1,2National Organization for Drug Control and Research (NODCAR), Medicinal Plants Dept.
(1Tissue Culture & 2Plant Physiology), Egypt.
2Northern Border University, Biology Dept. Division of Botany (Plant Physiology), Faculty of Science, KSA.
3Cairo University, Faculty of Agriculture, Biochemistry Dept., Egypt.
4Department of Environment and Plant Pasture, Desert Research Center, Mathef El-Mataria, Egypt.
5Northern Border University, Chemistry Department, Faculty of Science, Girl Division, Arar- Kingdom of Saudi Arabia
Article Publishing History
Received: 29/11/2016
Accepted After Revision: 28/12/2016
The physiological and phytochemical responses of Artemisia abrotanum to foliar application of different concentrations of amino acids (tryptophan and phenylalanine) and citric acids acid were studied during two successive seasons (2014-2015). The results showed that foliar application of either amino acids or citric acid significantly promoted the growth parameters in terms of plant height, a number of branches, fresh, and dry biomass. All treatments led to significant increments in essential oil content and yield. Plant growth and essential oil parameters gradually increased with amino acids or citric acid concentrations. Gas-liquid chromatographic analysis revealed that the major identified components of essential oil were Chamazulen, p-Cymen-8-ol and piperitone. Organic and amino acid treatments resulted in improving quantity and quality of essential oil components.
Amino Cids And Citric Acid, Artemisia Abrotanum L., In Vitro Culture
EL-Zefzafy M. M, Shahhat I. M. A, Yousef R. S, Elsharkawy E. R. Influence of Foliar Application With Amino Acids and Citric Acid on Physiological and Phytochemical Responses of Artemisia Abrotanum Produced by in Vitro Culture. Biosc.Biotech.Res.Comm. 2016;9(4).
EL-Zefzafy M. M, Shahhat I. M. A, Yousef R. S, Elsharkawy E. R. Influence of Foliar Application With Amino Acids and Citric Acid on Physiological and Phytochemical Responses of Artemisia Abrotanum Produced by in Vitro Culture. Biosc.Biotech.Res.Comm. 2016;9(4). Available from: https://bit.ly/2O5i6B2
INTRODUCTION
The genus Artemisia belongs to one of the largest and most widely distributed genera of the family Asteraceae. It is a diverse and economically important genus and it has more than 500 species. Artemisia is a wind pollinated cosmopolitan genus, mainly distributed in temperate areas of mid to high latitudes of the northern hemisphere, colonizing in arid and semiarid environmental landscape. Most plants within this genus possess ethnomedical and biological properties related to antiviral andantimalarial. antifungal, anticoagulant, hypoglycemic, and antispasmodic activities, some plants of the genus are used as foodstuff, ornamentals or soil stabilizers, some are allergenic or toxic, and some are weeds growing in the fields, (Zafar, et al.,1990 Nin,et al 1997, Valles & McArthur 2001, Bnouham et al., 2002 and Hayat et al., 2009).
Artemisia species invariably found as small fragrant shrubs or herbs and most yield essential oils. Some of these oils found used medicine .The plants themselves are popular among gardeners as cultivated ornamentals. essential oil of Artemisia arborescens has been used in the treatment of inflammation, intestinal trouble, and diarrhea and antioxidant. A. abrotanum from Cuba had trans-sabinyl acetate and á-terpineol as main oil compounds. The same species collected in Serbia displayed silphiperfol-5-en-3-one A (14.6%), ascaridole (13.1%), 1,8-cineole (10.5%), á-bisabolol oxide A acetate (8.7%) as main oil components . Similarly, an oil from Northwestern Italy had 1,8-cineole (34.7%), bisabolol oxide (18.4%) and ascaridole (16.0%) as main compounds. The dominant components in the oil from the Crimea were 1,8-cineole and camphor. A German A. abrotanum showed 1,8-cineol as main oil compound . Plant cultivated in Poland were rich in piperitone (17.5%), davanone (16.8%), 1,8-cineole (12.5%) and silphiperfol-5-en-3-ol A (6.3%) (Kordali et al 2005 and Jerkovic et al., 2003, Orav, et al., 2006, Lopes-Lutz et al., 2008 Sharopov et al 2012, and Ghasemi Pirbalouti et al., 2013).
