Department of Bioinformatics, Maulana Azad National Institute of Technology (MANIT), Bhopal- 462051 (M.P.), INDIA
Corresponding author Email: bioinfosunil@gmail.com
Article Publishing History
Received: 29/12/2016
Accepted After Revision: 23/03/2017
In this review, PubMed database has been explored to elucidate the problems related to HIV/AIDS, which have been solved previously using various machine learning approaches and some other techniques. Literatures from the epidemic years of HIV/AIDS till February, 2017 have been examined and problems such as prediction of HIV/AIDS protease cleavage sites and inhibitors, prediction of coreceptors usage for viral entry, development of anti-viral agents and prediction of response, resistance and adverse effect of antiretroviral therapy have been considered for the current study. Complications associated with HIV/AIDS infection as well as all three stages of HIV infection have been described. HIV virus binding to the coreceptors CCR5 and CXCR4 are delineated to show the significant role of the coreceptors for the anti-HIV drug development. After exploring various datasets, viral tropisms are found to be relevant to the viral third V3 region of the HIV virus binding.
Hiv, Machine Learning Techniques, V3 Region, Cd4 Receptor, Ccr5 And Cxcr4 Coreceptors
Kumari S, Chouhan U, Suryawanshi S. K. Machine Learning Approaches to Study HIV/AIDS Infection: A Review. Biosc.Biotech.Res.Comm. 2017;10(1).
Kumari S, Chouhan U, Suryawanshi S. K. Machine Learning Approaches to Study HIV/AIDS Infection: A Review. Biosc.Biotech.Res.Comm. 2017;10(1). Available from: https://bit.ly/30yKYoz
Introduction
Human immunodeficiency virus/acquired immune deficiency syndrome (HIV/AIDS) was originated from monkey in the United States in 1981. AIDS is a chronic and potentially most threatening infectious disease caused by human immunodeficiency virus in the 21st century. 78 million people were estimated to be suffering from HIV/AIDS and 35 million people have died since the start of the epidemic year but 36.7 million people were reported as HIV/AIDS infected and 1.1 million people have died in 2015 globally; 2.1 million people were found to be newly HIV/AIDS infected globally (Alkema 2016). Eastern and Southern Africa have a maximum increase since almost the start of the epidemic year. HIV/AIDS regularly decimated the population of the Africa shown in figure1 (HIV/AIDS 2016).
 |
Figure 1: New HIV infections among people aged 15 years and above in different regions since last six years from 2010 to 2015(Alkema et al. 2016) |
The current prevalence of the HIV/AIDS is 0.8% of the worldwide. 18.2 million people were accessing antiretroviral therapy in June 2016 (Alkema et al. 2016). Antiretroviral therapy (ART) has a very crucial role in the HIV treatment, but HIV infected individuals acquire resistance to the ART over a certain period. Antiretroviral therapy coverage increased from 7.51 million in 2010 to 17.03 million in 2015 shown in figure 2.
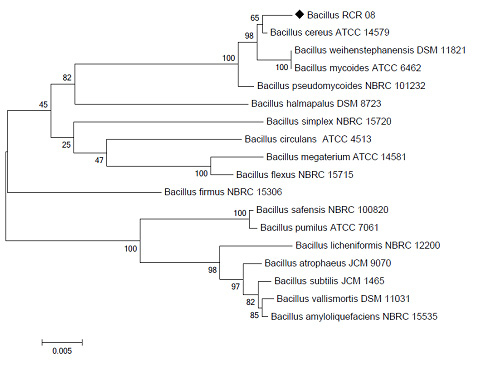 |
Figure 2: Antiretroviral therapy coverage % in different regions since last six years from 2010 to 2015(Bernard 2016) |
However, scientists are working to develop one vaccine to prevent HIV/AIDS. NIH supported clinical trial that was launched in 2016 to test a possible HIV/AIDS vaccine. This vaccine trial HVTN 072 is testing whether an experimental vaccine regimen safely prevents HIV/AIDS infection among South African adults (Health 2015).
The determination of the 90-90-90 treatment target is to be achievable by the reinforcement of the continuing momentum in 2020, whereby 90% of the HIV/AIDS infected people aware of their HIV/AIDS status, 90 % of the people with known their HIV/AIDS positive status are taking treatment and 90% of the people on HIV/AIDS treatment have suppressed viral loads (NASCOP 2014). Recent updated UNAIDS estimates indicate that US$ 26.2 billion will be required for the HIV/AIDS response in 2020, with US$ 23.9 billion required in 2030 (NASCOP 2014). The world has committed to ending the HIV/AIDS epidemic by 2030 (Bernard 2016).
Methods
We searched PubMed database with the keywords of “HIV” and “Machine Learning Approaches” up to February 22, 2017 and starting time was not given. The number of the articles retrieved from the PubMed database was 114, out of which there was 6 review articles. Efforts here made to review some uncertainties related to HIV infection. The search completeness was examined by the list of references of the reviewed articles. Somewhere, Book chapters were also included, some articles were excluded on criteria such as articles not in English language, campaign posters, newspaper articles and the articles irrelevant to the topic of this review. In numbers, 32 originally related articles were selected on the basis of this evaluation are listed in Table-1.
