Department of Biochemistry, Faculty of Science, King Abdulaziz
University Jeddah 21589, Saudi Arabia.
Corresponding author email: maaalghamdi3@kau.edu.sa
Article Publishing History
Received: 10/06/2021
Accepted After Revision: 24/09/2021
Traditional therapies for osteoporosis have undesirable side effects so that new therapeutic agents have been improved to use natural products and herbal extracts for therapeutic purposes. Therefore, the aim of this work was to evaluate the combined effects of Panax ginseng and Lepidum sativum (LS) seeds on biochemical changes and histological alterations of induced osteoporosis in female rats. Fifty adult female albino rats (200-320 g) were divided into five groups (n=10 rats/group). Group I, control group included non-ovariectomized untreated rats. Group II, ovariectomized (OVX) rats.
Group III, OVX rats administrated with KRG extract (100 mg\kg bw\day). Group IV, OVX rats administrated with LS seeds extract (20 mg\kg bw\day). Group V, OVX rats administrated with both extracts of KRG and LS seeds. In the present study, the ameliorative effects of Ginseng and LS seeds on bone remodeling against induced osteoporosis by ovariectomy were demonstrated by increasing serum levels of calcium (Ca), phosphorus (P), magnesium (Mg), vitamin D as well as decreasing serum alkaline phosphatase (ALP).
The histological study of cortical and trabecular femur bone revealed that the bone resorption and trabecular bone loss were increased in the OVX group. The administration of Ginseng, LS and their combination showed a marked improvement in trabecular bone and a significant reduction in bone loss. Ginseng and LS seed extracts and their combination possess an ameliorative effect against ovariectomy-induced osteoporosis which may be attributed to presence of wide range phytochemicals in the studied extracts.
Ginseng; Lepidium Sativum; Osteoporosis; Ovariectomy; Rats.
Al-Ghamdi M. A, Alshaikh A. T. Preventive Effects of Korean Red Ginseng and Lepidium sativum Seeds on Induced Osteoporosis in Female Rats. Biosc.Biotech.Res.Comm. 2021;14(3).
Al-Ghamdi M. A, Alshaikh A. T. Preventive Effects of Korean Red Ginseng and Lepidium sativum Seeds on Induced Osteoporosis in Female Rats. Biosc.Biotech.Res.Comm. 2021;14(3). Available from: <a href=”https://bit.ly/3kyltR2“>https://bit.ly/3kyltR2</a>
Copyright ©Al-Ghamdi Alshaikh This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Osteoporosis is a progressive skeletal disorder represented by reduced bone mineral density and microarchitectural bone deterioration (Qaseem et al., 2008), leading to increased bone fragility and fractures which is commonly occurred in the wrist, hip or spine (De Martinis et al., 2021). More than 200 million people have been affected by osteoporosis worldwide (Khan et al., 2019) and estimated at 21.4- 39.5% of Saudi population (Barzanji et al., 2013; Alwahhabi, 2015; Barake et al., 2021).
Several types of therapies have been designed to manage postmenopausal osteoporosis on using medications that inhibit bone resorption (e.g.estrogen, calcitonin and bisphosphonates)or stimulate bone formation (e.g. growth hormone, sodium fluoride, and parathyroid hormone) (Elkomy and Elsaid, 2015; Hagino et al., 2021). However, these medications have side effects (such as malignant reproductive tissues, cancers, gastrointestinal issues, and reduced skeletal strength) (Cao et al., 2014; Albert and Wood, 2021).
Accordingly, to reduce the side effect of currently used medicines, new therapeutic agents have been improved for efficacy therapy. There is a growing attitude in recent studies to use natural products and herbal extracts that possess active components (Khalil et al., 2008; Andargie et al., 2021).
Among the traditional herbs is Ginseng (Panax ginseng C. A. Meyer) which has been widely used in Eastern countries for thousands of years (Kim et al., 2015; Kim et al., 2016). Ginseng belongs to the Araliaceae family and has been widely grown in Korea (Siddiqi et al., 2013; Sharma and Goyal, 2015). Nowadays, all Ginseng products such as Korean Red Ginseng (KRG) have been used as alternative medicine in Europe as well as in Asian countries (Burden et al., 2021).
Ginsenosides are active components of Ginseng, which include protopanaxatriol, oleanane and protopanaxadiol (Bai et al., 2018). Ginseng has been reportedly a source of many effective substances including proteins, polysaccharides, polyacetylenes, and phenols compounds (Sharma and Goyal, 2015).
