Department of Environmental Health, Tehran Medical Branch, Islamic Azad University, Tehran, Iran
Corresponding author Email: gitikashani11@gmail.com
Article Publishing History
Received: 12/04/2017
Accepted After Revision: 28/06/2017
Phenol is a known prejudicial exotoxin and finds place in the environment due to intense activity of petrol industry. The aim of this applied-analytical study is to investigate phenol removal from urban drinking water using batch reactor with using powdered eggshell. Various operating variables are tested for their effects on phenol removal; these include pH, contact time, adsorbent doses, initial phenol concentration, reaction kinetics, and powdered eggshell characteristics. Sample of urban drinking water is prepared containing 5-15 mg/L phenol. Powdered eggshell is prepared in a laboratory oven at 105˚C for 12 h. The phenol-containing water enters batch reactor and phenol removal efficiency is studied in different cases of the variables pH (3-11), contact time (0-120 min), and adsorbent doses (3-5 gr/dl). Characteristics of the eggshell powder show that the average diameter size of eggshell powder is 2 μm. The main component of eggshell powder is calcium carbonate (CaCO3). The best conditions for phenol removal are obtained to be pH 3, contact time 80 min, and adsorbent dose 4 gr, and phenol concentration 5 mg/L. The adsorption of phenol on powdered eggshell is obtained from Langmuir isotherm.
Adsorption, Isotherm, Phenol, Powdered Eggshell, Urbane Drinking Water
Kashi G. On the Analysis of Phenol Removal from Drinking Water by Batch Reactor Using Powdered Eggshell. Biosc.Biotech.Res.Comm. 2017;10(2).
Kashi G. On the Analysis of Phenol Removal from Drinking Water by Batch Reactor Using Powdered Eggshell. Biosc.Biotech.Res.Comm. 2017;10(2). Available from: https://bit.ly/30tfxMv
Introduction
Chemically, phenol is acidic because of the influence of the aromatic ring. Phenol is an organic pollutant that finds in the industrial wastewater. Phenol has been measured in effluents (up to 53 ppm), ambient water (1.5 – >100 ppb), groundwater (1.9 – >10 ppb), rain (0.075-1.2 ppb), sediment (>10 ppb), and ambient air (0.03-44 ppb) (Al-Khalid, 2012 and American Public Health Association, 2015). The increase in phenol levels in groundwater has been mainly attributed to profligate utilization of phenolic pesticides, municipal and industrial sewage treatment systems such as oil refinery, and decaying organic matter or producing toxic poly chlorinated phenols during water chlorination process (Bazrafshan, 2016).
Phenol and its derivatives like Dinitrophenol and pentachlorophenol are very toxic substances with a toxicology rating of 4 (Cabaj, 2016). Phenol is famous due to its high toxicity for human life and aquatic life (Chand Meena, 2015). Phenol-laced water may lead to liver, kidney, and respiratory disorders in humans (Dakhil, 2013). Phenol-contaminated water above the permissible drinking water limit leads to phenolic tastes and doors problem, may be a cause of lethal to humans, and the drinking water standard have been promulgated as >1 μg/l at point of chlorination (Daraei, 2013). Processes such as activated carbon adsorption, ion exchange, liquid-liquid extraction, and chemical oxidation methods are effective for removing phenol and phenolic compounds from water (Dehghani, 2016).
The main disadvantage of ion exchange technique is low selectivity against anions. The main disadvantage of these techniques is high capital and operational cost (Fan, 2011). The advantages of adsorption technology are applicable in batch and continue arrangements, easier accessibility, designing flexibility, economical, and retain very effectiveness on high concentration and low of phenolic industrial wastewater. Many natural adsorbents are experimented as removal phenol agents including sawdust Yeast (Liao, 2010), and almond shell (Hsieh, 2008, Loganathan, 2013, Kashi, 2015 and Giti, 2015). The potential of annual egg, as biosorption of organic materials, production in 2011 is estimated to be 5.2 million tons in Iran country. The aim of this applied-analytical study is to investigate feasibility of phenol removal from drinking water using the batch reactor with using eggshell powder. The variables under study involve pH, contact time, adsorbent doses, initial phenol concentration, reaction kinetics, and eggshell powder characteristics.
