyeTM Terminator Cycle Sequencing Kits with AmpliTaq® DNA polymerase (FS enzyme) (Applied Biosystems), sequencing were performed. The sequences were assembled and edited using Bioedit software and deposited in the NCBI database for accession numbers. The sequences obtained were compared to find sequence similarity using GenBank program MUSCLE 3.7. PhyML 3.0 aLRT was used for phylogeny analysis. The program Tree Dyn 198.3 was used for tree rendering. The sequences obtained in this study were deposited in the GenBank database.
Test for synergism
In order to develop a bacterial consortium the selected strains must be tested for their compatibility. The test was performed as follows: three nutrient agar plates were taken and each plate was bored with two wells. The first plate was smeared with one of the three selected culture (TMB 2) and added with 10μl of the supernatant from TMB 6 and TMB 7 in the two wells respectively. The plates were then kept for incubation at 37˚C for 24 hours. Absence of any zone of inhibition around the wells showed that the cultures are compatible. The test was repeated by changing the swabbed organisms with the other two selected bacterial isolates and changing the supernatants from the organisms added in the bored wells accordingly (Ammar et al., 1998).
Development of Consortia for treatment textile effluent: Based
On the mutuality of the isolates, the Cayley’s tables were made. According to the Table-1, the culture consortium were developed and used for further study.
Table 1: Cayley’s table for consortia preparation
| Organisms | TMB 2 | TMB 6 | TMB 7 |
| TMB 2 | TMB 22 | TMB 26 | TMB 27 |
| TMB 6 | TMB 62 | TMB 66 | TMB 67 |
| TMB 7 | TMB 72 | TMB 76 | TMB 77 |
Treatment of effluent by individual strains and developed consortia
About 5ml of mixed culture was inoculated into 95 ml of textile effluent sample and incubated at 37ºC in shaking condition for 5 days. The degradation patterns of the effluent were measured in spectrophotometer in the range of 423nm.

Fourier Transformed Infrared Spectroscopy analysis
IR-spectra were obtained using a SHIMADZU FTIR 8400S. The untreated textile effluent (raw) and treated effluent with the potent consortia were assessed by FT-IR for the chemical modification. Effluent samples was recorded from 4000 – 400 as scanning range between wave number (cm‐1) and % Transmittance. All samples were run in triplicate and the data presented are the average of the three measurements.
Results and Discussion
The textile effluent sample was collected in sterilized container from CETP, Chinnakarai, Tiruppur and stored at 4°C for further studies.The autochthonous bacterial strains were isolated in minimal media plate containing the azo dye using spread plate technique. The result showed around 7 different colonies had the ability to degrade the azo dye which was observed through the zone around the colonies. The obtained colonies were designated as TMB1, TMB 2, TMB 6, TMB7, TMB 8, TMB 10 and TMB 11. It was clear from the results that the natural adaption of microorganism enabled them to survive in the presence such recalcitrant compounds (azo dyes). The ability of the organisms to decolourize was due to the utilization of the toxic dye which resulted in the breakdown of chromophore of the dyes. The isolated strains were then subjected to quantitative screening by inoculating the isolated strains in minimal media broth containing azo dyes. The results revealed that among the 7 strains screened, three strains were found to have ability to degrade all the four azo dyes used in the study. These strains were chosen because they showed percentage reduction more than 45% in 48 hours of incubation in all the four dyes used for the study (Fig. 1). Therefore, the strains TMB2, TMB 6 and TMB 7 were chosen for further study and were identified.
The isolated bacterial strains from effluent sample collected from the CETP were identified through morphologically different bacterial isolates cultures were preserved on nutrient agar medium at 4˚C. The three organisms which showed significant decolourization of the dye degradation were identified through morphological and biochemical analysis. The isolated bacterial cultures – TMB2, TMB 6 and TMB 7 were tentatively identified to be Pseudomonas sp., Klebsiella sp., Shigella sp., through biochemical test.