Ginseng presents different biological, medical and pharmaceutical functions such as anti-oxidant, anti-inflammatory, anti-allergic, anti-cancer, anti-fungal, anti-stress, memory enhancement; promoting liver function and protein synthesis, prevention of arteriosclerosis and hypertension (Sharma and Goyal, 2015; Karmazyn and Gan, 2021). In addition to its anti-osteoporosis activity by increasing bone formation in bone marrow cells and pre-osteoblast cells, amelioration of osteoporosis, as well as altering osteo-clastogenesis and bone resorption (Jung et al., 2021).
However, the mechanisms anti-osteoporotic effects of Ginseng are not clear. Lepidium sativum (LS) or Hab Al- Rashad “as locally termed in Saudi Arabia” belong to the Brassicaceae family and have widely grown in the Middle East (Elshal et al., 2013). LS seeds contain the most important phytochemicals and plant phytosterols have proved their potential anti-oxidant, anti-inflammatory and protection against certain diseases.
It also contains a flavonoid compound that protects the human body from oxidative stress (Yadav et al., 2011; Mahassni and Al-Reemi, 2013). LS has been used as a traditional herbal healer to diabetes control, hypertension, renal disease, cancer prevention, cardiovascular diseases protection and Phytotherap (Mahassni and Al-Reemi, 2013; Juma et al., 2007). LS seeds are particularly used in healing bone fracture (Juma et al., 2007; Abdul-Jabbar and Mohammed, 2021).
The aim of this work is to evaluate the combined effects of Ginseng and Lepidium sativum (LS) on biochemical changes and histological alterations of induced osteoporosis in female rats.
MATERIAL AND METHODS
Korean Red Ginseng (KRG) Panax ginseng was purchased from Korea Ginseng Corporation, Korea. KRG dose was prepared according to literature (Kim et al., 2015; Shin et al., 2021). The aqueous extract was given orally to experimental rats at a dose of 100 mg/kg bw/day for one month. Lepidium sativum (LS) seeds were obtained from a local herb shop in Jeddah, Saudi Arabia.
The aqueous extract of LS seeds was prepared according to the traditional Moroccan method described earlier (Eddouks et al., 2005). The obtained extract was administrated orally at a dose of 20 mg/kg bw/day for one month to different groups of rats. A total of 50 healthy female albino rats (200-320 g, 3 months old) were obtained from the Animal Experimental Unit at KFMRC (King Fahd Medical Research Center), King Abdulaziz University, Jeddah, Saudi Arabia with approved ethical permission.
The animals were fed with a standard diet manufactured by Grain Silos and Flour Mills Organization, Saudi Arabia. A bilateral ovariectomy was performed for 40 rats (called after as OVX rats) according to standardized procedures as previously described (Idris, 2012; Liu et al., 2015).
The animals were divided into 5 groups (n=10 rats/ group). Group I: untreated rats were fed on a standard diet and served as a negative control group. Group II (OVX group): OVX rats fed on a standard diet and served as a positive control group. Group III (Ginseng group): OVX rats administrated with the KRG extract (100 mg\kg bw\day). Group IV (LS group): OVX rats administrated with the extract of LS seeds (20 mg\kg bw\day). Group V (Ginseng+LS group): OVX rats administrated with a combination of KRG extract (100 mg\kg bw\day) and LS seeds extract (20 mg\kg bw\day).
Blood samples collected from rats were centrifuged at 3000 rpm for 15 min. The serum was separated and frozen at -20 °C until used for analysis. Serum phosphorus (P), calcium (Ca), magnesium (Mg) and alkaline phosphatase (ALP) were determined by commercial kits purchased from Siemens Healthcare Diagnostics Inc., Newark, USA. Serum Ca, P and Mg were measured as bone profile markers whereas ALP was measured as a bone turnover marker to monitor the bone metabolism. Serum levels of vitamin D were measured for the assessment of vitamin D sufficiency. [25(OH) D] kit was purchased from Roche Cobas Diagnostics International Ltd., Mannheim, Germany.
After blood collecting, rats were sacrificed, and the right and left femurs were harvested. Each femur was carefully cleaned, weight was recorded, and then stored in 10% neutral buffered formalin. The bone was decalcified with 5% aqueous nitric acid for one week. Once decalcified, the specimens were followed by routine histological processing and were embedded in paraffin. The paraffin sections (5 µm thick) were deparaffinized and stained by hematoxylin and eosin (H&E) for light microscopic examination as described in the literature (Mawhinney et al., 1984).
