1Department of Biochemistry, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia.
2Biology Department, Faculty of Science, King Abdulaziz University, Jeddah, Saudi Arabia.
Corresponding author email: smkhojah@kau.edu.sa
Article Publishing History
Received: 28/12/2021
Accepted After Revision: 25/03/2022
Inhibition of carbohydrate digestive enzymes by natural and non-toxic secondary products had less adverse effects than synthetic drugs. This study was aimed to inhibition of α-amylase activity to a significant level by some bacterial and plant extracts which decrease the digestion of carbohydrates, obesity, and diabetes side effects. Bacteria were isolated from soil and fermented milk and the most active isolate in inhibition of α-amylase was selected and identified. Also, more than ten plants were collected, extracted and screened for inhibition of α-amylase. Out of 30 bacterial isolates were tested as inhibitor for α-amylase in vitro, ten isolates showed inhibition of the α-amylase.
Furthermore, different aqueous and organic plant extracts were investigated as inhibitors of α-amylase. The active plants in α-amylase inhibition were rosemary, garlic, lepidium, white been, cumin, coffee peel, linseed, green tea, cinnamon, and chili pepper. The isolate Lactobacillus and Streptococcus extracts showed the highest enzyme inhibition compared to the other bacterial isolate while Bacillus sp. had the lowest inhibitory enzyme activity.
Also, coffee peel aqueous extracts cause the highest inhibition of α-amylase enzyme and the lowest activity compared to the other plant extracts (70.23%). In addition, it was found that the methanolic extracts rosemary, cumin and green tea completely inhibited the enzyme (100%) while linseed and cinnamon had lower inhibitory activity of α-amylase compared to the other plant extracts (2.38% and 3.57%, respectively). In conclusion, the increase uptake of sugars cause obesity and some plant and bacterial extracts can be used to inhibit α-amylase which treat obesity, and diabetes with minimal side effects.
Inhibition, Α-Amylase, Medicinal Plants, Extracts, Lactobacillus
Alqahttani N. A, Khojah S. M, Aburas M. M. A. Inhibition of α Amylase Activity by some Bacterial and Medicinal Plant Extracts In Vitro. Biosc.Biotech.Res.Comm. 2022;15(1).
Alqahttani N. A, Khojah S. M, Aburas M. M. A. Inhibition of α Amylase Activity by some Bacterial and Medicinal Plant Extracts In Vitro.. Biosc.Biotech.Res.Comm. 2022;15(1). Available from: <a href=”https://bit.ly/3ufSTrf”>https://bit.ly/3ufSTrf</a>
Copyright © This is an Open Access Article distributed under the Terms of the Creative Commons Attribution License (CC-BY). https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provided the original author and sources are credited.
INTRODUCTION
Human consumption of increased amounts of fats and polysaccharides which contained low micronutrients like vitamins and minerals and, at the same time, low physical activity had significant risk factors and led to obesity. Starch, bread, rice and sweets provide the body with the greatest amount of energy and in the developing courtiers, most of human diets is polysaccharides. The high uptake of these materials causes obesity which makes the body at risk of diabetes and other diseases. The famous health disorders, overweight and obesity are obtained due to an imbalance between energy intake and uptake and had significant risk factors for many chronic diseases as heart disease and cardiovascular diseases, type 2 diabetes, dyslipidemia, hypertension, and certain types of cancer like breast, and colon cancer (Barrett and Udani, 2011, Mahmood, 2016, Buchholz and Melzig, 2016, Moses et al., 2020).
Amylase history began in 1811 by Kirchhoff, when it was discovered as starch degrading enzyme α-amylase and α- glucosidase, which are the key enzymes in food digestion. Various studies have reported different materials for the significant inhibition of these enzymes to decrease digestion of carbohydrates. Inhibition of carbohydrate digestion can decrease their absorption and promote weight loss. The endoenzyme α-amylase is called 1,4-glucan-4-glucanohydrolase and considered one of the major products of the pancreas (about 5–6%) and salivary glands and acts on internal α-glycosidic bonds of polysaccharides ). The amylase enzyme has been detected in different microorganisms and many plants and have a significant role in the digestion of starch and glycogen through the hydrolysis of α-1,4-glycosidic linkage in starch, amylose, amylopectin, glycogen, and various malto oligosaccharides (Barrett and Udani, 2011; Feng et al., 2011; Prakash and Jaiswal, 2010; Sales, 2012; Mahmood, 2016; Sales, 2012, Mahmood, 2016).