Material and Methods
Phenol-contaminated water samples used for adsorption experiments are obtained from urbane distribution system situated the site of a laboratory in Islamic Azad University Tehran Medical Sciences Branch, in Tehran city. The samples are tested for the main physicochemical characteristics. The mean values of these water characteristics are presented in Table 1. All the reagents used are of analytical grade. A solution of 5, 10, and 15 mg/L phenol is prepared by dissolving appropriate amount of phenol (Merck, Germany) in deionized water.
| Table 1: The Main Physicochemical Characteristics of Phenol-Contaminated Urbane Water | ||
| Parameter | Unit | Value |
| Calcium | mg/L as CaCO3 | 162 |
| Dissolved oxygen | mg/L | 8.05 |
| Nitrate | mg/L | 9.5 |
| ORP | mV | 272 |
| pH | – | 7.19 |
| Sulphate | mg/L | 93.8 |
| Temperature | ˚C | 20 |
| Total Alkalinity | mg/L as CaCO3 | 122 |
After collecting chicken eggshells from local markets of Tehran city, removing waste matters such as colour and fat, boiling in deionized water for 30 minutes, and washing with deionized water, eggshell powder is prepared by heating the collected eggshells in a hot air oven (Dena, Iran) at 105˚C for 12 h, maybe, temperature higher than 105˚C leads to decreasing phenol removal due to damaging calcium carbonate structure, while temperature below 105˚C leads to developing bad taste and odour in finished water. Heat pre-treatment removes the organic matter, cause of taste and colour problems in water. After heating, the eggshells are crushed by a laboratory electrical crusher (AIKA, Germany) for 20-30 second and are sieved several times to get a uniform fraction of eggshell in a specific size (60-100 mesh/ 0.25-0.104 mm), according to ASTM standard (Mijan, 2014). The powdered eggshell is stored in the desiccator after pre-treating with a solution of sodium hypo chloride (NaOCl) (Merck, Germany), to eliminate the dust particles (Mourão, 2011).
Scanning Electron Microscopy (SEM) image (Philips, XL 30, Holand) is prepared from powdered eggshell. X-ray diffraction patterns are measured using RINT 2000 (Rigaku Instrument Corp.) with Cu Ká radiation for confirming the structure and mineral composition of powdered eggshells (Philips, Xpert, Holand). Powdered eggshell composition is obtained by energy dispersive X-ray (EDX) analysis. The surface area of powdered eggshell is analysed through nitrogen adsorption measurements at 77 K using Micromeritics Gemini 2370 equipment. The zeta potential is analysed with a Nano Zetasizer (Philips, Holand).
The batch reactor is a 250 ml glass rectangular container (10×6×6 cm). To evaluate the effect of adsorption, on the phenol removal process, samples undergo with different pH (ca. 3-11), different times (0-120 min), different concentrations of F (5-15 mg/L), and adsorbent dosage (3-5 gr/dl). The sample number is obtained 192. Magnetic stirrer (AIKA, Germany) is used for homogeneous mixing of water samples (120 rpm). For each test, 200 ml of sample water is poured into the reactor. All tests are performed at laboratory temperature (20˚C). Chloride acid and sodium hydroxide solutions (0.1 N) (Merck, Germany) are used for pH adjustment.
All tests were performed in triplicate, and the mean data values are reported. The water samples are tested for phenol after adsorption process by using spectrophotometer (Hach DR5000, America) at a wavelength of 500 nm. Phenol is determined by standard method 5530D (Ngah, 2011). The percentage phenol removal is calculated according to the following equation (1):
![]()
Where the percentage of phenol removal (R, percentage) and the phenol value before and after treatment (Ct0 and Ct, mg/L) expressed.