Data was analyzed by using SPSS statistics (version 15). All variables were double-checked for their values and outliers. Data were presented as mean ± SD. The parametric tests were used with normally distributed variables. The variance analysis was conducted to explore relationships between subgroups.
RESULTS AND DISCUSSION
Osteoporosis, represented by the rapid reduction and deterioration of the micro-architecture of bone tissue, causing an increased bone susceptibility to bone fracture has been a serious health problem worldwide (Genant et al., 2007; Hamed et al., 2021). Bone remodeling is described by the speed of two opposite processes; bone formation and bone deterioration. Any imbalance in bone remodeling may lead to bone mass loss (Hamed et al., 2021). With all their clinical and pharmacological advantages, the hormone replacement therapy has been widely applicable for osteoporosis. Recently, medicinal herbal extracts have been investigated for their therapeutic effects related to bone remodeling (Kurasawa, 2005; Guo et al., 2021).
Ginseng has been reported for its beneficial effects in the treatment of osteoporosis and increased bone formation although its effects and mechanism of action are not fully understood. Studies have shown that ginseng has an effective estrogenic activity on the OVX rats, due to its ability to stimulate the biosynthesis of estrogen and increase the quantity of receptor in the reproductive target tissues. The aglycone part of ginsenosides has shown to be similar to several steroids in structure, especially female hormones (Lee et al., 2021). This encouraged us to explore the beneficial effects of Ginseng, LS seed and their combination against OVX-induced osteoporosis in rats.
The results for the ameliorative effects of Ginseng, LS seed and their combined extract on some biochemical parameters of ovariectomized – induced bone loss were presentedin (Table 1). Serum Ca level was significantly increased with the treatment of Ginseng compared to control group and OVX group, (P< 0.01, Fig. 1 A). Level of serum P was significantly increased in Ginseng group (P< 0.001), LS group (P< 0.05), and their combination group (P< 0. 001) compared to the control group, Fig. (1 B). Furthermore, Ginseng administration increased the serum phosphorus concentration compared to the OVX group, P< 0.001.
Figure (1 C) showed that serum Mg was significantly decreased in the OVX group compared to the control group, at P<0.01. Comparing to the OVX group; Mg levels were significantly increased in Ginseng group (P<0.001), LS group (P<0.01) and Ginseng + LS group (P<0.01).
Due to the ovariectomy, vitamin D was showed a significant decreasing in the OVX group compared to control rats (P< 0.05); while the administration of herbal extracts (Ginseng, LS seed or their combination) caused a none-statistical improvement in its level, Fig. (1 D).
The serum ALP levels were significantly increased in ovariectomy rats (OVX group, P<0.01) and all treatment groups (Ginseng, LS and Ginseng + LS) compared with the control group (P<0.01, P<0.001 and P<0.001, respectively), Fig. (1 E). In preceding studies, the researchers demonstrated the ameliorative effect of Ginseng and LS seed against mass reduction and bone strength induced by ovariectomy in rats through assessing the serum levels of Ca, P, Mg, Vit D and ALP. These parameters and other bone turnover markers have been widely used as research tools to measure the effect of various drugs on bone remodel in (Bahlou, 2006; Nathawat et al., 2015; Gabr et al., 2017).
Table 1. Serum biochemical parameters among different rat groups
| Parameters | Control group | OVX
group |
Ginseng group | LS
group |
Ginseng + LS group |
| Serum Ca(nmol\L) | 2.46± 0.04 | 2.05 ± 0.02 | 2.59 ± 0.11 | 2.55 ± 0.06 | 2.55± 0.09 |
| Serum P (nmol\L) | 1.37± 0.18 | 1.54 ± 0.09 | 2.01 ± 0.32 | 1.65 ± 0.15 | 1.82± 0.13 |
| Serum Mg (nmol\L) | 1.02 ± 0.05 | 0.90 ± 0.02 | 1.05 ± 0.09 | 1.02 ± 0.05 | 1.02± 0.08 |
| Serum Vit D (nmol\L) | 106.17± 11.97 | 77.47±3.47 | 91.26 ± 21.31 | 85.95±12.50 | 82.17 ± 33.78 |
| Serum ALP (U\L) | 53.63 ± 16.39 | 128.70 ± 11.06 | 128.28 ± 39.40 | 145.25 ± 41.22 | 135.86± 54.90 |
Values are presented as (mean ± SD); Ca: calcium; P: phosphorus; Mg: magnesium; Vit D: vitamin D; ALP: alkaline phosphatase
Figure 1: Effect of ginseng, Lepidium sativum seed and their combination on biochemical parameters.