Amino acids as organic nitrogenous compounds are the building blocks in the synthesis of proteins (Hounsome et al., 2008). Amino acids are particularly important for stimulation cell growth, they act as buffers which help to maintain favorable PH value within the plant cell, since they contain both acid and basic groups; they remove the ammonia from the cell. (Abdel Aziz et al., 2010). Amino acid formulations, mixtures of nutrients, hydrolyzed proteins, humic acids, seaweed extracts and brassinolides are proposed as a commonly used growth promoters (Thomas et al., 2009). The application of amino acids can stimulate the performance of plant (Abdel-Mawgoud et al., 2011). The role of Tryptophan is well known: it has an indirect role in the growth via its influence on auxin synthesis, (Talaat, et al, 2002).The yield-contributing characters and quality of plants could be improved by foliar application of glutamine (Amin et al., 2011).
Citric acid is a six carbon organic acid, having a central role in CA cycle in mitochondria that creates cellular energy by phosphorylative oxidation reactions. It is created by the addition of acetyl-CoA to oxaloacetic acid that is converted to succinate and malate in next steps (Wills et al., 1981). Despite the proposed benefits of the application of amino acids on plant growth, there is not many studies about the physiological changes induced by foliar applied amino acids and organic acid together , in medicinal plants in which their biochemical constituents are important. Especially essential oil.
This research was conducted to evaluate the physiological changes induced by foliar applied active formulations of amino acids and citric acid on the Artemisa plant produced by in vitro culutur. The foliar applications of three different concentrations of Aminol-Forte (50,150, 250.) were applied to plant to study their effect on the growth, presence of phytochemical compounds and improving of quality and quantity of essential oil.
MATERIAL AND METHODS
A field experiment was carried out for two successive seasons (2014 – 2015) in Applied Research Center of Medicinal Plants (ARCMP), National Organization for Drug Control and Research (NODCAR), Egypt in order to examine the physiological and phytochemical responses of Artemisa plants to foliar applied of amino acids and citric. To achieve the study purpose, healthy plantlets (rooting stage in average tall 6 – 8 cm) were selected from tissue culture laboratory, (ARCMP), National Organization for Drug Control and Research (NODCAR), Egypt. Plant materials were washed from phytagel under running tap water and soaked in a solution of fungicide (0.2 % Benlet), then transferred to green house in plastic containers (5 cm) full of peat moss/sand (2/1 –V/V) for five weeks. Then plants were transferred to plastic containers (10 cm) full of peat moss /sand (2/1- V/V) for five weeks too.
After an acclimatization period finished (ten weeks), plants were transferred from a greenhouse to open field conditions in 1st Marsh of 2014/2015 season. The experimental soil was prepared and divided into ten plots, each plot area was 7 m2 (3.5 × 2), with six rows at 60 cm apart and 40 cm between hills. The mechanical and chemical analysis of the soil were carried out before planting, according to Jackson (1967), the obtained results were as shown in Table 1. Ammonium nitrate 33.5% N (200 Kg/fed), Calcium superphosphate 16 %P2O5 (150 Kg/fed) and potassium sulfate 48% K2O (50 Kg/fed) were added in three doses to fertilize the Artemesia plants. The first dose was added at 21 days before the first cut, the second and third doses were added after 21 days of the first and second cuttings, respectively.
The plants were sprayed twice after 60 days from planting and after first cut with freshly prepared solutions of amino acids (Tryptophan or phenylalanine) and citric acid at the concentrations of 50, 150 and 250 mg/l as well as untreated plants (control; distilled water). The spraying was done manually using a spraying bottle.
Physiological response parameters
Two cuts were taken during the growing seasons (the first one on June and the second on September).The physiological response parameters were recorded at each cut as plant height (cm), a number of branches / plant, herb fresh weight (g/plant) and herb dry weight (g/plant).
Phytochemical response parameters: Determination of essential oil percentage, %
The percentage volatile oil of air dried herb for each treatment was determined by hydro-distillation of vegetative parts of plants (100g) of each treatment for 3h according to British pharmacopeia (1980). The essential oil was dried over anhydrous Na2SO4, stored in a dark glass bottle and kept at 4°C until it was analyzed.
Chemical analysis of the volatile oil
The analysis of the volatiles was performed using a Hewlett-Packard 6890 GC linked to a Hewlett- Packard 5973 mass-selective detector. For the analysis a Zebron ZB-5MS, capillary column (27 m × 250 μm i. d., 0.25 μm film thickness) was used. The carrier gas was helium at 1.3 ml/min in constant flow mode. The injector temperature was 250°C, the injection volume 1 μl, and the split ratio 1:20. The initial oven temperature of 60°C was held for 1 minute, then increased at a rate of 5°C/min up to 220°C, and subsequently at a rate of 15°C/min up to 280°C, and finally was held isothermal for 1 min. The transfer line to the MSD was set at 280°C and the scan conditions were: M/Z 40–300, at 1.75 scans/sec. Prior to analysis 900 μl of the volatile fractions were mixed with 100 μl of biphenyl (2.0 mg/ml in hexane) as an internal standard. The components of essential oils were identified by comparing their relative retention times and mass spectra with those of Registry of Mass Spectral Data and literature citations.
