1*Student of Ph D, Department of Biotechnology, Iranian Research Organization for Science and Technology, Tehran, Iran
2Professor, Department of Biotechnology, Iranian Research Organization for Science and Technology, Tehran, Iran
3Assistant Professor, Department of Biotechnology, Iranian Research Organization for Science and Technology, Tehran, Iran
4Associate Professor, Department of Microbiology, College of Medicine, Tehran Medicine University, Tehran, Iran
5Professor, Department of Soil Science, Faculty of Agriculture, University of Tabriz, Tabriz, Iran
Article Publishing History
Received: 27/07/2016
Accepted After Revision: 10/09/2016
Most of polycyclic aromatic hydrocarbons (PAHs) are highly toxic, mutagenic, carcinogenic and teratogenic in nature and exposure to these PAHs represents public health risks and raises environmental concerns. Significant interest in the potential for bioremediation of PAHs on the contaminated sites has resulted in considerable progress in understanding biodegradation of PAHs and degradation pathways. Much of this research has focused on individual compounds, whereas on contaminated sites PAHs usually occur as mixtures of compounds. Four triplicate batch tests were used to establish the conditions for maximum degradation of naphthalene, phenanthrene and anthracene by changing the following four factors one at a time: temperature, pH, reaction time and pollutant concentrations. The optimal reactor conditions for the degradation of PAH, at the concentration of 1000 mg/L, were found to be 28ºC in pH= 7, under the reaction time of 7 days. The degradation of PAHs by these bacteria was confirmed by GC-MS analysis and UV- spectroscopy. These bacteria were capable of degrading a mixture of PAHs with low and high molecular weight and degradation efficiency was found tobe 57.1% for naphthalene, 82.1 % for phenanthrene and 55.2 % for anthracene at the end of 7 days. Fourteen types of bacteria were isolated and in 1000 mg/L of PAH destruction level, they achieved 3.3-82.1% of these compounds. Therefore this method is capable of removing PAHs from contaminated oily soil.
Biodegradation, Pahs, Pollutant, Degradation, Oily Soil
Sadighbayan K. H, Assadi M. M, Farazmand A, Monadi A. R, Aliasgharzad N. Biodegradation of Naphthalene, Phenanthrene and Anthracene (Pahs) With Bacteria in the Oily Soil of Tabriz. Biosc.Biotech.Res.Comm. 2016;9(3).
Sadighbayan K. H, Assadi M. M, Farazmand A, Monadi A. R, Aliasgharzad N. Biodegradation of Naphthalene, Phenanthrene and Anthracene (Pahs) With Bacteria in the Oily Soil of Tabriz. Biosc.Biotech.Res.Comm. 2016;9(3). Available from: https://bit.ly/2oZpdkK
Introduction
Nowadays, Petroleum products are one of the most widely used chemicals in society. With the massive quantity of fuel required to power automobiles and heat homes, and the number of times each gallon of petroleum is stored, transported or transferred, accidents and leakages are unavoidable (Andrea et al., 2001). Petroleum contamination results from leaking aboveground and underground storage tanks, spillage during transport of petroleum products, abandoned manufactured gasoline sites, other unplanned releases, and current industrial processes (Gilchrist et al., 1998). As petroleum contains hazardous chemicals such as benzene, toluene, naphthalene, anthracene and phenanthrene (PAHs), this contamination can be hazardous to the health of plants, animals, and humans. PAHs are a group of chemicals consisting of carbon and hydrogen, arranged in the form of two or more aromatic rings. The metabolism of PAHS in human body produces epoxide compounds with mutagenic and carcinogenic properties and in some cases, lung, intestinal, liver, pancreas and skin cancers have been reported (Zhang et al., 2004).