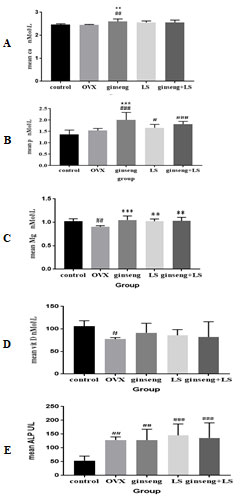
Values are mean ± SD.
#Significant P<0.05 versus control. *Significant P<0.05 versus OVX.
##Significant P<0.01 versus control. **Significant P<0.01 versus OVX.
###Significant P<0.001 versus control. ***Significant P<0.001 versus OVX.
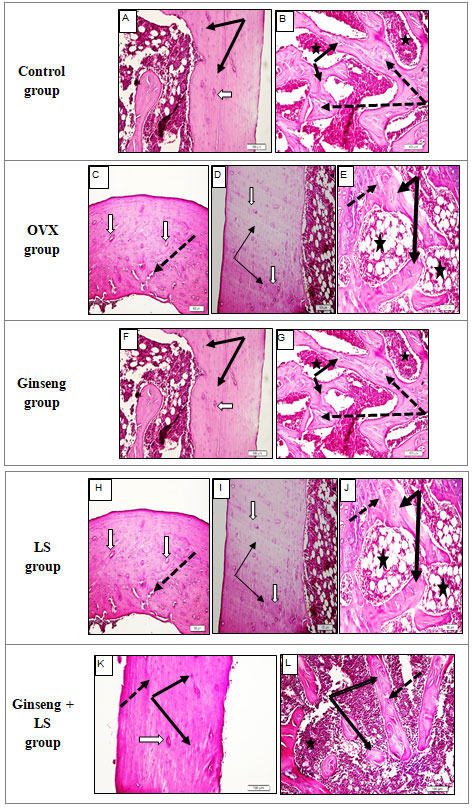
Figure 2: Histological sections of female rat femur of five groups, (H&E ×200).
The results of Table 1 showed the ameliorative effect of Ginseng, LS seed and their combined extracts on ovariectomized- induced bone loss. The results revealed that serum calcium level was decreased in OVX rats compared to untreated control rats. Serum calcium indirectly reflected the metabolism of bones. The administration of Ginseng and LS seed extract resulted a significantly increasing in the calcium level. These results are consistent with those that showed the serum Ca level was decreased in ovariectomized rats.
These findings are supported by the fact that ovarian hormone deficiency following ovariectomy, is marked by reduced intestinal calcium absorption and may contribute to the bone loss (Ouichou et al., 2008; Hassan et al., 2013). The results are also supported by the fact that, the level of calcitonin recorded a significant decrease in ovariectomized rats and may be attributed to the concentration of calcium level can be considered the principal stimulus for the secretion of calcitonin by C-cell. Low calcium in blood can stimulate for calcitonin secretion is diminished (Zhao et al., 2021).
The serum phosphate and magnesium were found significantly increased in the serum of the OVX rats who received the plant extracts compared to untreated OVX rats (Yu et al., 2021). The result of them showed gradually increase comparing with OVX, the highest increase shown in treatment with Ginseng. Serum vitamin D was found to be significantly decreased in OVX compared to control rats. The administration of Ginseng and LS seed extracts by the OVX rats caused none-significant increasing in its level compared to the OVX rats.
This increasing in the vitamin D level resulted from the stimulatory effect of the extract in vitamin D synthesis in the cell. The results are in line with the fact that herbal extracts can ameliorate the effect of OVX on biochemical parameters ( Zhao et al., 2011).
The serum of ALP, among the bone formation markers, increased in its level reflected bone turnover whereas its decreasing indicated to bone formation. In ovariectomized rats, ALP level was found to increase dramatically due to an increase in bone deterioration turnover. The results of this work also indicated a significant increasing in ALP level in the OVX rats compared to control rats, but the level of ALP tended to decrease after administration of herbal extracts. These suggest that herbal extracts possess compounds that stimulate bone density and inhibit bone resorption (Nagaki et al., 2021).