| Table 1: Literatures involved for the current study for machine learning approaches used to solve various HIV/AIDS problems. | ||||
| Number | Author name-Publication year | Methods | Objectives | Country |
| 1 | (Gupta 2015) | Machine learning methods | Prediction of tropism in HIV-1 subtype C V3 loop sequences using genotypic tools. | India |
| 2 | (Fogel 2015) | Artificial neural networks | Classification of R5-, X4- and R5X4-tropics HIV-1 using evolved neural networks. | US |
| 3 | (Antell 2016) | Machine learning | Identification of R5- and X4-specific Tat and LTR sequence signatures using HIV envelope V3. | USA |
| 4 | (Kumar 2013) | Support vector machine | Prediction of HIV-1 coreceptor usages using hybrid approach from its V3 loop amino acid sequences. | India |
| 5 | (Sander 2007) | Heuristic and Statistical learning methods | Prediction of HIV-1 coreceptor usage by structural descriptors of gp120 V3 loop. | Germany |
| 6 | (Dybowski 2010) | Two-level machine learning methods | Prediction of co-receptor usage of HIV-1 from genotype. | Germany |
| 7 | (Masso 2010) | Random forest, support vector machine, boosted decision tree, and neural network machine learning algorithms | Determination of HIV-1 co-receptor usage from accurate and efficient gp120 V3 loop structure based models. | USA |
| 8 | (Evans 2013) | Support vector machine, PSSM and 11/25 rule | Genotypic prediction of HIV-1 coreceptor tropism using a case-based reasoning system. | USA |
| 9 | (Kim 2010) | Feature selection subset method of multi-layered perceptron (FS-MLP) learning | Analysis of HIV-1 protease cleavage site an MLP-based feature subset selection. | Korea |
| 10 | (Singh 2016) | Artificial neural networks | HIV-1 protease cleavage site prediction using a combination of sequence, structural, and physiochemical properties. | Taiwan |
| 11 | (Öztürk 2013) | Support vector machine | Prediction of HIV-1 protease cleavage site using a consistency-based feature selection method allied with linear SVMs. | Turkey |
| 12 | (Wei 2015) | Support vector machine, shape, pharmacophore and docking methods | Multistage virtual screening and novel identification of HIV-1 protease inhibitors by integrating SVM, shape, pharmacophore and docking methods. | China |
| 13 | (Kirchmair 2011) | Virtual screening techniques. | Development of anti-viral agents using molecular modeling and virtual screening techniques. | Austria |
| 14 | (Zazzi 2012) | Machine learning approaches | Prediction of antiretroviral treatment response using machine learning: The EuResist project. | Italy |
| 15 | (Prosperi 2011) | Machine learning approaches | Prediction of antiretroviral treatment response using computational models. | USA |
| 16 | (Prosperi 2009) | Linear and Non-linear statistical learning models | Prediction of response to antiretroviral treatment by investigating of expert rule bases, logistic regression, and non-linear machine learning techniques. | Italy |
| 17 | (Zazzi 2016) | Expert’s Rules and Statistical/Machine learning algorithms. | Computer-Aided Optimization of Combined Anti-Retroviral Treatment for HIV: New Drugs, Drug Targets and Drug Resistance. | US |
| 18 | (Riemenschneider 2016a) | Support vector machine, Random forest and Statistical methods | Prediction of HIV Drug Resistance using Current Computational Approaches. | Germany |
| 19 | (Riemenschneider 2016b) | Machine learning techniques, binary relevance classifiers, classifier chains, and ensembles of classifier chains | Multiclass classification for HIV-1 drug resistance prediction by exploiting cross-resistance information with 662 protease sequences and 715 reverse transcriptase sequences. | Germany |
| 20 | (Heider 2013) | Machine learning techniques. | Multiclass classification for HIV-1 drug resistance prediction by exploiting cross-resistance information. | Germany |
| 21 | (Srisawat 2008) | Support vector machine, Radial basis function Networks and k-nearest neighbor methods | Prediction of HIV-1 drug resistance by combining classifiers. | Thailand |
| 22 | (Adrover et al. 2015) | Machine learning and crowdsourced human assessment | Identification of adverse effects of HIV drug treatment and related sentiments using Twitter. | US |
| 23 | (Evans 2014) | Boosted algorithms of consisting of multiple machine learnings and statistical models | Prediction of HIV-1 broadly neutralizing antibody epitope networks using neutralization titers and a novel computational method. | USA |
| 24 | (Hepler 2014) | Machine learning methods | IDEPI: fast prediction of HIV-1 antibody epitopes and other phenotypic features from sequence dataset using a flexible machine learning techniques. | USA |
| 25 | (Choi 2015) | Machine learning methods | Prediction of antibody feature: function relationships in RV144 vaccines using machine learning methods. | USA |
| 26 | (Poongavanam 2013) | Virtual screening | Prediction of HIV-1 RT related to RNase H inhibition using virtual screening. | Denmark |
| 27 | (Pancholi 2014) | SVM, Back propagation neural networks | Ligand based computational modeling studies on non-nucleoside reverse transcriptase inhibitors of HIV-1 | India |
| 28 | (Xuan 2013) | Support vector machine and Regression methods | Bioactivity of HIV-1 integrase ST inhibitors predicted using multilinear regression analysis and support vector machine. | China |
| 29 | (Yan 2012) | Machine learning methods | Support vector machine used for classification of active and weakly active ST inhibitors of HIV-1 integrase. | USA |
| 30 | (Mei 2013) | Support vector machine | Prediction of interactions between HIV-1 and human proteins using probability weighted ensemble transfer learning. | China |
| 31 | (Dawany 2014) | Support vector machine | Detection of accurate M. tuberculosis in patients with and without HIV co-infection by identification of a 251 gene expression signature. | USA |
| 32 | (Holman 2012) | Machine learning methods | Identification of amino acid signatures in the HIV env gene predictive of dementia using a machine learningapproach. | USA |
Stages Og HIV/AID
There are mainly three stages of HIV: Primary infection (Acute HIV), Clinical latent infection (Chronic HIV) and Early symptomatic HIV infection (Wawer 2005). In primary infection, a flu-like illness developed within one or two months after the virus entering the body of people, including signs and symptoms such as fever, headache, muscle aches, joint pain, rash, sore throat and swollen lymph glands mainly on the neck. Though, the symptoms of the first stage of HIV infection are mostly unnoticed, the amount of virus in the viral load or bloodstream spreads very highly at this time, resulting in dispersing the HIV infection more efficiently during primary infection than the next stage. In the clinical latent infection, generally persistent swelling of lymph nodes occurs and HIV remains in the human body but with no signs and symptoms. This stage lasts around 10 years in people not receiving antiretroviral medications and lasts for decades in people receiving antiretroviral therapy. Some people expedite to more intense disease much sooner. In the last stage of early symptomatic HIV infection, people probably progress to mild infections or chronic signs and symptoms such as fever, fatigue, swollen lymph nodes, diarrhea, weight loss, oral yeast infection (thrush) and shingles (herpes zoster) due to continuous replication of the virus in the human body and distortion of the immune cells.