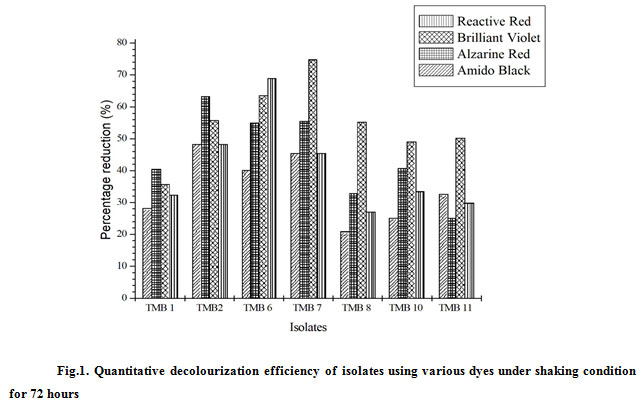 |
Figure 1: Quantitative decolourization efficiency of isolates using various dyes under shaking condition for 72 hours |
This was reconfirmed with 16s rRNA sequencing after extracting DNA.The sequences obtained were compared using GenBank program-Basic Local Alignment Search Tool (BLAST). The phylogenetic trees based on 16S rRNA gene sequences were constructed by the neighbour-joining method. The sequences obtained in this study were deposited in the GenBank database. GenBank accession numbers for the nucleotide sequences are KY788338,KY788341 and KY789442. The isolates obtained were found to be Pseudomonas aeruginosa, Klebsiella pneumonia, Shigella dysenteriae. From the results it is clear that the obtained strain are pathogenic to human, but still have the potency to degrade the chromophore of the toxic dyes. Several studies have reported that these pathogenic strains have efficiency in degrading the azo compounds. The decolourization Direct Blue 6 by Pseudomonas desmolyticum in 72 h under microaerophilic conditions was reported by Kalme et al. (2007). Similarly the decolorization and biodegradation of four different azo dyes, Reactive Yellow 107 (RY107), Reactive Black 5 (RB5), Reactive Red 198 (RR198), and Direct Blue 71 (DB71), in a sequential microaerophilic– aerobic treatment in 72, 120, 96, and 168 h, respectively, by a facultative Klebsiella sp was accounted by Franciscon et al. (2009).
The recent findings of Dixit and Garg (2018) clearly state the pathogenic strain posses’ potency to degrade the azo dye and completely mineralize them. In their study which is a two-stage sequential process that utilizes the isolated Klebsiella pneumoniae and successfully degrade the azo dyes and their metabolites completely in much less time as compared with the reported strains (Dixit and Garg, 2018). Decolourization of Acid Red 131 by using Shigella sp was reported by Sivaranjani et al (2013) in which they stated that 99% degradation was observed by the isolated strain (Sivaranjani et al., 2013). Therefore, further study of treating the effluent using the strains were carried out as to determine the efficiency of degradation and mineralization of azo dye containing effluents.
The untreated textile effluent was characterized and its physico – chemical parameters was determined and tabulated below (Table2).
Table 2: Physico – Chemical Characterization of Untreated Textile Effluent
| Parameters | Value | TNPCB standards |
| pH | 8.9 | 5.5-9.0 |
| Temperature | 35°C | 40°C at the point of discharge |
| Conductivity (µS/cm) | 13330 | 2250 |
| Turbidity (NTU) | 45.3 | 10 |
| Color (Hazen units) | 1200 | 400 |
| COD (mg/L) | 1160 | 250 |
| BOD3 (mg/L) | 70 | 30 |
| Hardness | 960 | 300 |
| Total suspended solids (TSS) (mg/L) | 3700 | 100 |
| Total dissolved solids (TDS) (mg/L) | 5970 | 2100 |
| Chloride (mg/L) | 2080 | 600 |
| Phosphate (mg/L) | 1.4 | 5 |
| Sulphate (mg/L) | 1745 | 1000 |
The collected sample had an unpleasant fishy odor and was turbid with greenish color. Unpleasant smell and taste in water, perhaps due to declining vegetation, inorganic constituents / organic substances, wastewater discharge into water bodies (Mohabansi et al., 2011). As reported by Patel et al., (2015) the primary sample has fishy, secondary aerated and processed has rotten egg as well as stagnant sample has fortified odor (Patel et al., 2015).The strong coloration of the effluent was sometimes influenced by the pH and temperature of the dyeing process which strengths the chromophore group. The presence of highly colored components affects the dissolved oxygen of the water. pH of a effluent is very important in determination of water quality since, it affects other chemical reactions such as solubility and metal toxicity (Fakayode, 2005). The dissolved oxygen levels are found to be very low and hence a lot of oxygen has been used up, which shows the increased concentration of organic matter. The collected effluent sample had a BOD3 70 mg/l which implies the content of organic matter is too high and oxygen gets depleted rapidly.COD is a critical parameter in assessing the water quality as it indicates the presence of non-biodegradable and organic matter present in the effluent. In the present study the investigations made indicate high COD value of around 1160 mg/l which is higher than the permissible level. As the physico-chemical parameters of the collected effluent are higher than the allowable limits, proper treatment is necessary before discharging. The elevated amount of industrial effluent pollution creates environmental issues that impact plant, animal and human life (Kolhe et al., 2011).