Release of PAHs into the environment can lead to the contamination of the natural resources. Their complex molecular structure and low solubility in water, limit the application of conventional remedial techniques (Abd El Latif et al., 2009). Iran has the 9% of the world’s petrol sources. Persian Golf and southern parts of Iran have been polluted by160000 tons of petrol and petroleum substances for years (Lakzian., 2007). In 1991, more than 5 tons of crude oil were spilled into Persian Golf waters and caused the death of plants and sea side ecosystem of the Persian Golf (Koochekzadeh., 2009). In Jan. 2010 an oil well in Maroon of Khuzestan started to leak and more than 20000 barrels of crude oil were spilled into the area 100 hectares of the surrounding area were contaminated. But unfortunately there’s no accurate information about these polluted areas (Koochekzadeh, 2009).
Bioremediation is a strategy for the removal of PAHs because some microorganisms can metabolize PAHs to inert substances, CO2 and water. Microbial PAH degradation occurs through successive oxidations catalyzed by mono and dioxygenase enzymes that require iron as cofactor. Bio- surfactant production by PAHs- degrading bacteria may enhance the bioavailability of PAHs in the environment (Samanta et al., 2002).
Although the aromatic hydrocarbons are generally more resistant to biodegradation, some aromatic hydrocarbons with low-molecular- weight such as naphthalene, may actually be oxidized before they are saturated. Mono aromatic hydrocarbons are toxic to some microorganisms due to their solvent action on cell membranes, but in low concentrations they are easily biodegradable under aerobic conditions (Eder et al., 2006). PAHs with 2–4 rings are less toxic and biodegradable at the rates that decrease with the level of complexity. PAHs with five or more rings can only be degraded through co- metabolism, in which microorganisms fortuitously transform non-growth substrates while metabolizing simpler hydrocarbons or other primary substrates in the oil (Jussara et al., 2015).
Alkylated aromatic are compounds degraded less faster than their parent compounds; the more highly alkylated groups are degraded less faster than less alkylated ones. The metabolic pathways for the biodegradation of aromatic compounds have been the subject of extensive study (Li et al., 2006). The bacterial gradation of aromatic compounds normally involves the formation of a diol, followed by ring cleavage and formation of a dicarboxylic acid. Fungi and other eukaryotes normally oxidize aromatic compounds using mono-oxygenases and forming trans-diols (Chanieau et al., 2005).
Material And Methods
Mixed Bacterial Culture Preparation
Different regions of contaminated soil in Tabriz refinery were sampled in order to isolate effective bacteria. After digging cavities of a depth of 30 cm in each specific area, approximately 400 g soil was sampled. Each sample was poured in an unclosed plastic bag and then transferred to the laboratory. Information about sample including sampling location, altitude and latitude was attached on each packet (Curl et al., 2007).
After preparing 10-1 to 10-4 concentrations from samples in physiologic serum, 100 µl of concentrations were cultured in a plate containing starch casein agar and incubated at 28°C for one week. Strains of g1, g2, g3, g4 and g5 (from Tabriz Refinery soil) were already isolated by enrichment culture technique on separate nutrient agar plates (Glucose, Malt extract and yeast extract agar) and incubated at 28 ºC for 24 h. Each colony of these microorganisms was transferred by TSB broth in order to prepare 0.5 Mac farland suspension (Burges., 2006; Bardi et al., 2000). They were put separately into Erlenmeyer flasks (250 ml) containing 100 ml of the Muller Hinton broth, 10 mg of PAHs and 2ml of bacteria suspension and incubated for 7 days at 28 ºC on an orbital shaker at 100 rpm, then culture medium was centrifuged for 15 min. PAH residues were separated with 5ml toluene and decantation (Bardi et al., 2000).