Complications of HIV/AIDS
The burden of HIV is to a large extent consequence of infections (tuberculosis (TB), cytomegalovirus, candidiasis, cryptococcal meningitis, toxoplasmosis and cryptosporidiosis), cancers (Kaposi’s sarcoma and lymphomas) and others (wasting syndrome, neurological complications and kidney dis How ease) (How). The increasing in the incidences of the HIV/AIDS and their consequences in terms of falling down the number of CD4 (Cluster of differentiation 4) receptors and coreceptors lead to damage the immune system of the human. Therefore, identification of the used coreceptor for the viral entry that can target to block the coreceptor to bind with the virus and maintenance of the CD4 receptors and coreceptors is being crucial for new therapeutic agents for the treatment of immune deficiency syndrome (Kaplan 2009).
Infections common to HIV/AIDS
Tuberculosis (TB)
Tuberculosis coinfection is associated with increase viral replications. TB is the most occurring infection related to HIV/AIDS and a leading cause of death (Pawlowski 2012).
Cytomegalovirus
This herpes virus is transmitted in human body fluids such as saliva, urine, blood, breast milk and semen. The virus remains inactive in a healthy immune system and remains resurfaces in weakens immune system causing damage to eyes, lungs, digestive tract or other organs (Mathevula 2013).
Candidiasis
Candidiasis causes inflammation and a thick, white coating on the mucous membranes of mouth, tongue, vagina or esophagus (Cutlan 2010).
Cryptococcal Meningitis
Cryptococcal meningitis is a common infection of the central nervous system associated with HIV/AIDS, caused by a fungus found in soil. Meningitis is an inflammation of the membranes and fluid surrounding brain and spinal cord (Park 2009).
Toxoplasmosis
This infection is caused by Toxoplasma gondii, a parasite dispersed primarily by cats. Infected cats successfully pass the parasites in their stools, and the parasites may then transfer to other animals and humans (Berger-Schoch 2011).
Cryptosporidiosis
An intestinal parasite usually found in animals and it is responsible for the infection. Cryptosporidiosis contracted when human ingests contaminated food or water. The parasite grows in the intestines of human and bile ducts, leading to severe, chronic diarrhea in HIV/AIDS infected people (Mathewos 2014).
Cancers common to HIV/AIDS
Kaposi’s sarcoma
Kaposi’s sarcoma is a blood vessel walls, a very rare tumor in HIV-negative people but a very common in HIV-positive people. It usually appears as pink, purple or red lesions on the skin and mouth of the HIV infected people (Page 2006). Internal organs can also be affected by Kaposi’s sarcoma (Di Benedetto 2008).
Lymphomas
Lymphomas cancer originates in white blood cells and usually appears in lymph nodes. Painless swelling of the lymph nodes in neck, armpit or groin is most common early sign (Daniels 2012).
Other complications
Wasting syndrome
Aggressive treatment regimens have decreased the number of cases of wasting syndrome, but it still affects many HIV/AIDS infected people. It leads to a loss of at least 10 percent of human body weight, often escorted by diarrhea, chronic weakness and fever (Bass 2015).
Neurological complications
It can cause neurological symptoms such as confusion, depression, forgetfulness, anxiety and difficulty walking, whereas AIDS doesn’t infect the nerve cells. AIDS dementia complex is one of the most common neurological complications, which leads to changes in behavior and diminishes in mental function (Ances 2007).
Kidney disease
HIV-associated nephropathy (HIVAN) is an inflammation of the small filters in kidneys that remove extra fluid and wastes from bloodstream and pass them to your urine. The risk of developing HIVAN is much higher in black people due to a genetic predisposition. A antiretroviral therapy should be started in those diagnosed with HIVAN in any case of CD4 count (Scherzer 2012).
Receptors
CD4 receptors are particularly attractive target molecules, since they have been found to play a certain role in maintaining the immune system of the human. CCR5 and CXCR4 are two coreceptors of the receptor present on the T-cell, which is useful for viral cellular entry shown in figure 3.
 |
Figure 3: HIV virus cellular entry through binding with the CD4 receptor and CXCR4 and CCR5 coreceptor in T-tropic and M-tropic HIV respectively(Gorry 2005) |
CCR5 (C-C chemokine receptor type 5)
The CCR5 protein belongs to the family of the beta chemokine receptors of the integral membrane proteins (Samson 1996). The CCR5 coreceptors are G protein-coupled coreceptors (Aavikko 2014). CCR5 coreceptors are also known as CD195 proteins expressed on the surface of white blood cells, T helper cells, macrophages, dendritic cells, eosinophils and microglia. T helper cells are specific tissues and organ targets by which HIV virus causes AIDS using CCR5 coreceptors to enter and infect into the immunological cells. The viral entry of HIV-1 into a target host cell is enabled by the essential HIV-1 envelope glycoprotein structure (Alkhatib 2009). The Gp120 external subunit and Gp41 transmembrane subunit are two subunits of the envelope glycoprotein structure cleaved from a Gp160 protein precursor encoded by the HIV-1 env gene (Checkley 2011).