Identification of essential oil components
The components of the were identified by comparison of their mass spectra with those of a computer library or with the authentic compounds and confirmed by comparison of their retention indices with those of authentic compounds. Kovats, indices (Kova´ ts, 1958) were determined by co-injection of the sample with a solution containing a homologous series of hydrocarbons, at a temperature run identical to that described above.
Statistical analysis
The experimental treatments were laid out in a randomized complete block design (RCBD) with 3 replicates. Data on plant height (cm), number of branches per plant, herb fresh weight (g/plant), herb dry weight (g/plant), essential oil (%) and yield of essential oil (ml/plant) were statically analyzed using the COSTAT 6311Win and means were compared using LSD at a probability level of 5%.
RESULTS AND DISCUSSION
Artemisia abrotanum plant was sprayed with phenylalanine, tryptophan, and citric acid at different concentrations and the results were presented in (Table 2). Results showed that all treatments significantly increased plant height, the number of branches, fresh and dry weights of aerial parts per plant. The most pronounced effect on these growth criteria was obtained at treatment 250 mgl-1 phenylalanine and 250 mgl-1 of citric acid. Foliar application of phenylalanine resulted in the greatest effect as compared with other treatments (tryptophan and citric acid). Data in Table 2 also clearly indicated that the lowest records of A. abrotanum growth characteristics were observed in plants cultivated in soil without any foliar application control).
Data in Table 2 show that all applications of amino acids (phenylalanine or tryptophan) and citric acid significantly enhanced the height of A. abrotanum plants as compared with control in both seasons, (Fig. 1). The highest plant height (71.50 and 69.56 cm) was recorded at 250 mgl-1 of phenylalanine and tryptophan, respectively, while the control achieved a minimum plant height (50.58 cm). It was also observed that there was no significant difference between control and the results obtained at 50 mgl-1 of all amino acids and citric acid.It is obvious from the data in Table 2 that, all amino acids and citric acid treatments (50, 150 and 250 mgl-1) gradually increased the number of branches/plant (Fig. 2). Moreover, this increasing was significant, except that at 50 mgl-1 with tryptophan and citric acid, where there was no significant effect of these treatments on the number of branches. The average minimum of branches number (31.71±3.55) was recorded with control, while the high concentration of tryptophan (50 mgl-1) gave the maximum result of this parameter (52.23±6.37) compared with control. Nevertheless, it was shown that there was no significant difference between tryptophan and phenylalanine at high concentration (250 mgl-1). Application of all treatment concentrations of amino acids and citric acid observed a higher significantly increased the number of branches in the second season than the first season.
 |
Figure 1: Effect of amino acids and citric acid (type and concentration) on plant height of A. abrotanum |
 |
Figure 2: Effect of amino acids and citric acid (type and concentration) on No. of branches of A. abrotanum |
Herb fresh weight significantly responded to foliar application of tryptophan, phenylalanine and citric acid at different concentrations compared with untreated plants (Table 2). This response was gradual with acid concentrations (Fig. 3). Phenylalanine was superior to the other acids (tryptophan and citric acid) at all concentrations in fresh biomass production. This acid achieved the highest fresh weight of herb (105.30±9.14 g/plant) with increment about 88.8% at 250 mgl-1 compared with the control which, gave the lowest herb fresh weight (80.35±10.56 g/plant). According to the statistical analysis conducted, it was cleared that there was no significant difference between the best treatment, 250 mgl-1, of tryptophan (33.34±0.71 g/plant) and citric acid (135.40±6.45 g/plant).
 |
Figure 3: Effect of amino acids and citric acid (type and concentration) on herb fresh weight of A. abrotanum |
The results of herb dry weight presented in Table 2, it was shown clearly that the amino acid and citric acid had a highly positive significant effect on the average herb fresh yield of A. abrotanum. This effect was observed in the two growing seasons, as well as the second season was superior to the first. Like all previous growth parameters, dry weight of Artemesia herb was also gradually increased by increasing the concentrations of all amino acids and citric acid (Fig 4). The increments of herb dry weight of Artemesia were estimated by range from 24.7% to 84.86% at 50 mgl-1 and 250 mgl-1 respectively, with citric acid compared with the control. Moreover, untreated plants gave the lowest herb dry weight (29.92±4.24 g/plant). On the other hand, it was shown that there were no significant differences between all treatments at the same concentration.