The removal phenol capacity of the regenerated powdered eggshell is calculated according to the following equation (2)
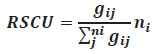
Where the removal phenol capacity of the regenerated powdered eggshell (DCFC, mg/g) and the phenol concentration before and after treatment (S0 and St, mg/L) expressed. To optimize runs and data analysis, based on Taguchi model, is used to study vestiges of the selected variables and minimize the number of experimental runs (Tzvetkova, 2016).
Results and Discussion
The reduction of phenol from urbane drinking water is investigated in an adsorption reactor with filler particles made of charring chicken eggshells in batch mode. Several operational variables are examined for the effects on process reduction efficiency. The following results are obtained from the experiments.
Figure 1 illustrates SEM images of the eggshell powder. As is observed that, the average diameter size of eggshell powder is 2 μm. Figure 2 illustrates X-ray dispersive (XRD) analysis of the eggshell powder. As is found in figure 2, the analysed eggshell powder is composed of the elements including calcium (Ca) and phosphor (P) as the main and partial elements respectively. Figure 3 illustrates EDX analysis of the eggshell powder. As is found in figure 3, the analysed eggshell powder is composed of elements including calcium (Ca), oxygen (O), magnesium (Mg), carbon (C), and so on.
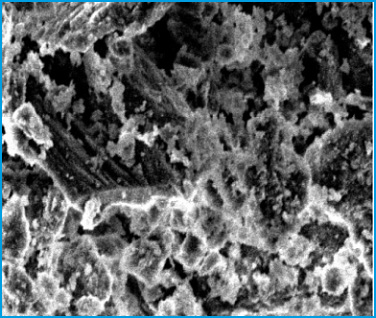 |
Figure 1: SEM image of eggshell powder (a) before treatment, (b) after treatment |
 |
Figure 1b: |
 |
Figure 2: XRD analysis of eggshell powder |
The characteristics of eggshell powder are showed in Table 2. Specific surface area, BET measurements has also been performed, and the highest BET is obtained for eggshell powder 7.43 m2/g. The spectrum of the analysed eggshell powder is adapted to 2370 standards. Therefore these peaks confirmed that the main component of eggshell powder is calcium carbonate (CaCO3). The calcium to carbon (Ca/C) ratio of eggshell powder is 2.9. SEM analysis is another helpful instrument for the analysis of the surface morphology of an adsorbent. The agglomerate, non-adhesive, porous and irregular surface structure of the adsorbent can be distinctly shown in SEM image indicates in figure 1a. Furthermore, the pores on the surface of the adsorbent are regular, adhesive, and very heterogeneous as indicates in figure 1b. The heterogeneous pores leads to producing a lager exposed surface area for the adsorption of phenol. In other hand the heterogeneous pores leads to producing a large affinity for the adsorption of phenol.
| Table 2: Characterization of Eggshell Particles | ||||
| Particle | pHZPC | Density
(g/cm3) |
Specific Surface Area, BET (m2/g) | Diameter
(μm) |
| Eggshell | 8.2 | 1.148 | 7.43 | 2 |
Mijan et al. (2014) report that the eggshell powder particles are irregular in shape and their surface is rough. The diameter of pore is symptom of the anticipated adsorption of phenol molecule onto the surface of the adsorbent. It distinctly indicates the porous surface formation which supports the adsorbent with enhanced surface area and very adsorption capacity. The SEM analysis shows that the removal of phenol affects the orientation of the eggshells powder. The treated sample illustrates regular, adhesive appearance causing higher adsorption of phenol. Bhaumik et al report that the particle size of eggshell powder is 150-350 μm. Zulfikar et al. (2013) report that BET of eggshell powder is 3.23 m2/g. Gaonkar and Chakraborty report that composition of eggshell consists of calcium carbonate (91%). Agarwal and Gupta report that the most compound in the eggshell powder is calcium carbonate. Functional groups of eggshells are diagnosed by infrared analysis. The peaks at about 710, 875, 1420, 1807, and 2520 cm-1 adopt with those of pure CaCO3. The XRD pattern displays six characteristic peaks at 2è=34.5°, 42.2°, 46°, 50.9°, 56°, and 57.8° in crystalline structure and corroborates the presence of calcite (CaCO3) in the eggshell (Zulfikar, 2013).