The Gp120 subunit binds to a CD4 glycoprotein and a HIV-1 coreceptor CCR5 expressed on a target host cell forming a heterotrimeric complex (Murphy 2001). The binding of Gp120 envelope protein to CCR5 coreceptor consists two crucial steps: The tyrosine sulfated amino terminus of this coreceptor is an “essential determinant” of binding to Gp120 in the first step and there must be reciprocal action (synergy, intercommunication) between gp120 and the CCR5 transmembrane domains in a second step (Ji 2007). Some individuals lead to a mutation as CCR5-delta 32 in the CCR5 gene by the genetic deletion of a portion of the CCR5 gene to protect them against these HIV strains. This mutation in homozygous carriers is resistant to the M-tropic strains of the HIV-1 infected individuals (De Silva 2004 Hütter 2009 Allers 2011 Zhen 2013 Kay 2014 and Tebas 2014).
The CCR5 gene encodes the CCR5 protein, which is located on the short (p) arm at the position of 21 on chromosome 3.The cognate ligands of CCR5 include CCL3, CCL4 and CCL3L1 (Struyf 2001 and Miyakawa 2002) and CCR5 interacts with CCL5 (Struyf et al. 2001 Miyakawa et al. 2002 and Slimani 2003).The general ligands for the receptor RANTES, MIP-1á and MIP-1â are able to suppress HIV-1 infection in vitro (Nakayama 2012).
CXCR4 (C-X-C chemokine receptor type 4)
CXCR4 is an alpha-chemokine receptor specific for stromal-derived-factor-1 (SDF-1) also known as CD184 (cluster of differentiation 184) proteins. SDF-1 is a molecule endowed with effective chemotactic activity of lymphocytes. During the implantation and hormone replacement therapy cycle in the endomentrium, producing, in the presence of human blastocyst and a surface polarization of the CXCR4 coreceptors suggest that these coreceptors are connected in the adhesion phase of the human implantation, CXCR4 is upregulated. The ligand of CXCR4, SDF-1 is known to be significant in hematopoietic stem cell homing to the bone marrow and in hematopoietic stem cell quiescence. CXCR4 signaling coreceptors found to regulate the CD20 expression on B cells (Pavlasova 2016).
SDF-1 and CXCR4 were believed to be a relatively monogamous ligand-receptor pair (other chemokines are promiscuous, tending to use several different chemokine receptors) until recently. Recent evidence demonstrates ubiquitin is also a natural ligand of CXCR4 (Saini 2010). Ubiquitin is a small (76-amino acid) protein highly conserved among eukaryotic cells. It is best known for its intracellular role in targeting ubiquitylated proteins for degradation via the ubiquitin proteasome system. Evidence in numerous animal models suggests ubiquitin is anti-inflammatory immune modulator and proinflammatory damage endogenous opponent associated molecular pattern molecules (Majetschak 2011). It is speculated this interaction may be through CXCR4 mediated signaling pathways. MIF is an additional ligand of CXCR4 (Bernhagen 2007).
CXCR4 plays a role in neurological guidance by presenting in newly generated neurons during embryogenesis and adult life. The receptor levels decrease as neurons mature. CXCR4 mutant mice have aberrant neuronal distribution. This has been implicated in disorders such as epilepsy (Bagri 2002).
The viral regions
Coreceptor consideration holds the V1-V2 region oh Gp120 and the bridging sheet such as antiparallel and 4-stranded â sheet that joints the inner and outer domains of the Gp120. The coreceptor usage through its peptide composition and the degree of N-linked glycosylation can be influenced by the V1-V2 stem. In contrast with V1-V2 region, the V3 loop region is highly mutable and therefore it is the most significant determinant of coreceptor specificity (Bozek 2013).
The HIV enzymes role
The viral genome reverse transcription is required to generate the proviral DNA and the integration into the target genome cell for successful HIV-1 virus replication. The viral encoded enzymes such as reverse transcriptase (RT) and integrase (IN) put their potential for these events and act sequentially during viral replication. The double stranded proviral DNA produced by replicating both RNA and DNA templates using RT enzyme which is a heterodimer of p66 and p51 subunits (Arts 1997). A defined set of DNA cleavage and combined events to insert the proviral DNA into the host genome directed by the IN enzyme which is 32 kDa polypeptide (Wiskerchen 1995). HIV-1 protease (PR) is important for the life- cycle of the HIV virus and this enzyme is responsible for cleavage of the newly synthesized polyproteins at the appropriate places to create the mature protein components of an infectious HIV virion. HIV virions remain uninfected without effective HIV PR (Doitsh
2010).
The review highlights the scarcity of information on the HIV/AIDS problems which are possible to solve using various machine learning techniques are shortlisted in Table 1. Classification of X4, R5 and dual (R5X4) tropic HIV-1 from V3 loop amino acid sequences of HIV-1 subtype B using artificial neural networks up to accuracy of 81.8% (Fogel et al. 2015) and classification of X4 and R5tropic HIV-1 from V3 loop amino acid sequences of HIV-1 subtype C using SVM showed a good concordance of 85% (Gupta et al. 2015) are done in USA and India respectively. Prediction of coreceptor usage for viral entry from Gp120 V3 loop amino acid sequences using SVM, heuristic and statistical learning methods, two-level machine, random forest, boosted decision tree, and neural network machine learning algorithms has been done in India, Germany and USA respectively (Raghava 2013 Sander et al. 2007 Dybowski 2010 and Vaisman 2010). Prediction of HIV-1 protease cleavage site and inhibitors using Feature selection subset method of multi-layered perceptron (FS-MLP) learning, SVM, ANN pharmacophore and docking methods have been done 80.0% ~ 97.4% accuracy in Taiwan, Korea, Turkey and China respectively (Kim et al. 2010 Singh Su 2016 Öztürk et al. 2013 and Wei et al. 2015).