In the present study, histology of Group I (Control group): In the present study, the structure of female rat femur was similar to those described in the literatures. The cortical (compact) bone of femur shaft was showed smooth outer surface (Thick black arrows) and characterized by normal Haversian system architecture (white arrows) (Fig. 2A). Trabecular (spongy) bones showed normal branching and anastomosing trabecula (black arrows) separated by the bone marrow spaces (stars) (Fig. 2B).
Histology of Group II (OVX group): In this study, the femur bone of OVX rats showed random irregular cracking of femur cortical (compact) bone (black arrows), widening of Haversian canals (white arrow), areas of bone rarefication (star), lines of calcification (dotted arrows), and in severe cases areas of bone fracture and loss (Fig. 2C & D). Trabecular (spongy) bone showed wide bone marrow cavities (stars) and thin cracked trabeculae (white arrows) a thin cracked trabecula (Fig. 2E).
Histology of Group III (Ginseng group): Administration of Ginseng to OVX rats provided marked protection against OVX- induced boney changes in the rat femur. Cortical (compact) bone showed a normal appearance with absence of any bone cracking that seen in untreated rats.
Figure (2 F) showed cortical bone with narrow Haversian canals (white arrow) and normal osteocyte lacunae (black arrows). Although trabecular (spongy) bone looked to be thicker, it showed central regions of incomplete ossification. Figure (2 G) showed normal wide trabeculae (black arrows) with bluish areas of calcification (dotted arrows) and bone marrow cavities (stars).
Histology of Group IV (LS group): Administration of LS seeds to OVX rats provided more protection against OVX- induced bone changes in rat femur compared to Ginseng. Compact cortical bone showed a well-organized Haversian system with absence of any bone cracking that seen in untreated rats. Figure (2H&I) showed narrow Haversian canals (white arrow) and normal osteocyte lacunae (black arrows).
Fine cracking was still observed (dotted arrow). Spongy trabecular bone looked thicker and more ossified than Ginseng group. Figure (2 J) showed cortical bone with normal wide trabeculae (black arrows) with bluish areas of calcification (dotted arrows) and bone marrow cavities (stars). Histology of Group V (Ginseng + LS group): The administration of a combination of Ginseng and LS seed to OVX rats provided more protection against OVX- induced boney changes in rat femur compared to Ginseng or LS seeds alone.
Compact cortical bone showed a well-organized Haversian system with absence of any bone cracking. Figure (2 K) showed cortical bone with narrow Haversian canals (white arrow) and normal osteocyte lacunae (black arrows). Spongy trabecular bone looked thicker and more ossified compared to previous groups. Spongy bone showed in Fig. (2 L) normal wide trabeculae (black arrows) with areas of complete calcification (dotted arrows) and bone marrow cavities (stars).
The histological study of both cortical and trabecular femur bone revealed that the bone resorption and trabecular bone loss were increased in the OVX group. The administration of Ginseng, LS and their combined extracts showed a marked improvement in trabecular bone and a significant reduction in bone loss. These results were agreed with the biochemical investigations. Microscopically, the control rats revealed no histopathological alteration in the cortical and trabecular femur bone which is in agreement with Vallet et al., (2021) who demonstrated that the left femur of the normal control rats did not show any histopathological changes in the cortical bone with osteoblasts proliferation as well as normal bony trabeculae.
The present study suggested an increase in bone loss which was supported by osteoporotic changes found through histological examination in OVX rats. The histopathological findings in Ginseng group are well supported by the findings of Leiu et al. (2015) as Ginseng stimulates the process that increases bone density by inhibiting bone resorption. The positive effect on bone density of the Ginseng extract is probably due to increasing the protein biosynthesis and nucleic acid (Han et al., 2021).
Previous histopathological studies supported a potential protective role of LS against induced osteoporosis in male and female rats. El-Zawahry et al., (2017), and Abdul-Jabbar et al., (2021) reported that the sections of rats treated with LS extract revealed a marked improvement compared with osteoporotic rats, and the cortical bone thickness was very similar to the normal control group. In both studies, the bony trabeculae were partially recovered near the normal structure and appeared to be more persistent with the smaller bone marrow spaces.
CONCLUSION
In conclusion, the present results indicated that Ginseng and LS seed extracts and their combined extracts reduced the bone loss in ovariectomized rats, possibly through inhibiting bone resorption process. Phytochemical constituents of extracts such as flavonoids, saponins, and phenolics could probably be responsible for the anti-osteopathic activities of plants. Therefore, the extract from Ginseng and LS has the potential to be used in the development of clinical anti-osteoporotic agents.