Adsorption experiments are carried out an initial pH values in the range of 3 to 11 at the experimental conditions such as contact time (80 min), phenol concentration (5 mg/L) and amount of eggshell powder adsorbent (4 g). The mean phenol removal decreases from 100% to 87% when the pH increased from 3 to 7 (Figure 4). Phenol removal in the adsorption reactor is mainly influenced by the water pH. The pH has a significant effect on phenol reduction due to surface charge of the absorbent, with the highest reduction obtained at pH 3. It is related to phenol ionization, and adsorbent surface. Phenol is a weak acid and produces phenoxide ions. Due to more exchangeable ion rate, and better adsorbent, the optimum pH for reaching to phenol standard (>1 μg/L) is pH 3. The phenol removal of in alkaline condition diminishes which can be due to increasing turbidity.
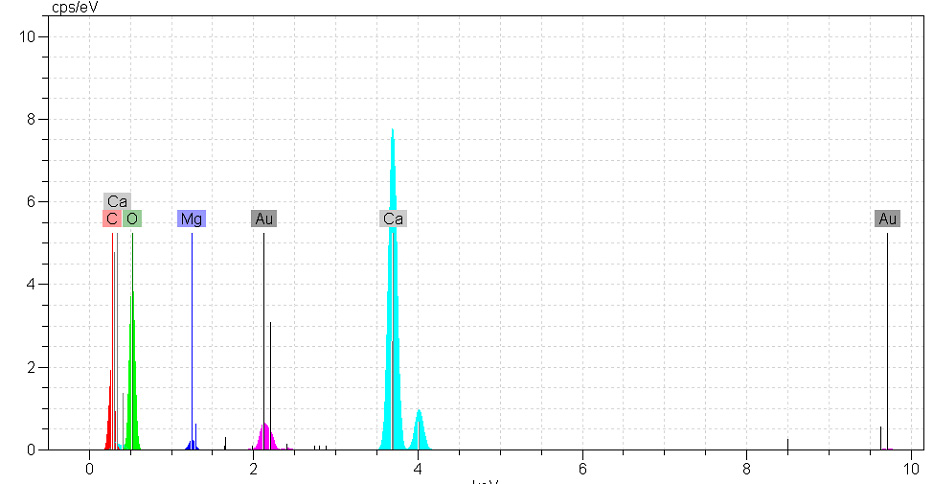 |
Figure 3: EDX analysis of eggshell powder |
This is in agreement with Daraei et al. (Daraei, 2013), who report that the fastest removal rate occurs at pH equal to 3.5. The pH of point of zero charge (pHZPC) is an important parameter in phenol sorption, for it is the pH in which the sorbent has a neutral charge. When pH is increased above the pHZPC, phenol sorption decreases due to electrostatic repulsion between the surface and phenoxide ions as well as a result of competition with hydroxides in solution.
Adsorption experiments are carried out an initial adsorbent dosage in the range of 3 to 5 g at the experimental conditions such as contact time (80 min), phenol concentrations (5 mg/L) and pH value (3). The mean phenol removal increases from 57% to 100% when the adsorbent dosage increases from 3 to 4 (Figure 5). Enhancing the adsorbent dosage increases the percentage of phenol that is attributed to enhancing of sportive surface area, and sorption capacity since more active adsorption sites, more exchangeable sites, and more proper porosity are available. The optimum dosage of eggshell powder adsorbent is 4 g. It is seen that 4 g is a better adsorbent dosage than 3 g. This phenomenon can be due to exposure of the active sites of adsorbent which allow phenoxide ions of water to have a direct contact with eggshell powder, therefor increasing adsorption capacity. Increasing the adsorbent dosage also leads to decreasing surface area between eggshell powder adsorbent and phenol adsorbate due to the formation of aggregates. Any change in the adsorbent dosage will increase pH. Zulfikar et al (2013) reported that the optimum dosage of eggshell is 5.0 g. At constant pH, activity of phenoxide is directly proportional with the concentration of Ca+2 and thereby increasing the adsorption of phenol over eggshell surface significantly (Zulfikar, 2013).