 |
Figure 4: Effect of amino acids and citric acid (type and concentration) on herb dry weight of A. abrotanum |
The results of essential oil content (%) and its yield (ml/plant) were presented in (Table 4, Fig. 5 and Fig. 6). Data in this table shown that the biosynthesis of essential oil and its average yield were significantly promoted as a result of foliar spraying with amino acids and citric acid. The increments in essential oil content are ranged between 10.3% and 47.84%, while in essential oil yield was between 41.46% and 182.93% compared with the control. It is also clear from the obtained data that essential oil content and its yield gradually increased with amino acid and citric acid level increases (Fig. 5 and 6). The highest recorded value of the average essential oil content (4.45±0.35 %) and yield of essential oil (1.16±0.12 ml/plant) were obtained in the herb of plants treated with phenylalanine at 250 mg/l. While the control gave minimum essential oil content (3.01±0.44 %) and oil yield (0.41±0.15 ml/plant). On the other hand, there are no significant differences between all the three acids (phenylalanine, tryptophan and citric) at the same concentration in terms of essential oil contents and essential oil yield.
 |
Figure 5: Effect of amino acids and citric acid concentrations on essential oil content |
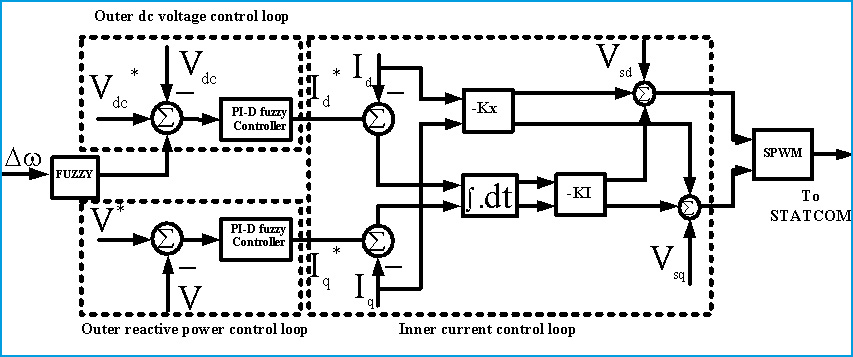 |
Figure 6: Effect of amino acids and citric acid concentrations on essential oil yield |
In this respect, several investigators have studied the effect of different amino acids on the total oil percentage and oil yield (ml/plant) and found the same results; Gamal El.Din et al., (1997) on Cymbopogon citratus Hort, Talaat and Youssef (2002) on basil plant, Karima et al., (2005) on Matricaria chamomilla L. Amino acids accumulation in plants plays different roles, an osmolyte, regulation of iron transport, modulating stomata opining and detoxification of heavy metals. Moreover, amino acids affect synthesis and activity of some enzymes, gene expression and redox-homeostasis (Rai, 2002).
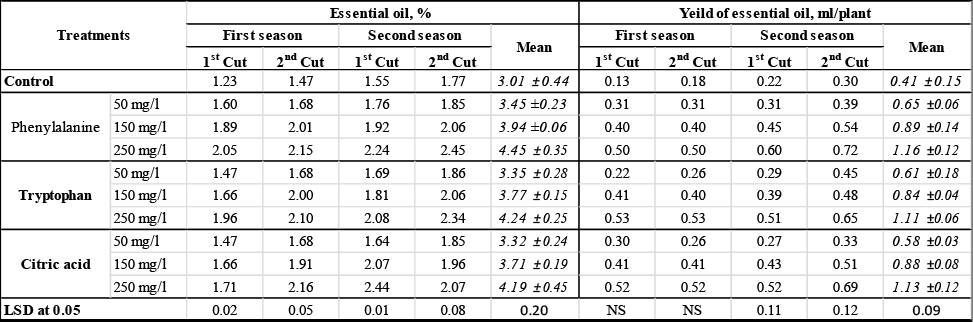 |
Table 1: Mechanical and chemical analysis of experimental soil |
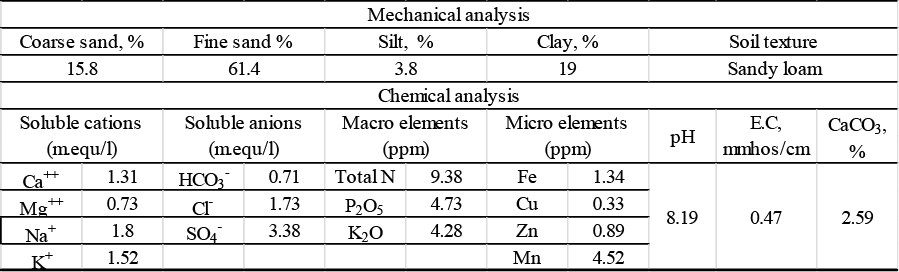 |
Table 2: Effect of amino acids and citric acid on physiological responses of A. abrotanum |