Analysis of compatibility was performed to verify whether the chosen strains used in biodegradation were suitable for effective bioremediation when used as a consortium. The results showed that there was no inhibition zone around the wells for any of the plates after incubation. The main reason for this compatible nature of the selected bacterial strains may be that they have co-existed for a longer period of time in a common environment. This clearly illustrates that the compatible nature of these strains would be effective in degrading the recalcitrant azo dyes and the complex organics present in the effluent. About 4 different consortia containing equal amount of the strains were prepared and designated as TMB 26, TMB 27, TMB 67 and TMB 267 and used for the study. On observing the reduction in the various physico – chemical parameters of the treated effluent sample using bacterial consortia, it could be concluded that the bioremediation efficiency of the consortium TMB 27 was found to be reduced to a significant extent (Table 3) compared to the other strains or developed consortia. This could be due to the fact that the consortium of adapted microorganism were able to exert more enzymes that have a wide spectral range which in turn could have degraded the complex organic compounds and dye content present in the effluent. Similarly investigation done by Puvaneshwari et al., (2006) revealed that mixed bacterial strains showed an efficient degradation of the organic compounds.
Table 3: Percentage Reduction of Physicochemical Parameters of Effluent Treated using Individual Strains and Developed Bacterial Consortia
| Treatments | Percentage of reduction in physico-chemical parameters (%) after 72 hours of treatment | |||
| COD | TDS | Colour | Turbidity | |
| Untreated | – | – | – | – |
| TMB 2 | 71.1 | 48.07 | 65.7 | 39.2 |
| TMB 6 | 64.9 | 29.6 | 50.5 | 49.2 |
| TMB 7 | 62.8 | 27.1 | 52.6 | 38.1 |
| TMB 26 | 62.8 | 35.5 | 57.8 | 53.6 |
| TMB 27 | 79.3 | 59.7 | 73.6 | 66.8 |
| TMB 67 | 73.1 | 53 | 68.4 | 67.9 |
| TMB 267 | 75.2 | 56.4 | 71.05 | 56.9 |
FTIR gives information regarding the structural changes that occurs during biodegradation process with the help of functional groups present (Ladwani et al., 2016). IR Spectra of the untreated effluent showed many bands of 3394.72 cm-1, 3174.83 cm-1, 2607.76 cm-1,2110.12 cm-1, and 1269.16 cm-1 which are representatives of functional group OH, N-H, O-H with strong and very broad intensity, and C-N. The peak position of 3498.12 cm-1, 2858.51 cm-1, 2607.76 cm-1, 2144.84 cm-1,1597.059 cm-1, 1435.04 cm-1, 1095.57 cm-1 and 898.82 cm-1which are the characteristics of O-H, C-H, O-H of strong and very broad intensity, , C=C, ether C-O and C-Cl alkyl halide group respectively.