ACKNOWLEDGEMENTS
The authors acknowledge King Abdulaziz University, Jeddah, for scientific research.
Disclosure Statement: The authors declare no conflict of interest.
REFERENCES
Abdul-Jabbar, Z.S. and Mohammed, J.A., (2021). Study of Histological Changes in the Bones of Front and Hind Limbs of White Rat Treated with Ibuprofen and Lepidium sativum. Revista Geintec-Gestao Inovacao E Tecnologias, 11(2), Pp.1988-2003.
Albert, S.G. and Wood, E., (2021). Meta-analysis of clinical fracture risk reduction of anti-osteoporosis drugs: direct and indirect comparisons and meta-regressions. Endocrine Practice.
Alwahhabi, B.K., (2015). Osteoporosis in Saudi Arabia: Are we doing enough?. Saudi medical journal, 36(10), p.1149.
Andargie, M., Vinas, M., Rathgeb, A., Möller, E. and Karlovsky, P., (2021). Lignans of Sesame (Sesamum indicum L.): a comprehensive review. Molecules, 26(4), p.883.
Bahlous, A., et al., (2006). Biochemical markers of bone remodeling: recent data of their applications in managing postmenopausal osteoporosis. La Tunisie medicale, 84(11): 751-757.
Bai, L., Gao, J., Wei, F., Zhao, J., Wang, D. and Wei, J., (2018). Therapeutic potential of ginsenosides as an adjuvant treatment for diabetes. Frontiers in pharmacology, 9, p.423.
Barake, M., El Eid, R., Ajjour, S., Chakhtoura, M., Meho, L., Mahmoud, T., Atieh, J., Sibai, A.M. and Fuleihan, G.E.H., (2021). Osteoporotic hip and vertebral fractures in the Arab region: a systematic review. Osteoporosis International, pp.1-17.
Barzanji, A.T., Alamri, F.A. and Mohamed, A.G., (2013). Osteoporosis: a study of knowledge, attitude and practice among adults in Riyadh, Saudi Arabia. Journal of community health, 38(6), pp.1098-1105.
bin Abdullah Juma, A.B.H., (2007). The effects of Lepidium sativum seeds on fracture-induced healing in rabbits. Medscape general medicine, 9(2), p.23.
Burden, A.M., Tanaka, Y., Xu, L., Ha, Y.C., McCloskey, E., Cummings, S.R. and Glüer, C.C., (2021). Osteoporosis case ascertainment strategies in European and Asian countries: a comparative review. Osteoporosis International, 32(5), pp.817-829.
Cao, P.C., Xiao, W.X., Yan, Y.B., Zhao, X., Liu, S., Feng, J., Zhang, W., Wang, J., Feng, Y.F. and Lei, W., (2014). Preventive effect of crocin on osteoporosis in an ovariectomized rat model. Evidence-Based Complementary and Alternative Medicine.
De Martinis, M., Sirufo, M.M. and Ginaldi, L., (2020). Osteoporosis: Current and emerging therapies targeted to immunological checkpoints. Current medicinal chemistry, 27(37), pp.6356-6372.
Eddouks, M., Maghrani, M., Zeggwagh, N.A. and Michel, J.B., (2005). Study of the hypoglycaemic activity of Lepidium sativum L. aqueous extract in normal and diabetic rats. Journal of Ethnopharmacology, 97(2), pp.391-395.
Elkomy, M.M. and Elsaid, F.G., (2015). Anti-osteoporotic effect of medical herbs and calcium supplementation on ovariectomized rats. The Journal of Basic & Applied Zoology, 72, pp.81-88.
Elshal, M.F., Almalki, A.L., Hussein, H.K. and Khan, J.A., (2013). Synergistic antiosteoporotic effect of Lepidium sativum and alendronate in glucocorticoid-induced osteoporosis in Wistar rats. African Journal of Traditional, Complementary and Alternative Medicines, 10(5), pp.267-273.
Gabr, G.A., et al., (2017). The Potential Protective Effects of Vigna radiata and Lepidium sativum against Bone Loss Induced by Prednisolone Acetate in Male and Female Rats. Indo American Journal of Pharmaceutical Sciences, 4(5): 1085-1094.
Genant, H.K., Delmas, P.D., Chen, P., Jiang, Y., Eriksen, E.F., Dalsky, G.P., Marcus, R. and San Martin, J., (2007). Severity of vertebral fracture reflects deterioration of bone microarchitecture. Osteoporosis international, 18(1), pp.69-76.