Many investigators, Talaat et. al. (2002) and Orner et.al. (2013), mentioned that foliar application of amino acids significantly increased essential oil percentage and yield. Also, Taraf et.al.(1999) on lemongrass, Eid et.al. (2010) on Jasminum grandiflorm and Safaa et.al. (2011) on geranium reported that foliar application of citric acid caused a pronounced increment in the percentage and yield of essential oil. It has been documented that the exudation of citrate and malate from roots of calcicole plants (plants growing in alkaline soils) enables them to extract P and Fe from such soils (Lopez-Bucio and Nieto-Jacobo, 2000). Use of citric acid alone or in combinations with malic acid increased the essential oil production of sweet basil (Jaafari and Hadavi, 2012). Foliar sprays of citric acid alone or in combination with Fe sources have been used to recover many plants from the iron chlorosis (Eidyan et al., 2014). Later studies revealed that the effect of citric acid is not just due to pH change and there are a variety of physiological responses to applied citric acid.
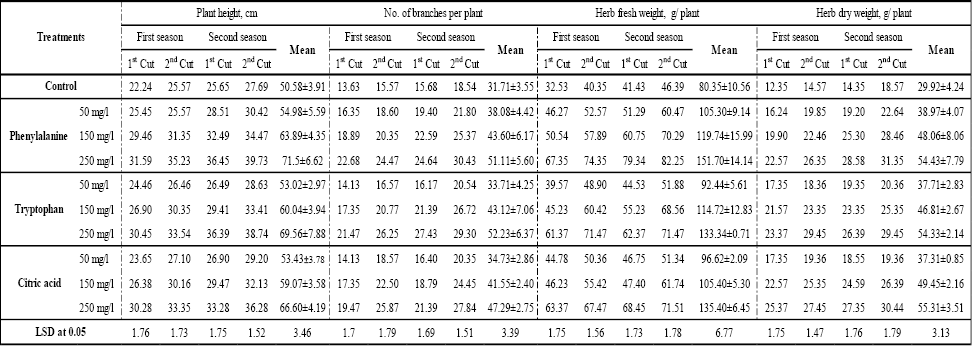 |
Table 3: Effect of amino acids and citric acid on essential oil % and yield of A. abrotanum |
The results of the GC/MS analysis of the essential oil of A. abrotanum. during the first and second season as are shown in Table (4,5). There are 18 identified compounds. Chamazulen was identified as the major compound in the different treatments ranged from 19.3 % to .23.5 %, the second main component, p-Cymen-8-ol ranged (12.39% and 12.55. %) in the essential oil followed with piperiton which was identified as the third main constituent in the essential oil and its relative percentage accounted for (12.16% and 12.23. %), while some compound found in traces amount as Camphen (0.79% and 1.61%).
Foliar application of amino acid indicate that phenylalanine at 250 mg/1 revealed that, maximum relative percentage of Chamazulen was (22.37% and 23.5 % ) for 1st and 2nd cuts, respectively during 1st season). In the 2nd season, the foliar application of phenylalanine at 250 mg/1 resulted in the highest relative percentage of Chamazulen 2nd cut, (Table, 4, 5) in both seasons. The data indicated that Cymen-8-ol reached to its maximum values 11.3% and 12.5. % for 1st and 2nd cuts, respectively) as a result of application of phenylalanine at 250 mg/ L during the 1st season. On the other hand, tryptophan at 250 mg /l gave the highest relative percentage 1,8-cineol which ranged between (10.22 to 10.95%) in the 1st and 2nd cuts during two seasons, as compared with control. while citric acid did show any significant change in some essential oil concentration than amino acid, like Eudesmol, pipeitone, Terpenol, camphene, Gerenyl isobutrate, terpenol and 1.8-cineol in First season, while second season citric acid showed significant decrease in the concentration of some oil component especially at low concentration 50 mg of citric acid.