Alpha-amylase inhibitors are widely used by the pharmaceutical and agricultural industries. In comparison, this enzyme’s inhibitors can be used to lower blood glucose and in the treatment of obesity, adipose, hyperlipemia, diabetes, pre- diabetes, gastric ulcer, duodenal ulcer, and as insect control. Natural α-amylase inhibitors are distributed in microorganisms, higher plants, or animal’s secretions. Microorganisms represent a vast resource of novel compounds with many agricultural and medical applications. Natural inhibitors of α-amylase attract considerable interest as excellent sources of bioactive compounds due to their longtime clinical practice and reliable therapeutic efficacy. Some extracts from the microbial origin like members of the family Bacillaceae and Streptomycetaceae were reported as α-amylase inhibitors (Obiro et al., 2008; Feng, et al., 2011; Liu et al., 2015; Sun et al., 2015; Moses et al., 2020).
Moreover, from the yeast cells, a functional α-amylase inhibitor was detected and purified from culture the culture supernatant using a two-step chromatographic method and can be used for biotechnological and pharmaceutical applications. Similarly, some plant extracts are sources of α-amylase inhibitor, including White been, Cumin and Rosemary. There are many types of plants reported to show α-amylase inhibitory activity, and so maybe relevant to the treatment of type 2 diabetes and obesity. About 800 plant species have been antidiabetic properties. A wide range of plant-derived principles belonging to compounds, mainly alkaloids, glycosides, galactomannan gum, polysaccharides, hypoglycins, peptidoglycans, guanidine, steroids, glycopeptides, and terpenoids, have demonstrated bioactivity against hyperglycemia (Sales, 2012; Brain-Isasi et al., 2017; Moses et al., 2020).
Among the new chemical structure approved as drugs by the U.S. Food and Drug administration, a total of 46% are relevant to natural products, including 4% natural products, 22% natural-product derivatives, and 20% natural mimic compounds (Liu et al., 2015). In a survey conducted in 2017, the results indicated that α-amylase could inhibit glycoprotein in common beans (Phaseolus vulgaris L). Another study in 2016 performed on Basil seed was found to contain bioactive peptides, which have antioxidative and a-amylase inhibitory activities, and three novel inhibitor peptides successfully identified.
These peptides can use therapeutic agents to reduce the risk of oxidative stress and prevent type-2 diabetes (Saufi, 2016). In another study performed on the essential oil from Cedrus deodara cones in 2017 reported that this evergreen tree was distributed in western Himalaya also found abundantly in Asia, including China, Afghanistan, Pakistan, India, and Nepal. The results indicated that cones essential oils act as a potential α-amylase inhibitors due to the presence of long pinene which is the primary active component in the essential oil from Cedrus deodara cones (Xu et al., 2017).
Another study was conducted in 2017, it discover the bioactive peptides derived from cumin seed, with antioxidative and antidiabetic activities (Siow et al., 2017). In summary, most of the studies approved that there are several plants that have chemical structures and bioactive compounds that inhibit amylase enzyme that leads to development for food and pharmaceutical applications (Moses et al., 2020).
The present research aimed to analyze the inhibition of amylase enzyme by specific bacterial and some medicinal plant extracts, and to help the selection of the most active one that inhibits the enzyme.
MATERIALS AND METHODS
α-amylase enzymes Kit was purchased from α-amylase liquid color Human Gesell schaft, Germany Kit and it is stored at 2-4°C.
Soil and fermented milk samples: In this study, 5 soil samples of agricultural soil, approximately 5 g each, were obtained from different sites which included: Qurayyat Governorate, Taif, and Abha, Saudi Arabia in addition to Jumeirah Beach in Dubai, United Arab Emirates. of each sample was collected in a sterile plastic package and transferred to the Microbiology laboratory at King Abdulaziz University to isolate bacteria. Five samples from the fermented milk were collected from the supermarket to isolate lactic acid bacteria.
Plants samples: Ten kinds from plants were selected and used to inhibit amylase enzyme, which included: rosemary, garlic, Lepidium, white been, cumin, coffee peel, linseed, green tea, cinnamon, and chili pepper. They were collected from the local market in Jeddah, Saudi Arabia. Approximately 10 g of each plant was taken to be extracted with water or methanol. The extracts were saved in the refrigerator at 4°C before testing.
Isolation and characterization of bacteria: The medium used for bacteria isolation was nutrient agar medium which containing (g/l) peptone (5.0), beef extract (3.0), and sodium chloride (5.0), pH 7.4 at 25°C. Agar 15.0 g/L was added to prepare solid medium (Gowsalya et al., 2014). Lactobacillus was isolated from AL-Maraee yogurt using de Man, Rogosa and Sharpe medium (MRS) at pH 6 under anaerobic condition (10% CO2) and 37°C. All isolated bacteria were stored on Slant agar in a refrigerator at 4ºC to be used a later (Carrim, 2006).
Finally, ten pure bacteria samples were isolated, and they were used for inhibition of amylase. The morphology, physiology and biochemistry of the bacterial colonies like shape, color microscopic appearances and Gram’s type were recorded as described by Cheesbrough (2005). API20 was used for Gram negative bacterial isolates. Electron microscope and molecular method using 16SrRNA were used to study the bacterial isolate B11(Al-Haik et al., 2017a, b).