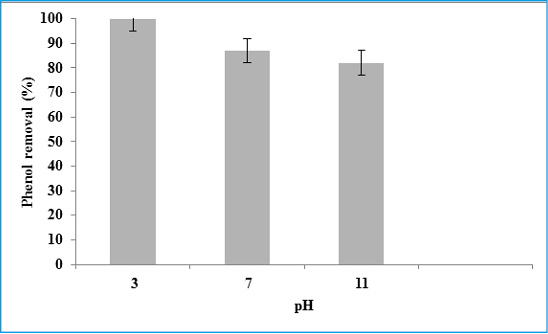 |
Figure 4: The effect of water pH on the phenol removal in the batch adsorption reactor |
Adsorption experiments are carried out as a function of the time levels in the range of 0 to 120 min at the experimental conditions such as amount of eggshell powder adsorbent (4 g/dL), phenol concentration (5 mg/L) and pH value (3). The efficiency of phenol removal increases as the contact time increases. The mean phenol removal increases from 79% to 100% when the contact time increases from 40 to 80 min (Figure 6). The efficiency of phenol removal initially increases as the contact time increases, but then gradually approaches a more or less constant value, indicating obtainment of equilibrium. As there is no enhance in phenol removal efficiency between 80 and 120 minute, equilibrium time of 80 minute is selected for eggshell powder adsorbents.
 |
Figure 5: The effect of eggshell powder adsorbent dosage on the phenol removal in the batch adsorption reactor |
These variations can be due to the fact that initially, all absorbent sites are empty and active, the solute concentration gradient is high and attraction of active functional groups towards phenol which results in stronger surface binding is high. Then, the phenol adsorption rate on to eggshell powder adsorbent noticeably diminishes due to decreasing in absorbent sites. This phenomenon indicates a monolayer of phenoxide ions on the external surface and pores of eggshell powder and pore diffusion on to the internal surface eggshell powder through the film due to continues turbulent maintains during the experiments. During this period residual phenol fluctuates from a maximum value of 1.05 mg/L at 40 min contact time to a minimum value of 0.0 mg/L for a contact time of 80 min. The highest removal capacity of eggshell powder (4 mg/g) is obtained for duration of 80 min, respectively. An equilibrium time of 60 minute is selected for chitosan-H2SO4 beads by Ngah et al., (2011). An equilibrium time of 120 minute is selected for carbonate hydroxyapatite are extracted from eggshell waste by Liao (Liao, 2010). If the phenoxide ion is more than absorbent sites, adsorption decreases due to saturating absorbent sites at a constant concentration.
Adsorption experiments are carried out an initial phenol concentration in the range of 5 to 15 mg/L at the experimental conditions such as contact time (40-120), amount of eggshell powder adsorbent (4 g/dL) and pH value (3). The mean phenol removal decreases from 100% to 81% when the initial phenol concentration increases from 5 to 15 mg/L for duration 80 minute (Figure 7). The phenol removal as a function of contact time is proportional to the phenol ions in water. At higher concentration, due to saturating active adsorption sites by phenol the mass transfer rate decreases. Due to enhancing concentration gradient, performed as enhancing driving force to predominant all mass transfer resistances of the phenol between the solution and solid phase, resulting in an enhancing equilibrium sorption until sorbent saturation is obtained. This is in agreement with Balasubramani and Sivarajasekar who report that the removal rhodamine B efficiency decreases when the initial rhodamine B concentration increases. In other hands, increasing concentration of phenol leads to shifting the pHZPC to lower value and decreasing the electrostatic attraction between sorbent surfaces and phenoxide ions.