Spectroscopic Analysis
Cultured fluid without algae incubated under the same conditions since the test specimens were used as the control. The percentage of PAH destruction by bacteria was calculated by the following relationship:
Destruction percentage = A1 – A2 × 100
A1
A1: hydrocarbon absorption before destruction
A2: hydrocarbon absorption after destruction by microorganism
Gc Mass Analysis For Determination Of Metabolites
The GC mass analysis was preformed on Shimadzu GC 2010 equipped with spilt (rate: 100) as an injection mode in order to determine the metabolites resulted from bacterial degradation. The CPED1-M25-025 column was used and the length of column was 24.9 m. Temperature within column which was 8°C for 9 minutes, increased by 150°C. The maximum temperature was 325°C (temperature program begins from 80°C) and system conditions were as follows:
Hold time = 1 min, Detection system FID= 310°C, Injection system STL= 300°C (Kishore et al., 2007). The wavelengths of PAHs were as follows, Naphthalen (288 nm), Phenanthrene (296 nm), and Anthracene (360 nm).
Three complete metabolites from most biological destructions were identified by GC-Mass (Farinazleen., 2004). The findings of this research reveal that by improving and optimizing the conditions, having effective bacteria, analyzing and identifying produced metabolites from destruction processes and employing a pilot study, the rectification of contaminated soil by PAHs and production of useful metabolites such as different acids, alcohols and other harmless substances can be carried out.
Results And Discussion
The present results showed that different viable species belonging to bacteria possess varied activities in biological biodegradation of a variety of PAHs depending on the type of the aromatics, its concentration and the bacteria species. Table 1 and Fig. 1-3 indicate biodegradation of Naphthalene, Phenanthrene and Anthracene by g1, g2, g3, g4 and g5 and exhibit ability to degradate of 10 ppm PHAs after 7 days.
 |
Figure 1: Absorption spectra of biodegradation of Naphthalene |
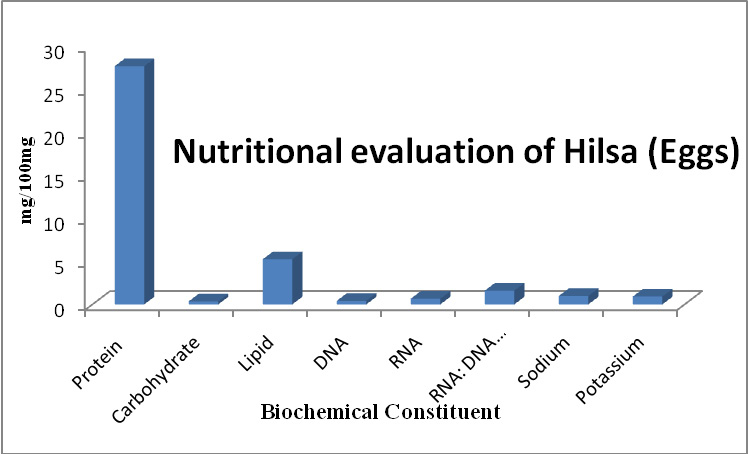 |
Figure 2: Absorption spectra of biodegradation of Phenanthrene |
 |
Figure 3: Absorption spectra of biodegradation of Anthracene |
| Table 1: Percentage of degradation of different PAHs by different bacteria | ||||||
| Degradation % | ||||||
| PAHs | Day | g1 | g2 | g3 | g4 | g5 |
| Naphthalene | 7 | 41.4 | 35.3 | 30.1 | 24.1 | 57.1 |
| Phenanthrene | 7 | 23.4 | 29.1 | 33.3 | 30.3 | 82.1 |
| Anthracene | 7 | 28.6 | 29.2 | 6.2 | 12.6 | 55.2 |
Gas chromatography analysis of the PAHs (Fig. 4-6) suggest that the high biodegradation activity in 7 days, which could be a consequence of synergistic interactions among different strains composing the bacteria, allowed degradation of aromatic compounds with high molecular weight.