Some of the literatures are regarding of drug development, anti-viral agents development (Kirchmair et al. 2011), antiretroviral response prediction (Zazzi et al. 2012 Prosperi 2011 and Prosperi et al. 2009), antiretroviral resistance prediction (Zazzi 2016 Riemenschneider 2016a Riemenschneider 2016b Heider et al. 2013 and Kijsirikul 2008), antiretroviral adverse effects prediction (Adrover et al. 2015) has been analyzed which were used machine learning approaches, SVM, Expert’s Rules and Linear and Non-linear statistical learning algorithms, Radial basis function networks (RBF networks), k-nearest neighbor (kNN) and Virtual screening method to improve their result in Austria, Italy, USA, Germany and Thailand. Prediction of antibody of HIV epitope networks using neutralization titers and a novel computational methods or a simple machine learning methods has been done in USA (Evans et al. 2014 Hepler et al. 2014 and Choi et al. 2015.
Prediction of HIV-1 RT associated RNase H inhibition (Poongavanam 2013) shown good enrichment (80-90%) by receptor-based flexible docking experiments compared to ligand-based approaches such as FLAP (74%), shape similarity (75%) and random forest (72%) in Denmark. Ligand based computational modeling studies on non-nucleoside reverse transcriptase inhibitors of HIV-1 (Pancholi et al. 2014) has been done India respectively. Prediction of bioactivities of HIV-1 integrase ST inhibitors (Xuan et al. 2013) and classification of active and weakly active ST inhibitors of HIV-1 integrase has (Yan et al. 2012) been done using machine learning approaches in China and USA respectively. Prediction of interactions between HIV-1 and human proteins using SVM in China has also done (Wei et al. 2015). One of the literatures has worked on the detection of M tuberculosis in patients with and without HIV coinfection by identifying a 251 gene expression signatures using SVM with 81.4% and 88.9-94.7% accuracy respectively in USA (Dawany et al. 2014).
Conclusion
After reviewing all the literature, the coreceptor which has been used for cellular viral entry is very necessary to identify to develop the drugs that can target the coreceptor and prevent the coreceptor from binding with HIV virus. Maraviroc has been found to be an as successful barrier in blocking the CCR5 coreceptor but fails in blocking the CXCR4 coreceptor. Prediction of HIV-1 coreceptor usage is necessary to identify the number of CD4 counts remained into the body of HIV-1 infected people. HIV-1 protease is a retroviral aspartyl protease (retropepsin) that is essential for the HIV-1 lifecycle, the retrovirus that is responsible for AIDS. The integral role of the HIV-1 protease in the viral replication, HIV-1 protease has become a prime target for drug therapy. Identification of the coreceptor which has been used for cellular viral entry with different machine learning approaches and more high accuracy than the previous findings is our future work for contribution in the drug development of HIV-1.
Acknowledgement
We are thankful to the Department of Biotechnology (DBT), New Delhi for providing support for this work under Bioinformatics Infrastructure Facility of DBT at Maulana Azad National Institute Technology, Bhopal.
References
- Aavikko, M. (2014) Identification of novel tumor predisposition families and underlying genetic defects.
- Adrover, C., Bodnar, T., Huang, Z., Telenti, A. ,Salathé, M. (2015) Identifying adverse effects of HIV drug treatment and associated sentiments using Twitter. JMIR public health and surveillance, Vol. 1, No. 2.
- Alkema, L., Chou, D., Hogan, D., Zhang, S., Moller, A.-B., Gemmill, A., Fat, D.M., Boerma, T., Temmerman, M. ,Mathers, C. (2016) Global, regional, and national levels and trends in maternal mortality between 1990 and 2015, with scenario-based projections to 2030: a systematic analysis by the UN Maternal Mortality Estimation Inter-Agency Group. The Lancet, Vol. 387, No. 10017, pp. 462-74.
- Alkhatib, G. (2009) The biology of CCR5 and CXCR4. Current Opinion in HIV and AIDS, Vol. 4, No. 2, p. 96.
- Allers, K., Hütter, G., Hofmann, J., Loddenkemper, C., Rieger, K., Thiel, E. ,Schneider, T. (2011) Evidence for the cure of HIV infection by CCR5Δ32/Δ32 stem cell transplantation. Blood, Vol. 117, No. 10, pp. 2791-9.
- Ances, B.M. ,Ellis, R.J. (2007) Dementia and neurocognitive disorders due to HIV-1 infection, Seminars in neurology, Vol. 27, Copyright© 2007 by Thieme Medical Publishers, Inc., 333 Seventh Avenue, New York, NY 10001, USA., pp. 086-92.
- Antell, G.C., Dampier, W., Aiamkitsumrit, B., Nonnemacher, M.R., Jacobson, J.M., Pirrone, V., Zhong, W., Kercher, K., Passic, S. ,Williams, J.W. (2016) Utilization of HIV-1 envelope V3 to identify X4-and R5-specific Tat and LTR sequence signatures. Retrovirology, Vol. 13, No. 1, p. 32.
- Arts, E.J., Le Grice, S.F. (1997) Interaction of retroviral reverse transcriptase with template–primer duplexes during replication. Progress in nucleic acid research and molecular biology, Vol. 58, pp. 339-93.
- Bagri, A., Gurney, T., He, X., Zou, Y.-R., Littman, D.R., Tessier-Lavigne, M. ,Pleasure, S.J. (2002) The chemokine SDF1 regulates migration of dentate granule cells. Development, Vol. 129, No. 18, pp. 4249-60.
- Bass, L.H., Washington, C.M. (2015) Infection Control in Radiation Oncology Facilities. Principles and Practice of Radiation Therapy, p. 178.
- Berger-Schoch, A., Herrmann, D., Schares, G., Müller, N., Bernet, D., Gottstein, B. ,Frey, C. (2011) Prevalence and genotypes of Toxoplasma gondii in feline faeces (oocysts) and meat from sheep, cattle and pigs in Switzerland. Veterinary parasitology, Vol. 177, No. 3, pp. 290-7.
- Bernard, E., Cameron, S. (2016) Advancing HIV justice 2. Building momentum in global advocacy against HIV criminalisation.