 |
Figure 6: The effect of contact time on the phenol removal in the batch adsorption reactor |
The proposed mechanism of phenol uptake rate onto eggshell powder surface involves the replacement (ion exchange adsorption) of calcium of the eggshell powder, by phenoxide ion to form an insoluble matter. The regression coefficient for the fitted line is calculated to be R2= 0.8715 for phenol. The apparent rate constant, K1 and the half-life time, t1/2 are calculated to be 0.5357 min-1 and 1.29 min. Phenol reduction follows a first order kinetic model. Phenol reduction follows a Freundlich isotherm model (R2>0.9). This is agreement with Kumaraswamy et al who report that chromium by eggshell powder follows a Langmuir isotherm model. Therefore, the eggshell powder adsorption reactor, in batch mode, is showed to be an efficient and viable process for meeting a high degree of phenol reduction from drinking water and be considered as a promising technology for treating phenol-polluted drinking water in developing countries (Figure 8, Figure 9).
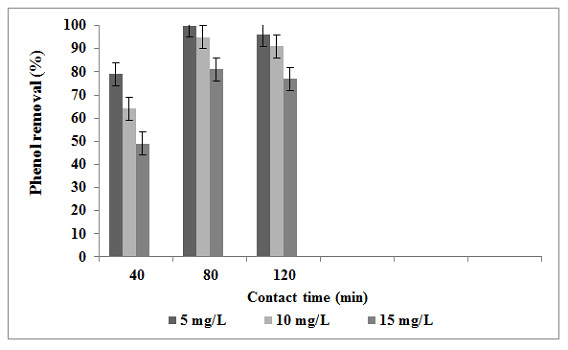 |
Figure 7: The effect of initial phenol concentration on the fluoride removal in the batch adsorption reactor |
 |
Figure 8: Kinetic models: (a) first order (b) second order |
 |
Figure 9: Conceptual model of DPSIR framework |
 |
Figure 10: Effect of contact time and initial concentration on the phenol removal (Sorbent dosage 4 g; pH 3) |
Conclusion
The experimental results suggest that batch chicken eggshell powdered reactor is a practical and promising method for the phenol-contaminated water. Phenol removal is affected by pH, the concentration of phenol, the concentration of adsorbent, and reaction time. This reactor are capable of phenol removal at the pH value (3) investigated, with a reaction time 80 min. It is purposed that performance of process is studied the other material.
Acknowledgments
The authors thank the Department of Environmental Health of Islamic Azad University, Tehran Medical Sciences Branch for financial and instrumental supports.
References
- Al-Khalid, T. and El-Naas, M.H., 2012. Aerobic biodegradation of phenols: a comprehensive review. Critical reviews in environmental science and technology, 42(16), pp.1631-
- American Public Health Association, American Water Works Association, Water Pollution Control Federation and Water Environment Federation, 2015. Standard methods for the examination of water and wastewater (Vol. 2). American Public Health Association..
- Bazrafshan, E., Amirian, P., Mahvi, A.H. and Ansari-Moghaddam, A., 2016. Application of adsorption process for phenolic compounds removal from aqueous environments: a systematic review. Global NEST Journal, 18(1), pp.146-63.
- Cabaj, J., Je˛drychowska, A., Zajłc, D., Krawiec, S. and Sołoducho, J., 2016. Phenolic Compounds Determination Using Laccase-based Electrode Modified with Conducting Polymer Support. International Journal of Electrochemical Science, 11(1), pp.609-620.
- Chand Meena, M., Band, R. and Sharma, G., 2015. Phenol and Its Toxicity: A Case Report. Iranian Journal of Toxicology, 8(27), pp.1222-1224.
- Dakhil, I.H., 2013. Removal of phenol from industrial wastewater using sawdust.International Journal of Engineering And Science, 3(1), pp.25-31.