 |
Figure 4: Gas chromatography analysis for biodegradation of Naphthalene |
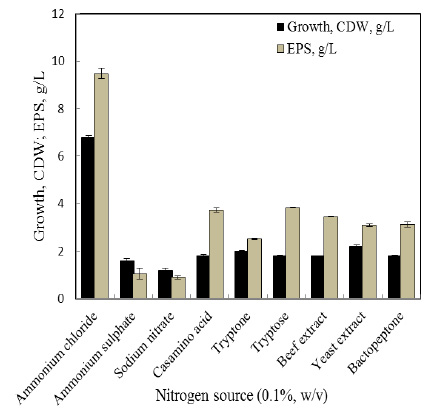 |
Figure 5: Gas chromatography analysis for biodegradation of Phenanthrene |
 |
Figure 6: Gas chromatography analysis for biodegradation of Anthracene |
In order to study, bacteria were isolated from farmland sludge of Tabriz Refinery which had potential degradation mechanisms. The bacteria with codes of g1, g2, g3, g4 and g5 increased biodegradation rate of the PAHs from oily soil. Inoculation of the oil-degrading bacteria complimented oil degradation and thus enhanced biodegradation of PAHs. The synergetic interaction among the oil degrading bacteria degraded higher amount of the polycyclic aromatic hydrocarbons. The bacteria isolated from different locations can be used to formulate bacterial consortia. The purpose of the present study was to assess the ability of an indigenous amount of bacteria from a petroleum refinery waste site to degrade PAHs and to emulsify other hydrocarbon substrates.
Bioremediation is a natural process by which pollutants are recycled rather being buried. Furthermore, from the public point of view, bioremediation is more desirable and most of the world organizations disseminate this method to correct and improve damaged regions by removing environmental contaminants (Kishore et al., 2007). One of the best bioremediations are biologic methods and the use of microorganisms. Bacteria are of the greatest importance compared with other microorganisms because of their different reductive enzymes (Chanieau et al., 2005). Considering the results of the present study and conducted studies, soil bacteria, more or less, have the potential of reduction and destruction of polycyclic aromatic hydrocarbons.
In the study conducted by our colleagues on reductive microorganisms of PAHs, they succeeded to identify a yeast (AH70) isolated from oil-polluted soils which was determined as a 100% alkaline sequence homologue in determining genomic sequence of 26S rRNA. This organism is able to destruct Naphthalene at the rate of 89.76%, Phenanthren at the rate of 77.21%, Pyrene at the rate of 60.77%, and Benzopyrene at the rate of 55.53% during 10 days (Abd El Latif et al., 2009).
Andrea R.Clement et al. reported that two yeast species are identified among studied soil microorganisms that the species 984 was able to destruct Anthracene at the rate of 64%±10 and the species 870 was able to destruct Naphthalene at the rate of 69%±10 (Andrea et al., 2001).
C.H.Chaineau et al. in their research titled “The effects of nutritions on crude oil biological destruction by soil microbial population in a farm soil”, increased the rate of these materials’ reduction from 47% to 62% (Chanieau et al., 2005).
Zhang.H.Kallimanis et al. demonstrated that a species of Pseudomonas was able to solve 35 mg/l Phenanthrene at the presence of surfactant produced by that bacterium and finally caused the destruction of Phenanthrene (Zhang et al., 2004).
In the present study, isolation of some of soil bacteria from polluted soil of Tabriz refinery was conducted; then, treatment was done and finally the destruction of hydrocarbons PAHs by these microorganisms was performed, and different percentages of destruction were observed and reported. Considering the results and findings of the present study, isolated bacteria from soil have the potential to reduce the oil hydrocarbons invitro. Rich farmlands which have significant resources of phosphorous, nitrogen and sulfur, also confirm this
finding.
Bhattacharya et al. isolated 150 stubs of oil-hydrocarbons destructive bacteria from India oil-polluted soils and demonstrated that Ps.citronellolis are dominant considering the destructive ability of aromatic and aliphatic compounds (Bhattacharya et al., 2003).
Barati and Vasudevan in their research titled “using oil-hydrocarbons by isolating Ps. fluorescence from oil-polluted soils” demonstrated that the microbial stub had the significant power to destruct short and long-chain alkalenes (Barathi et al., 2001).
Eder et al. isolated a stub of Pseudomonas from oil-polluted soil of refinery which had the 72% of destruction power by producing surfactants (Eder et al., 2006).