- Bernhagen, J., Krohn, R., Lue, H., Gregory, J.L., Zernecke, A., Koenen, R.R., Dewor, M., Georgiev, I., Schober, A. ,Leng, L. (2007) MIF is a noncognate ligand of CXC chemokine receptors in inflammatory and atherogenic cell recruitment. Nature medicine, Vol. 13, No. 5, pp. 587-96.
- Bozek, K., Lengauer, T., Sierra, S., Kaiser, R. ,Domingues, F.S. (2013) Analysis of physicochemical and structural properties determining HIV-1 coreceptor usage. PLOS Comput Biol, Vol. 9, No. 3, p. e1002977.
- Checkley, M.A., Luttge, B.G. ,Freed, E.O. (2011) HIV-1 envelope glycoprotein biosynthesis, trafficking, and incorporation. Journal of molecular biology, Vol. 410, No. 4, pp. 582-608.
- Choi, I., Chung, A.W., Suscovich, T.J., Rerks-Ngarm, S., Pitisuttithum, P., Nitayaphan, S., Kaewkungwal, J., O’Connell, R.J., Francis, D. ,Robb, M.L. (2015) Machine learning methods enable predictive modeling of antibody feature: function relationships in RV144 vaccinees. PLOS Comput Biol, Vol. 11, No. 4, p. e1004185.
- Cutlan, J., Saunders, N., Olsen, S. ,Fullen, D. (2010) White sponge nevus presenting as genital lesions in a 28‐year‐old female. Journal of cutaneous pathology, Vol. 37, No. 3, pp. 386-9.
- Daniels, V.G. (2012) AIDS: the acquired immune deficiency syndrome, Springer Science & Business Media.
- Dawany, N., Showe, L.C., Kossenkov, A.V., Chang, C., Ive, P., Conradie, F., Stevens, W., Sanne, I., Azzoni, L. ,Montaner, L.J. (2014) Identification of a 251 gene expression signature that can accurately detect M. tuberculosis in patients with and without HIV co-infection. PloS one, Vol. 9, No. 2, p. e89925.
- De Silva, E. ,Stumpf, M.P. (2004) HIV and the CCR5-Δ32 resistance allele. FEMS microbiology letters, Vol. 241, No. 1, pp. 1-12.
- Di Benedetto, F., Di Sandro, S., De Ruvo, N., Berretta, M., Masetti, M., Montalti, R., Ballarin, R., Cocchi, S., Potenza, L. ,Luppi, M. (2008) Kaposi’s sarcoma after liver transplantation. Journal of cancer research and clinical oncology, Vol. 134, No. 6, pp. 653-8.
- Doitsh, G., Cavrois, M., Lassen, K.G., Zepeda, O., Yang, Z., Santiago, M.L., Hebbeler, A.M. ,Greene, W.C. (2010) Abortive HIV infection mediates CD4 T cell depletion and inflammation in human lymphoid tissue. Cell, Vol. 143, No. 5, pp. 789-801.
- Dybowski, J.N., Heider, D. ,Hoffmann, D. (2010) Prediction of co-receptor usage of HIV-1 from genotype. PLOS Comput Biol, Vol. 6, No. 4, p. e1000743.
- Evans, M.C., Paquet, A.C., Huang, W., Napolitano, L., Frantzell, A., Toma, J., Stawiski, E.W., Goetz, M.B., Petropoulos, C.J. ,Whitcomb, J. (2013) A case-based reasoning system for genotypic prediction of hiv-1 co-receptor tropism. Journal of bioinformatics and computational biology, Vol. 11, No. 04, p. 1350006.
- Evans, M.C., Phung, P., Paquet, A.C., Parikh, A., Petropoulos, C.J., Wrin, T. ,Haddad, M. (2014) Predicting HIV-1 broadly neutralizing antibody epitope networks using neutralization titers and a novel computational method. Bmc Bioinformatics, Vol. 15, No. 1, p. 77.
- Fogel, G.B., Lamers, S.L., Liu, E.S., Salemi, M. ,McGrath, M.S. (2015) Identification of dual-tropic HIV-1 using evolved neural networks. Biosystems, Vol. 137, pp. 12-9.
- Gorry, P.R., Churchill, M., Crowe, S.M., Cunningham, A.L. ,Gabuzda, D. (2005) Pathogenesis of macrophage tropic HIV-1. Current HIV research, Vol. 3, No. 1, pp. 53-60.
- Gupta, S., Neogi, U., Srinivasa, H. ,Shet, A. (2015) Performance of genotypic tools for prediction of tropism in HIV-1 subtype C V3 loop sequences. Intervirology, Vol. 58, No. 1, pp. 1-5.
- Heider, D., Senge, R., Cheng, W. ,Hüllermeier, E. (2013) Multilabel classification for exploiting cross-resistance information in HIV-1 drug resistance prediction. Bioinformatics, Vol. 29, No. 16, pp. 1946-52.
- Hepler, N.L., Scheffler, K., Weaver, S., Murrell, B., Richman, D.D., Burton, D.R., Poignard, P., Smith, D.M. ,Pond, S.L.K. (2014) IDEPI: rapid prediction of HIV-1 antibody epitopes and other phenotypic features from sequence data using a flexible machine learning platform. PLOS Comput Biol, Vol. 10, No. 9, p. e1003842.
- Holman, A.G. ,Gabuzda, D. (2012) A machine learning approach for identifying amino acid signatures in the HIV env gene predictive of dementia. PloS one, Vol. 7, No. 11, p. e49538.
- Hütter, G., Nowak, D., Mossner, M., Ganepola, S., Müßig, A., Allers, K., Schneider, T., Hofmann, J., Kücherer, C. ,Blau, O. (2009) Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. New England Journal of Medicine, Vol. 360, No. 7, pp. 692-8.