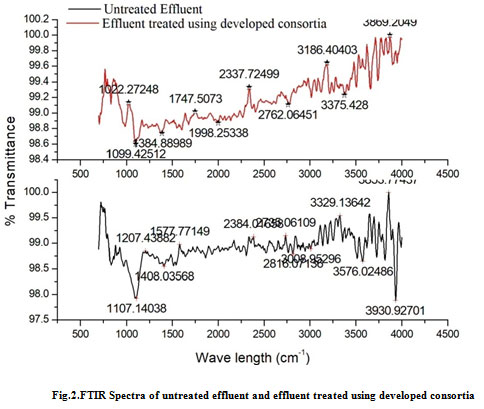 |
Figure 2: FTIR Spectra of untreated effluent and effluent treated using developed consortia |
IR spectrum of the textile effluent treated with consortia shows difference in bandings when compared with the untreated sample. During the biodegradation of effluent, the IR spectrum of the treated sample shows variations such as appearance and disappearance of peaks 2754.34, 3429.43, 2144.34, 1693.50, 1435.03, 1269.16,898.82 in treated sample. The absorption bands showed variation in 3930.27, 3853.77, 3722.61,3649.31, 3251.98, 3194.12, 3140.11, 1577.77,1107.14 cm-1, due to N-H stretching, O-H stretching (alcohol), N-H stretching (Amide), N-H (bending), C-O stretching (Alcohol)respectively (Figure 2). From the above results it is clear that changes in the FTIR spectrum are evidence for the degradation of the dyes and complex organic matter present in the effluent into simpler molecules which is due to the metabolism of the mixed bacterial strains. Therefore, the study concludes that when mixed bacterial population when used in degradation of effluents containing recalcitrant compounds, the rate of degradation would be faster as more number of various enzymes produced by the strains would act on the molecules and aid in degradation. Similar studies on degradation of recalcitrant’s by mixed microbial strains rather than individual have shown better degradation efficacy (Lade et al., 2015; Nachiyar et al., 2016; Chan et al., 2011) The strains isolated in the study are said to be pathogenic in nature which might pollute the environment further, in order to overcome this biotechnological tools can be used and the gene responsible for degradation can be cloned into non-pathogenic strains.
Acknowledgment
The authors gratefully acknowledge DST-SSTP (Ref. No – C/6318IFD/2016-17) for funding the research project.
References
American Public Health Association, American Water Works Association, Water Pollution Control Federation and Water Environment Federation, (1920). Standard methods for the examination of water and wastewater. American Public Health Association.
Ammar M.S., El-Esawey, M., Yassin, M. and Sherif, Y.M.(1998). Hydrolytic Enzymes of fungi isolated from certain Egyptian Antiquities objects while utilizing the Industrial Wastes of Sugar and Integrated Industries Company (SIIC). J. Biotechnol, 3(1998), pp.60-90.
Chan, G.F., Rashid, N.A., Koay, L.L., Chang, S.Y. and Tan, W.L., (2011). Identification and optimization of novel NAR-1 bacterial consortium for the biodegradation of Orange II. Insight Biotechnol, 1(1), pp.7-16.
Dixit, S. and Garg, S.(2018). Biodegradation of environmentally hazardous azo dyes and aromatic amines using Klebsiella pneumoniae. Journal of Environmental Engineering, 144(6), p.04018035
Fakayode, S.O. (2005). Impact of industrial effluents on water quality of the receiving Alaro River in Ibadan, Nigeria
Franciscon, E., Zille, A., Fantinatti-Garboggini, F., Silva, I.S., Cavaco-Paulo, A. and Durrant, L.R.(2009). Microaerophilic–aerobic sequential decolourization/biodegradation of textile azo dyes by a facultative Klebsiella sp. strain VN-31. Process biochemistry, 44(4), pp.446-452.
G Cappuccino, J. and Sherman, N. (2014). Microbiology: A Laboratory Manual TENTH EDITION.
He, F., Hu, W. and Li, Y., (2004). Biodegradation mechanisms and kinetics of azo dye 4BS by a microbial consortium. Chemosphere, 57(4), pp.293-301.
Kalme, S., Ghodake, G. and Govindwar, S. (2007). Red HE7B degradation using desulfonation by Pseudomonas desmolyticum NCIM 2112. International Biodeterioration & Biodegradation, 60(4), pp.327-333.