Guo, Y., Liu, Y., Shi, C., Wu, T., Cui, Y., Wang, S., Liu, P., Feng, X., He, Y. and Fu, D., (2021). Remote-controllable bone-targeted delivery of estradiol for the treatment of ovariectomy-induced osteoporosis in rats. Journal of nanobiotechnology, 19(1), pp.1-18.
H El-Zawahry, B., M El-Shawwa, M. and F Hikal, S., (2017). Effect of Lepidium sativum on blood levels of apelin and some metabolic and oxidative parameters in obese male rats. Al-Azhar Medical Journal, 46(3), pp.723-738.
Hagino, H., Tanaka, K., Silverman, S., McClung, M., Gandra, S.R., Charokopou, M., Adachi, K., Johnson, B. and Stollenwerk, B., (2021). Cost effectiveness of romosozumab versus teriparatide for severe postmenopausal osteoporosis in Japan. Osteoporosis International, pp.1-11.
Hamed, K., Roaldsen, K.S. and Halvarsson, A., (2021). Fear of falling serves as protection and signifies potential danger”: a qualitative study to conceptualise the phrase fear of falling in women with osteoporosis. Osteoporosis international, pp.1-8.
Han, M.J., Shin, J.E., Park, S.J. and Choung, S.Y., (2021). Synergetic effect of soluble whey protein hydrolysate and Panax ginseng berry extract on muscle atrophy in hindlimb-immobilized C57BL/6 mice. Journal of Ginseng Research.
Hassan, H.A., A.M.E. Wakf, and N.E.E. Gharib, (2013). Role of phytoestrogenic oils in alleviating osteoporosis associated with ovariectomy in rats. Cytotechnology, 65(4): 609-619.
Idris, A.I., (2012). Ovariectomy/orchidectomy in rodents. In Bone research protocols (pp. 545-551). Humana Press, Totowa, NJ.
Inagaki, Y., Kido, J.I., Nishikawa, Y., Kido, R., Sakamoto, E., Bando, M., Naruishi, K., Nagata, T. and Yumoto, H., (2021). Gan-Lu-Yin (Kanroin), Traditional Chinese Herbal Extracts, Reduces Osteoclast Formation In Vitro and Prevents Alveolar Bone Resorption in Rat Experimental Periodontitis. Journal of clinical medicine, 10(3), p.386.
Jung, B.H., Ma, D.S. and Yoo, K.Y., (2021). Inhibition of Alveolar Bone Destruction by Red Ginseng Extract in an Experimental Animal Periodontitis Model.
Karmazyn, M. and Gan, X.T., (2021). Chemical components of ginseng, their biotransformation products and their potential as treatment of hypertension. Molecular and Cellular Biochemistry, 476(1), pp.333-347.
Khalil, W.K., Ahmed, K.A., Park, M.H., Kim, Y.T., Park, H.H. and Abdel-Wahhab, M.A., (2008). The inhibitory effects of garlic and Panax ginseng extract standardized with ginsenoside Rg3 on the genotoxicity, biochemical, and histological changes induced by ethylenediaminetetraacetic acid in male rats. Archives of toxicology, 82(3), pp.183-195.
Khan, J.A., McGuigan, F.E., Akesson, K.E., Ahmed, Y.M., Abdu, F., Rajab, H. and Albaik, M., (2019). Osteoporosis knowledge and awareness among university students in Saudi Arabia. Archives of osteoporosis, 14(1), p.8.
Kim, H., Hong, M.K., Choi, H., Moon, H.S. and Lee, H.J., (2015). Chemopreventive effects of korean red ginseng extract on rat hepatocarcinogenesis. Journal of Cancer, 6(1), p.1.
Kim, J., Lee, H., Kang, K.S., Chun, K.H. and Hwang, G.S., (2015). Protective effect of Korean Red Ginseng against glucocorticoid-induced osteoporosis in vitro and in vivo. Journal of ginseng research, 39(1), pp.46-53.
Kim, S.J., Shin, J.Y. and Ko, S.K., (2016). Changes in the contents of prosapogenin in Red ginseng (Panax ginseng) depending on the extracting conditions. Journal of ginseng research, 40(1), pp.86-89.