- Ji, C., Zhang, J., Dioszegi, M., Chiu, S., Rao, E., Cammack, N., Brandt, M. ,Sankuratri, S. (2007) CCR5 small-molecule antagonists and monoclonal antibodies exert potent synergistic antiviral effects by cobinding to the receptor. Molecular pharmacology, Vol. 72, No. 1, pp. 18-28.
- Kaplan, J.E., Benson, C., Holmes, K.K., Brooks, J.T., Pau, A., Masur, H., Control, C.f.D., Prevention, Health, N.I.o. ,America, H.M.A.o.t.I.D.S.o. (2009) Guidelines for prevention and treatment of opportunistic infections in HIV-infected adults and adolescents. MMWR Recomm Rep, Vol. 58, No. RR-4, pp. 1-207.
- Kay, M.A., Walker, B.D. (2014) Engineering cellular resistance to HIV. N Engl J Med, Vol. 370, No. 10, pp. 968-9.
- Kim, G., Kim, Y., Lim, H. ,Kim, H. (2010) An MLP-based feature subset selection for HIV-1 protease cleavage site analysis. Artificial intelligence in medicine, Vol. 48, No. 2, pp. 83-9.
- Kirchmair, J., Distinto, S., Roman Liedl, K., Markt, P., Maria Rollinger, J., Schuster, D., Maria Spitzer, G. ,Wolber, G. (2011) Development of anti-viral agents using molecular modeling and virtual screening techniques. Infectious Disorders-Drug Targets (Formerly Current Drug Targets-Infectious Disorders), Vol. 11, No. 1, pp. 64-93.
- Kumar, R., Raghava, G.P. (2013) Hybrid approach for predicting coreceptor used by HIV-1 from its V3 loop amino acid sequence. PloS one, Vol. 8, No. 4, p. e61437.
- Majetschak, M. (2011) Extracellular ubiquitin: immune modulator and endogenous opponent of damage-associated molecular pattern molecules. Journal of leukocyte biology, Vol. 89, No. 2, pp. 205-19.
- Masso, M., Vaisman, I.I. (2010) Accurate and efficient gp120 V3 loop structure based models for the determination of HIV-1 co-receptor usage. Bmc Bioinformatics, Vol. 11, No. 1, p. 494.
- Mathevula, H.M. (2013) Factors affecting adherence to treatment in patients on chronic medication at Mokopane Hospital, University of Limpopo (Turfloop Campus).
- Mathewos, M. (2014) Prevalence of opportunistic intestinal parasitic infections among hiv/aids patients attending Othona hospital, Wolayita Sodo, Southern Ethiopia.
- Mei, S. (2013) Probability weighted ensemble transfer learning for predicting interactions between HIV-1 and human proteins. PloS one, Vol. 8, No. 11, p. e79606.
- Miyakawa, T., Obaru, K., Maeda, K., Harada, S. ,Mitsuya, H. (2002) Identification of amino acid residues critical for LD78â, a variant of human macrophage inflammatory protein-1á, binding to CCR5 and inhibition of R5 human immunodeficiency virus type 1 replication. Journal of Biological Chemistry, Vol. 277, No. 7, pp. 4649-55.
- Murphy, P.M. (2001) Viral exploitation and subversion of the immune system through chemokine mimicry. Nature immunology, Vol. 2, No. 2, pp. 116-22.
- Nakayama, K., Nakamura, H., Koga, M., Koibuchi, T., Fujii, T., Miura, T., Iwamoto, A. ,Kawana-Tachikawa, A. (2012) Imbalanced production of cytokines by T cells associates with the activation/exhaustion status of memory T cells in chronic HIV type 1 infection. AIDS research and human retroviruses, Vol. 28, No. 7, pp. 702-14.
- Öztürk, O., Aksaç, A., Elsheikh, A., Özyer, T. ,Alhajj, R. (2013) A consistency-based feature selection method allied with linear SVMs for HIV-1 protease cleavage site prediction. PloS one, Vol. 8, No. 8, p. e63145.
- Page, J., Louw, M. ,Pakkiri, D. 2006, Working with HIV/Aids, Juta and Company Ltd.
- Pancholi, N.J., Gupta, S., Sapre, N. ,Sapre, N.S. (2014) Design of novel leads: ligand based computational modeling studies on non-nucleoside reverse transcriptase inhibitors (NNRTIs) of HIV-1. Molecular BioSystems, Vol. 10, No. 2, pp. 313-25.
- Park, B.J., Wannemuehler, K.A., Marston, B.J., Govender, N., Pappas, P.G. ,Chiller, T.M. (2009) Estimation of the current global burden of cryptococcal meningitis among persons living with HIV/AIDS. Aids, Vol. 23, No. 4, pp. 525-30.
- Pavlasova, G., Borsky, M., Seda, V., Cerna, K., Osickova, J., Doubek, M., Mayer, J., Calogero, R., Trbusek, M. ,Pospisilova, S. (2016) Ibrutinib inhibits CD20 upregulation on CLL B cells mediated by the CXCR4/SDF-1 axis. Blood, Vol. 128, No. 12, pp. 1609-13.
- Pawlowski, A., Jansson, M., Sköld, M., Rottenberg, M.E. ,Källenius, G. (2012) Tuberculosis and HIV co-infection. PLoS Pathog, Vol. 8, No. 2, p. e1002464.
- Poongavanam, V., Kongsted, J. (2013) Virtual screening models for prediction of HIV-1 RT associated RNase H inhibition. PloS one, Vol. 8, No. 9, p. e73478.
- Prosperi, M., De Luca, A. (2011) Computational models for prediction of response to antiretroviral therapies. AIDS reviews, Vol. 14, No. 2, pp. 145-53.
- Prosperi, M.C., Altmann, A., Rosen-Zvi, M., Aharoni, E., Borgulya, G., Bazso, F., Sönnerborg, A., Schülter, E., Struck, D. ,Ulivi, G. (2009) Investigation of expert rule bases, logistic regression, and non-linear machine learning techniques for predicting response to antiretroviral treatment. Antivir Ther, Vol. 14, No. 3, pp. 433-42.