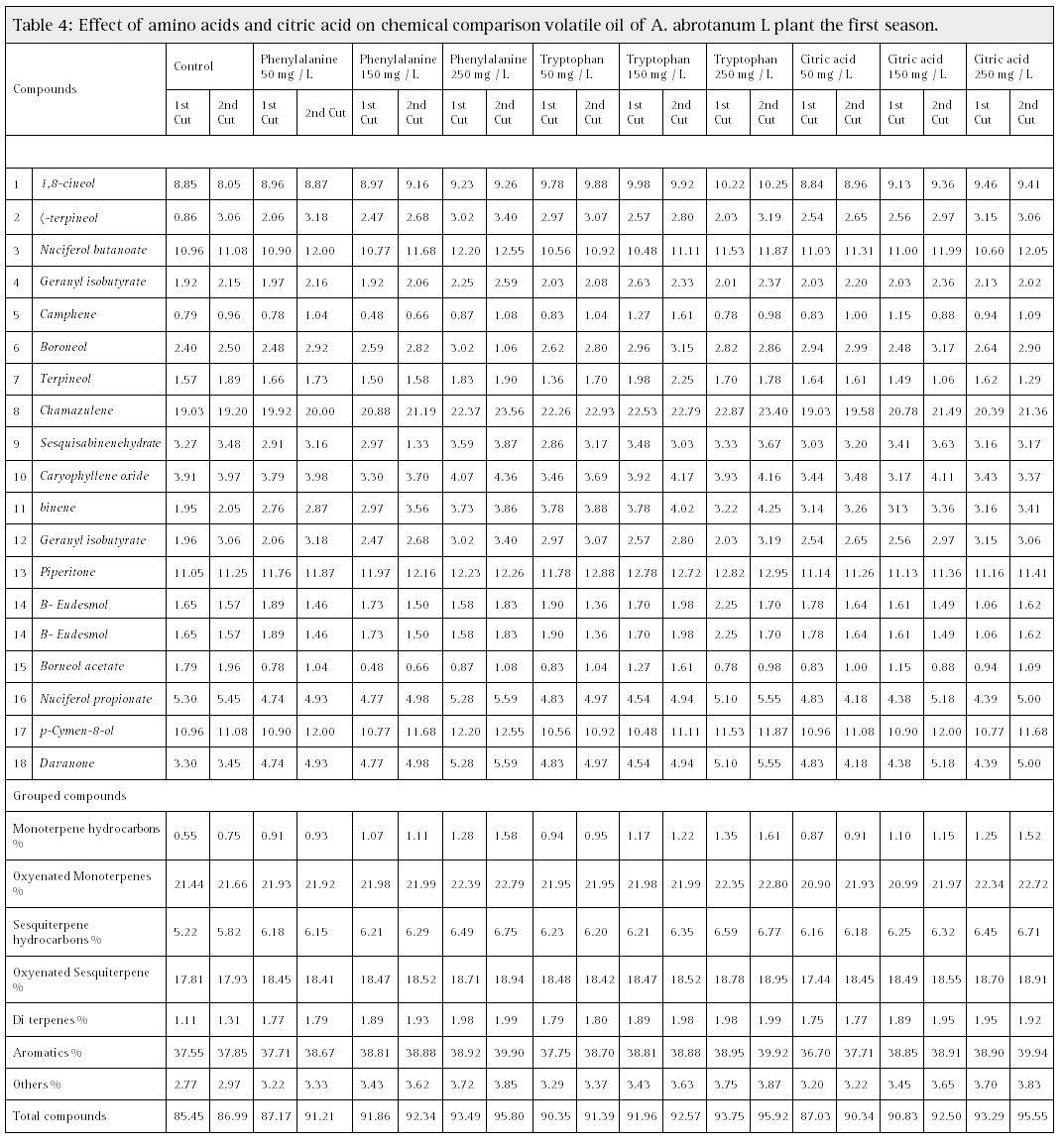 |
Table 4: Effect of amino acids and citric acid on chemical comparison volatile oil of A. abrotanum L plant the first season |
 |
Table 5: Effect of amino acids and citric acid on chemical comparison volatile oil of A. abrotanum L plant the second season |
The essential oil composition varies according to cuts or different treatments and was characterized by a high percentage of oxygenated compounds ranged from 21.9 to 22.8 %. The components of the essential oil in herb for different treatments during 2 cuts during both seasons were shown in Tables (4 and 5). The increment in oil% might be due to the increase in vegetative growth and nutrient uptake. Similar results were reported by Gharib (2006). The chemical composition of A. abrotanum oils is very variable, the chemovarieties and the environmental conditions caused to this fact. The major components from different origins were found to be linalool, 1,8-cineol, piperitone, davanone and silphiperfol-5-en-3-ol, several workers reported that essential oil components of are varied according to growth conditions., foliar application of salicylic of (Ocimum basilicum L.) and (Majorana hortensis) increased the production of quantity and quality of oil ( Gharib et al 2006). These results are similar to those obtained by (Orav, et al., 2006).