Considering the mentioned studies, the present study has had similar and confirmative results and innovation aspects due to the use of native and wild microorganisms of Tabriz refinery soils.
Acknowledgements
This study was adopted from a research plan which was supported financially by Tabriz Petroleum Refinery. Authors of this essay acknowledge the Research Vice President of Refinery and Laboratory Technicians of Chemical Science Faculty of University of Tabriz for their sincere cooperation.
References
Abd El Latif H, Saad A, Alamri S, Motamed E, Hashem M (2009). Isolation and molecular genetic characterization of a yeast strain is able to degrade petroleum polycyclic aromatic hydrocarbons, Afr. J. Biotechnology, 8(10): 2218-2223.
Andrea R, Clemente T, Anazawa A, Lucia R (2001). Biodegradation of Polycyclic Aromatic Hydrocarbons by Soil Fungi, Braz-J- Microbiol, 23(4): 127-133.
Gilchrist T.L (1998). Heterocyclic chemistry, Second edition, Longman, Scientific & Technical.
Barathi S, Vasudevan N (2001).Utilization of petroleum hydrocarbons by Ps. fluorescens isolated from a petroleum- contaminated soil, Jour. Environment International, 26(5-6): 413-416.
Bardi L, Mattei A, Steffan S, Marzona M (2000). Hydrocarbon degradation by a soil microbial population with β-cyclodextrin as surfactant to enhance bioavailability, J. Enzyme and Microbial Technology, 27(9): 709-713.
Bhattacharya D, Sarma P.M, Krishanan S, Mishra S, Lal B (2003). Evaluation of genetic diversity among Pseudomonas sp, Jour. African Journal of Biotechnology, 20: 120-130.
Burges A (1998). The microorganisms in the soil, Hutchinson, London.
Farinazleen Mohamad Ghazali (2004). Biodegradation of hydrocarbons in soil by microbial consortium, Jour. International Biodeterioration & Biodegradation, 54(1):61-67.
Chanieau C.H, Rougeux G, Yepremian C (2005). Effects of nutrient concentration on the biodegradation of crude oil and associated microbial populations in the soil, Soil Biology and Biochemistry, 37(8): 1490-1497.
Curl E.A, Truclove B (2007). The Rhizosphere [Springer- 15th ed]. Verlag, Berlin.
Eder C, Rodrigo J.S, Jacques F.M, Bento M, Carmo R, Peralba P.A, Selbach E, Flavio A.O (2006). Anthracene biodegradation and surface activity by an iron – stimulated Pseudomonas sp, Braz-J-Microbial, 50: 88-93.
Farinazleen M.G (2004). Biodegradation of hydrocarbons in soil by microbial consortium, International Biodeterioration & Biodegradation, 54(1): 61-67.
Jussara P, Francisca P (2015). Biodegradation of crude oil in sandy sediment, International Biodeterioration & Biodegradation, 44(2-3): 87-92.
Kishore D, Ashis K (2007). Crude petroleum-oil biodegradation eYciency of Bacillus subtilis and Pseudomonas aeruginosa strains isolated from a petroleum-oil contaminated soil from North-East India, Bioresource Technology, 98: 1339–1345.
Koochekzadeh M (2009). Oil and petroleum microbiology, Adabestan jahan no, Tehran –Iran: 83-88.
Lakzian A (2007). Principle and utilization of soil microbiology, Ferdosi University, Mashhad – Iran: 234-241.
Li W, Suzelle B and Jin-Woo K (2006). Biodegradation of pentyl amine and aniline from petrochemical wastewater, Environmental Management, 83: 191-197.
Samanta SK, Singh OV and Jain RK (2002). Polycyclic aromatic hydrocarbons: Environmental pollution and bioremediation, J. Trends in Biotechnology, 20: 243-248.
Zhang H, Kallimanis A, Koukkou A.I, Drainas C (2004). Isolation and characterization of novel bacteria degrading polycyclic aromatic hydrocarbons from polluted Greek soils, Applied Microbiology and Biotechnology, 65: 124-131.