Kolhe, A.S. and Pawar, V.P. (2011). Physico-chemical analysis of effluents from dairy industry. Recent Research in Science and Technology, 3(5).
Lade, H., Kadam, A., Paul, D. and Govindwar, S. (2015). Biodegradation and detoxification of textile azo dyes by bacterial consortium under sequential microaerophilic/aerobic processes. EXCLI journal, 14, p.158.
Ladwani, K.D., Ladwani, K.D., Ramteke, D.S. and Deo, S. (2016) Detection and Identification of Organic Compounds in Wastewater of Final Effluent Treatment Plant by FTIR and GC-MS. Journal of Advanced Chemical Sciences, pp.246-247.
Lu, X. and Liu, R.(2010). Treatment of azo dye-containing wastewater using integrated processes. In Biodegradation of azo dyes (pp. 133-155). Springer, Berlin, Heidelberg.
Mansour, H.B., Ghedira, K., Barillier, D., Ghedira, L.C. and Mosrati, R.(2011). Degradation and detoxification of acid orange 52 by Pseudomonas putida mt-2: a laboratory study. Environmental Science and Pollution Research, 18(9), pp.1527-1535.
Mansour, H.B., Houas, I., Montassar, F., Ghedira, K., Barillier, D., Mosrati, R. and Chekir-Ghedira, L. (2012). Alteration of in vitro and acute in vivo toxicity of textile dyeing wastewater after chemical and biological remediation. Environmental Science and Pollution Research, 19(7), pp.2634-2643.
Mohabansi, N.P., Tekade, P.V. and Bawankar, S.V. (2011). Physico-chemical parameters of textile mill effluent, Hinganghat, Dist. Wardha (MS). Current World Environment, 6(1), pp.165-168.
Nachiyar, C.V., Sunkar, S., Karunya, A., Ananth, P.B. and Jabasingh, S.A., (2016). Aerobic bacterial consortium CN-1: Potential degrader of azo dyes. Journal of Environmental Biology, 37(3), p.361.
Ogugbue, C.J. and Sawidis, T. (2011). Bioremediation and detoxification of synthetic wastewater containing triarylmethane dyes by Aeromonas hydrophila isolated from industrial effluent. Biotechnology research international, 2011.
Omar, H.H., (2008). Algal decolorization and degradation of monoazo and diazo dyes. Pak J Biol Sci, 11(10), pp.1310-1316.
Patel, R., Tajddin, K., Patel, A. and Patel, B.(2015). Physicochemical analysis of textile effluent. International Journal of Research and Scientific Innovation, II, pp.33-37.
Puvaneswari, N., Muthukrishnan, J. and Gunasekaran, P., (2006). Toxicity assessment and microbial degradation of azo dyes.
Rice, E.W. ed., (2012). Standard methods for the examination of water and wastewater, 10. Washington, DC: American Public Health Association.
Santisi, S., Cappello, S., Catalfamo, M., Mancini, G., Hassanshahian, M., Genovese, L., Giuliano, L. and Yakimov, M.M., (2015). Biodegradation of crude oil by individual bacterial strains and a mixed bacterial consortium. Brazilian Journal of Microbiology, 46(2), pp.377-387.
Sarkar, S., Banerjee, A., Halder, U., Biswas, R. and Bandopadhyay, R.(2017). Degradation of synthetic azo dyes of textile industry: a sustainable approach using microbial enzymes. Water Conservation Science and Engineering, 2(4), pp.121-131.
Sivaranjani, A., Madhan, B. and Barathidasan, K. (2013). Decolourization of Acid Red 131 by using Shigella sp. Isolated from Tannery Effluent. International Journal of Pharmaceutical & Biological Archives, 3(5), pp.142-146.
Yaseen, D.A. and Scholz, M. (2019). Textile dye wastewater characteristics and constituents of synthetic effluents: a critical review. International journal of environmental science and technology, 16 (2), pp.1193-1226.
Yin, H., Qiu, P., Qian, Y., Kong, Z., Zheng, X., Tang, Z. and Guo, H., (2019) Textile Wastewater Treatment for Water Reuse: A Case Study. Processes, 7(1), p.34.