- Riemenschneider, M., Heider, D. (2016a) Current Approaches in Computational Drug Resistance Prediction in HIV. Current HIV research, Vol. 14, No. 4, pp. 307-15.
- Riemenschneider, M., Senge, R., Neumann, U., Hüllermeier, E. ,Heider, D. (2016b) Exploiting HIV-1 protease and reverse transcriptase cross-resistance information for improved drug resistance prediction by means of multi-label classification. BioData mining, Vol. 9, No. 1, p. 10.
- Saini, V., Marchese, A. ,Majetschak, M. (2010) CXC chemokine receptor 4 is a cell surface receptor for extracellular ubiquitin. Journal of Biological Chemistry, Vol. 285, No. 20, pp. 15566-76.
- Samson, M., Labbe, O., Mollereau, C., Vassart, G. ,Parmentier, M. (1996) Molecular cloning and functional expression of a new human CC-chemokine receptor gene. Biochemistry, Vol. 35, No. 11, pp. 3362-7.
- Sander, O., Sing, T., Sommer, I., Low, A.J., Cheung, P.K., Harrigan, P.R., Lengauer, T. ,Domingues, F.S. (2007) Structural descriptors of gp120 V3 loop for the prediction of HIV-1 coreceptor usage. PLOS Comput Biol, Vol. 3, No. 3, p. e58.
- Scherzer, R., Estrella, M., Yongmei, L., Deeks, S.G., Grunfeld, C. ,Shlipak, M.G. (2012) Association of tenofovir exposure with kidney disease risk in HIV infection. AIDS (London, England), Vol. 26, No. 7, p. 867.
- Singh, O., Su, E.C.-Y. (2016) Prediction of HIV-1 protease cleavage site using a combination of sequence, structural, and physicochemical features. Bmc Bioinformatics, Vol. 17, No. 17, p. 279.
- Slimani, H., Charnaux, N., Mbemba, E., Saffar, L., Vassy, R., Vita, C. ,Gattegno, L. (2003) Interaction of RANTES with syndecan-1 and syndecan-4 expressed by human primary macrophages. Biochimica et Biophysica Acta (BBA)-Biomembranes, Vol. 1617, No. 1, pp. 80-8.
- Srisawat, A., Kijsirikul, B. (2008) Combining classifiers for HIV-1 drug resistance prediction. Protein and peptide letters, Vol. 15, No. 5, pp. 435-42.
- Struyf, S., Menten, P., Lenaerts, J.P., Put, W., D’Haese, A., De Clercq, E., Schols, D., Proost, P. ,Van Damme, J. (2001) Diverging binding capacities of natural LD78â isoforms of macrophage inflammatory protein‐1â to the CC chemokine receptors 1, 3 and 5 affect their anti‐HIV‐1 activity and chemotactic potencies for neutrophils and eosinophils. European journal of immunology, Vol. 31, No. 7, pp. 2170-8.
- Tebas, P., Stein, D., Tang, W.W., Frank, I., Wang, S.Q., Lee, G., Spratt, S.K., Surosky, R.T., Giedlin, M.A. ,Nichol, G. (2014) Gene editing of CCR5 in autologous CD4 T cells of persons infected with HIV. New England Journal of Medicine, Vol. 370, No. 10, pp. 901-10.
- Wawer, M.J., Gray, R.H., Sewankambo, N.K., Serwadda, D., Li, X., Laeyendecker, O., Kiwanuka, N., Kigozi, G., Kiddugavu, M. ,Lutalo, T. (2005) Rates of HIV-1 transmission per coital act, by stage of HIV-1 infection, in Rakai, Uganda. Journal of Infectious Diseases, Vol. 191, No. 9, pp. 1403-9.
- Wei, Y., Li, J., Chen, Z., Wang, F., Huang, W., Hong, Z. ,Lin, J. (2015) Multistage virtual screening and identification of novel HIV-1 protease inhibitors by integrating SVM, shape, pharmacophore and docking methods. European journal of medicinal chemistry, Vol. 101, pp. 409-18.
- Wiskerchen, M. ,Muesing, M.A. (1995) Human immunodeficiency virus type 1 integrase: effects of mutations on viral ability to integrate, direct viral gene expression from unintegrated viral DNA templates, and sustain viral propagation in primary cells. Journal of virology, Vol. 69, No. 1,
pp. 376-86. - Xuan, S., Wu, Y., Chen, X., Liu, J. ,Yan, A. (2013) Prediction of bioactivity of HIV-1 integrase ST inhibitors by multilinear regression analysis and support vector machine. Bioorganic & medicinal chemistry letters, Vol. 23, No. 6, pp. 1648-55.
- Yan, A., Xuan, S. ,Hu, X. (2012) Classification of active and weakly active ST inhibitors of HIV-1 integrase using a support vector machine. Combinatorial chemistry & high throughput screening, Vol. 15, No. 10, pp. 792-805.
- Zazzi, M., Cozzi-Lepri, A. ,Prosperi, M.C. (2016) Computer-Aided Optimization of Combined Anti-Retroviral Therapy for HIV: New Drugs, New Drug Targets and Drug Resistance. Current HIV research, Vol. 14, No. 2, pp. 101-9.
- Zazzi, M., Incardona, F., Rosen-Zvi, M., Prosperi, M., Lengauer, T., Altmann, A., Sonnerborg, A., Lavee, T., Schülter, E. ,Kaiser, R. (2012) Predicting response to antiretroviral treatment by machine learning: the EuResist project. Intervirology, Vol. 55, No. 2, pp. 123-7.
- Zhen, A. ,Kitchen, S. (2013) Stem-cell-based gene therapy for HIV infection. Viruses, Vol. 6, No. 1, pp. 1-12.