Kurasawa, K., (2005). Bone quality and biochemical markers. Clinical calcium, 15(6), pp.959-964
Lee, Y.H., Choi, H.J., Kim, J.Y., Kim, J.E., Lee, J.H., Cho, S.H., Yun, M.Y., An, S., Song, G.Y. and Bae, S., (2021). Ginsenoside Rg4 enhances the inductive effects of human dermal papilla spheres on hair growth via the AKT/GSK-3β/β-catenin signaling pathway.
Liu, X.L., Li, C.L., Lu, W.W., Cai, W.X. and Zheng, L.W., (2015). Skeletal site‐specific response to ovariectomy in a rat model: change in bone density and microarchitecture. Clinical oral implants research, 26(4), pp.392-398.
Mahassni, S.H. and Al-Reemi, R.M., (2013). Apoptosis and necrosis of human breast cancer cells by an aqueous extract of garden cress (Lepidium sativum) seeds. Saudi journal of biological sciences, 20(2), pp.131-139.
Mawhinney, W.H., Richardson, E. and Malcolm, A.J., (1984). Control of rapid nitric acid decalcification. Journal of clinical pathology, 37(12), p.1409.
Nathawat, R., P. Mishra, and V. Patni, (2015). Identification and Qualitative Analysis of β-Sitosterol and Some Phytoestrogens in. Vivo.
Ouichou, A. and A. El Hessni, (2008). Does flaxseed uptake reverse induced-bone loss in ovariectomized rats? International Journal of Osteoporosis and Metabolic Disorders, 1(1): 24-30.
Qaseem, A., Snow, V., Shekelle, P., Hopkins Jr, R., Forciea, M.A. and Owens, D.K., (2008). Pharmacologic treatment of low bone density or osteoporosis to prevent fractures: a clinical practice guideline from the American College of Physicians. Annals of internal medicine, 149(6), pp.404-415.
Sharma, J. and Goyal, P.K., (2015). Chemoprevention of chemical-induced skin cancer by Panax ginseng root extract. Journal of ginseng research, 39(3), pp.265-273.
Sharma, J. and Goyal, P.K., (2015). Chemoprevention of chemical-induced skin cancer by Panax ginseng root extract. Journal of ginseng research, 39(3), pp.265-273.
Shin, S.J., Nam, Y., Park, Y.H., Kim, M.J., Lee, E., Jeon, S.G., Bae, B.S., Seo, J., Shim, S.L., Kim, J.S. and Han, C.K., (2021). Therapeutic effects of non-saponin fraction with rich polysaccharide from Korean red ginseng on aging and Alzheimer’s disease. Free Radical Biology and Medicine, 164, pp.233-248.
Siddiqi, M.H., Siddiqi, M.Z., Ahn, S., Kang, S., Kim, Y.J., Sathishkumar, N., Yang, D.U. and Yang, D.C., (2013). Ginseng saponins and the treatment of osteoporosis: mini literature review. Journal of ginseng research, 37(3), p.261.
Vallet, M., Sophocleous, A., Törnqvist, A.E., Azfer, A., van’t Hof, R., Albagha, O.M. and Ralston, S.H., (2021). Targeted Inactivation of Rin3 Increases Trabecular Bone Mass by Reducing Bone Resorption and Favouring Bone Formation. Calcified tissue international, pp.1-11.
Yadav, Y.C., Jain, A., Srivastava, D.N. and Jain, A., (2011). Fracture healing activity of ethanolic extract of Lepidium sativum L. seeds in internally fixed rats’ femoral osteotomy model. International Journal of Pharmacy and Pharmaceutical Sciences, 3(2), pp.193-197.
Yu, S., Chun, E., Ji, Y., Lee, Y.J. and Jin, M., (2021). Effects of red ginseng on gut, microbiota, and brain in a mouse model of post-infectious irritable bowel syndrome. Journal of Ginseng Research.
Zhao, X., Wu, Z.X., Zhang, Y., Yan, Y.B., He, Q., Cao, P.C. and Lei, W., (2011). Anti-osteoporosis activity of Cibotium barometz extract on ovariectomy-induced bone loss in rats. Journal of ethnopharmacology, 137(3), pp.1083-1088.
Zhao, Y., Wang, Y., Zhang, M., Gao, Y. and Yan, Z., (2021). Protective Effects of Ginsenosides (20R)‐Rg3 on H2O2‐Induced Myocardial Cell Injury by Activating Keap‐1/Nrf2/HO‐1 Signaling Pathway. Chemistry & Biodiversity, 18(4), p.e2001007.