CONCLUSIONS
We conclude that there is a pure effect by sprayed of all treatments enhanced growth parameters, plant height ,the number of branches, the fresh and dry weight of plant, oil percentage and yield per plant for two cuttings relative to untreated controls. from the above explanation the important role of amino acids in enchaning the growth and production of different plant constituents which is in accordance with our results, also our results are in accordance with many investigators who reported that the application of amino acids increases the concentration of essential oil and growth rate. Foliar application of phenylalanine, gave the best effect on growth parameters at 250 mg,and increase the quantity and quality of essential oil, by incrasing the yield percentage of oil and increasing the concentration of major compounds in the oil, while the application of citric acid shows variable effect according to concentration and seasons.
DISCLOSURE STATEMENT
No potential conflict of interest was reported by the authors.
REFERENCES
Attoa, G.E., H.E. Wahba and A.A. Farahat, (2002). Effect of some amino acids and sulphur fertilization on growth and chemical composition of Iberis amara L. plants. Egyption J. Hort., 29:17-37.
Audesirk G. and Audesirk T. (1986). Biology Life and Earth. MACmillan Publising Company, New York, London. pp: 494-507.
Bnouham, M., Mekhfi, H., Legssyer, A., Ziyyat, A. (2002). Forummedicinal plants used in the treatment of diabetes in Morocco. Int. J. Diabetes & Ethnopharmacology.
Chialva F, Liddle PAP, Doglia G. (1983)Chemotaxonomy of wormwood (Artemisia absinthium L.) Composition of the essential oil of several chemotypes. Z Lebensm UntersForsch., 176: 363-366.Google Scholar.
Eid RA. (2010) Physiological Properties studies on essential oil of Jasminum grandiflorum L. as affected by some vitamins. Ozean Journal of Applied Sciences, 3,1: 87- 96.
Eidyan, B., Hadavi, E., and Moalemi, N. (2014). Pre-harvest foliar application of iron sulfate and citric acid combined with urea fertigation affects growth and vase life of tuberose (Polianthes tuberosa L.)‘por-par’. Hortic. Environ. Biotechnol. 55, 9–13. doi: 10.1007/s13580-014-0061-2.
El-Bahar, K.M., A.S. Ghanem and E.A. Omer (1990). Responses of Datura metel L. cell cultures to some amino acids. Egypt J. Appl. Sci., 5: 192-199.
Fatmaa A. E. G. (2006).Effect of Salicylic Acid on the Growth, Metabolic Activities and Oil Content of Basil and Marjoram, Int. J. Agri. Biol., Vol. 8, No. 4, 2006.
Gamal El-Din (1997), Physiological studies on the effect of some amino acids and microelements on growth and essential oil content in lemongrass (Cymbopogon citrates Hort). Agric. Sci. Mansoura Vniv., 22, 4229-4241.
Ghasemi Pirbalouti A, Firoznezhad M, Craker L, Akbarzadeh M. 2013. Essential oil compositions, antibacterial and antioxidant activities of various populations of Artemisia chamaemelifolia at two phenological stages. Rev Bras Farmacogn; 23:861-9.
Hayat MQ, Ashraf M, Khan MA, Jabeen S, 2009, Ethnobotany of the genus Artemisia L. (Asteraceae) in Pakistan. Ethnobot Res, 7: 147-162.
Hendawy, S.F. (2000): Physiological and chemical studies on Echinaceae purpurea L., plant. Ph.D. Thesies, Fac., Agric., Moshtohor, Zaga. Univ. Egypt.
Jaafari, N., and Hadavi, E. (2012). Growth and essential oil yield of basil (Ocimum basilicum L.) as affected by foliar spray of citric acid and salicylic acid. Z. Arznei Gewurzpfl. 17, 80–83.
Jackson, M. L.(1967). Soil chemical analysis.printice – Hall of India, P.144 – 197.
Jerkovic I, Mastelic M, Milos M, Juteau F, Masotti V, Viano J (2003) Chemical variability of Artemisia vulgaris L. essential oils originated from the Mediterranean area of France and Croatia. Flavour Fragrance J.18: 436-440. 10.1002/ffj.1246.