- Daraei, H., Mittal, A., Noorisepehr, M. and Daraei, F., 2013. Kinetic and equilibrium studies of adsorptive removal of phenol onto eggshell waste.Environmental Science and Pollution Research, 20(7), pp.4603-4611.
- Dehghani, M.H., Mostofi, M., Alimohammadi, M., McKay, G., Yetilmezsoy, K., Albadarin, A.B., Heibati, B., AlGhouti, M., Mubarak, N.M. and Sahu, J.N., 2016. High-performance removal of toxic phenol by single-walled and multi-walled carbon nanotubes: Kinetics, adsorption, mechanism and optimization studies. Journal of Industrial and Engineering Chemistry, 35, pp.63-74.
- Fan, J., Zhang, J., Zhang, C., Ren, L. and Shi, Q., 2011. Adsorption of 2, 4, 6-trichlorophenol from aqueous solution onto activated carbon derived from loosestrife. Desalination, 267(2), pp.139-146.
- Giti, K. and Narges, J., 2015. Optimization electrophotocatalytic removal of acid red 18 from drinking water by the Taguchi model.
- Hsieh, F.M., Huang, C., Lin, T.F., Chen, Y.M. and Lin, J.C., 2008. Study of sodium tripolyphosphate-crosslinked chitosan beads entrapped with Pseudomonas putida for phenol degradation.Process Biochemistry, 43(1), pp.83-92.
- Kashi, G., Mehree, A., Zaeimdar, M., Khoshab, F. and Madaree, A.M., 2015. Removal of fluoride from urban drinking water by eggshell powder.
- Liao, D., Zheng, W., Li, X., Yang, Q., Yue, X., Guo, L. and Zeng, G., 2010. Removal of lead (II) from aqueous solutions using carbonate hydroxyapatite extracted from eggshell waste. Journal of Hazardous Materials, 177(1), pp.126-130.
- Loganathan, P., Vigneswaran, S., Kandasamy, J. and Naidu, R., 2013. Defluoridation of drinking water using adsorption processes. Journal of Hazardous materials, 248, pp.1-19.
- Mijan, M.A., Kim, D.H. and Kwak, H.S., 2014. Physicochemical properties of nanopowdered eggshell. International Journal of Food Science & Technology, 49(7), pp.1751-1757.
- Mourão, P.A.M., Laginhas, C., Custódio, F., Nabais, J.V., Carrott, P.J.M. and Carrott, M.R., 2011. Influence of oxidation process on the adsorption capacity of activated carbons from lignocellulosic precursors. Fuel Processing Technology, 92(2), pp.241-246.
- Ngah, W.W., Fatinathan, S. and Yosop, N.A., 2011. Isotherm and kinetic studies on the adsorption of humic acid onto chitosan-H 2 SO 4 beads. Desalination, 272(1), pp.293-300.
- Tzvetkova, P.G., Nickolov, R.N., Tzvetkova, C.T., Bozhkov, O.D. and Voykova, D.K., 2016. Diatomite/carbon adsorbent for phenol removal. Journal of Chemical Technology and Metallurgy, 51(2), pp.202-209.
- Zazouli, M.A., Taghavi, M. and Bazrafshan, E., 2012. Influences of Solution Chemistry on Phenol Removal From Aqueous Environments by Electrocoagulation Process Using Aluminum Electrodes. Health Scope, 1(2), pp.66-70.
- Zulfikar, M.A., Mariske, E.D. and Djajanti, S.D., 2012. Adsorption of lignosulfonate compound using powdered eggshells. Songklanakarin Journal of Science and Technology, 34(3), pp.309-316.
- Zulfikar, M.A., Novita, E., Hertadi, R. and Djajanti, S.D., 2013. Removal of humic acid from peat water using untreated powdered eggshell as a low cost adsorbent. International Journal of Environmental Science and Technology, 10(6), pp.1357-1366.