Karima MG and MSA Abd EL-Wahed.(2005)Effect of Some Amino Acids on Growth and Essential Oil Content of Chamomile Plant. International Journal of Agriculture & Biology 7, 3: 376 – 380.
Kordali S, Kotan R, Mavi A, Cakir A (2005) Determination of the chemical composition and antioxidant activity of the essential oil of Artemisia dracunculus L. and of the antifungal and antibacterial activities of turkish A. dracunculus, A. absinthium and Santonicum essential oil. J Agric Food Chem. 53: 9452-9458..View ArticleGoogle Scholar.
Ljung Karin (2013). Auxin metabolism and homeostasis during plant development. Development 140, 943-950.
Lopes-Lutz D, Alviano DS, Alviano CS, Kolodziejczyk PP. (2008). Screening of chemical composition, antimicrobial and antioxidant activities of Artemisia essential oils. Phytochemistry; 69:1732-8.
McArthur E.D & D.J. Fairbanks. (2000). Proceedings of Provo, Utah. US Department of Agriculture, Rocky Mountain Research Station, Ogden.
Omer, E.A.; H.A.H. Said-Al Ahl; A.G. El Gendy; Kh. A. Shaban and M.S. Hussein (2013). Effect of amino acids application on production, volatile oil and chemical composition of chamomile cultivated in saline soil at Sinai. J. Appl. Sci. Res., 9(4): 3006-3021. Metabolism, 10: 33-50.
Opez-Bucio, J., and Nieto-Jacobo, M. I. (2000). Organic acid metabolism in plants: from adaptive physiology to transgenic varieties for cultivation in extreme soils. Plant Sci. 160, 1–13. doi: 10.1016/S0168-9452(00)00347-2.
Orav A, Raal A, Arak E, Müürisepp M, Kailas T. (2006). Composition of the essential oil of Artemisia absinthium L. of different geographical origin. Proc. Estonian Acad. Sci. Chem. 55 (3): 155-165. Google Scholar.
Phillips, L.D.J., (1971). Introduction to the Biochemistry and Physiology of Plant Growth Hormones. Mc. Graw-Hill Book Co.
Rai, V.K. (2002). Role of amino acids in plant responses to stresses. Biologia Plantarum, 45(4): 481-487.
Saffaa RE et. al. (2011). Effect of riboflavin, ascorbic acid and dry yeast on vegetative growth, essential oil pattern and antioxidant activity of geranium (Pelargonium graveolens L.). American-Eurasian J. Agric. & Environ. Sci.10,5: 781-786.
Sharopov FS, Sulaimonova VA, Setzer WN. Composition of the essential oil of Artemisia absinthium from Tajikistan. Rec Nat Prod 2012;6:127-34.
Sun, Y.L., Hong, S.K., (2011): Effects of citric acid as an important component of the responses to saline and alkaline stress in the halophyte Leymus chinensis (Trin.). Plant Growth Reg. 64, 129-139.
Talaat, IM. and AA. Youssef (2002) The role of the amino acids lysine and ornithine in growth and chemical constituents of Basil plants.Egyptian J. Appl. Sci. 17: 83–95.
Taraf, S.A., (1999). The response of vegetative growth and essential oil of lemongrass (Cymbopogon citrates Hort) to foliar application of ascorbic acid, nicotinamid and some micronutrients. Arab Univ. of Agric. Sci.; 7: 247-259.
Tucker, A.O., Maciarello, M.J. and Sturtz, G. (1993). The essential oils of Artemisia Powis Castle and its putative parents, A. absinthium and A. arborescens. Journal of Essential Oil Research, 5: 239-242.
Valles, J. & E.D. McArthur. 2001. Artemisia systematic and phylogeny: Cytogenetic and molecular insights. Pp 67-74 in Shrubland Ecosystem Genetics and Biodiversity.
Wahba H. E., et. al.( 2015) Growth and chemical composition of Urtica pilulifera L. plant as influenced by foliar application of some amino acids. 1. Mater. Environ. Sci. 6 (2) 499-506.
Waller, G.R. and E. Nawacki, (1978). Alkaloid Biology and Metabolism in Plants. Phanum, Press, New York, pp; 152.
Youssef, AA. and Talaat I M,. (2003). Physiological response of rosemary plants to some vitamins. Egypt.Pharm.J.;1:81-93.
Zafar, M.M., M.E. Hamdard and A. Hameed. (1990). Screening of Artemisia bsenthium for antimalarial effects on Plasmodium berghei in mice: a preliminary report. J. Ethnopharmacol., 30:223-226.