Bacterial growth in liquid medium: Nutrient broth or MRS broth media (50 ml) were inoculated with 0.1 ml (4×104 CFU/ml) of each bacterial suspension in 250 ml Erlenmeyer flasks, incubated at 37°C for 2 days at 120 rpm. At the end of the fermentation period, each bacterial filtrate was collected, sterilized by 0.45 µm bacterial filter and preserved at 4°C until used for amylase assay.
Preparation of plants extract: The selected plants, used for inhibition of amylase enzyme, included: rosemary, garlic, Lepidium sativum, white been, cumin, coffee peel, linseed, green tea, cinnamon, and chili pepper. Plants were ground to a fine powder. Then, they were stored at room temperature and protected from light until extraction. Extraction of samples with water was carried out where 5g of the powdered plant parts were mixed with 50mL of boiling deionized water for 24h at room temperature. Then, the aqueous solution was filtrated and lyophilized immediately and stored dry at 4°C. It was protected from the light. A refluxed condenser was then prepared with methanolic extracts by extracting 5g powdered plants with 50ml of methanol for 24 h. After that, the extract was filtrated, dried and stored in the refrigerator at 4°C until used for amylase assay.
Enzyme assay procedure: Inhibition of α-amylase was measured based on the hydrolytic cleavage of a modified starch derivative material (Buchholz and Melzig, 2016). By micropipette, 1000 μl of enzyme reagent kit was put in the cuvette and mixed with 10 μl of the sample solution (bacteria filtrate, aqueous or methanolic plant extracts). The mixture was incubated for 2-3 min at 37°C and finally, the absorbance was detected at 405 nm (A405 nm). Distilled water was used as a positive control and all testes were carried out in triplicate and mean value was calculated.
The enzyme activity was determined by the increase of fluorescence per minute. The inhibitory activity of the extracts was the difference between the enzyme activity in the 100% activity control (no inhibitor added) and the enzyme activity in the reaction mixture containing the bacteria filtrate or plant extract and was expressed as a percentage of the enzyme activity of the positive control. Then, each extract was tested for the inhibitory activity. The results were expressed as the average ± standard deviation (Buchholz and Melzig, 2016).

Statistical Analysis: The results were obtained as the average (mean value) and standard deviation. The results are expressed as mean ± SD (n =3) of three experiments. Statistical Analysis was performed using S.P.S.S. for Windows, the means of results were compared by one-way Analysis of variance (ANOVA), and the significant level was P<0.05 (Orhan, and Akincioglu, 2020).
RESULTS AND DISCUSSION
Alpha-amylase is an enzyme which hydrolyzes carbohydrate into monosaccharides then into glucose, and in advance, it is easily absorbable. From one of current methods for prevention of obesity is reduction of caloric intake during the food digestion and absorption, and inhibition of α-amylase is important for prevention of starch hydrolysis to small parts such as maltose, maltotriose, and maltotetraose. Inhibition of α-amylase enzymes is one of the most widely studied for the treatment of obesity that was became one of the most common problems in the world (Kellogg et al., 2014, Mahmood, 2016; Buchholz and Melzig, 2016; Rasouli et al., 2017).
Analyzing the inhibition of α-amylase is required because they stimulate the absorption of glucose and the associated postprandial hyperglycemic spike. Alpha amylase inhibition is a policy in diabetes administration as it can manage the serum glucose level (Hidayati et al., 2018). In this study, a colorimetric method, based on 2-cloro-4-nitrofenil-maltotriosido (CNPG3) which mainly react with 𝛼- amylase, forming 2, 2 dichloro-4-nitrophenol from the substrate, resulting in the increase in the absorbance/ minute. There is a directly proportional between the measured absorbance and the concentration of 𝛼-amylase, found in the tested sample.
In this study, the selected bacteria on nutrient agar were characterized based on cell shape and colony shape, elevation, size and diameter (Figure1). Some bacterial colonies were white, yellowish white, orange or pink on nutrient agar while on MRS agar they were white or yellowish white. The elevation of the colony was smooth, flat, or serrated. The colonies diameter was ranged from 1.0 to 8.0 mm, and some the edge was round or irregular. Similarly, five bacterial isolates were obtained from fermented milk on MRS agar. All bacterial isolates were selected for further screening for inhibition of amylase enzyme. Table 1 showed the source of the isolated bacteria, colony color, cell shape, presence or absence of endospore and gram reaction.
Figure 1: The isolated bacteria obtained from collected soil on nutrient agar medium

Table 1. The selected bacterial isolates from different sources
| Soil sample | Source | Shape | Color | Medium used | Endospore | Gram stain | Isolate identification |
| B1S1 | Fermented milk | Bacilli | White | MRS agar | Absent | -ve | Lactobacillus |
| B1S4 | Taif soil | Bacilli | Orange | Nutrient agar | Present | +ve | Bacillus |
| B1S5 | Jumeirah Beach soil | Bacilli | White | Nutrient agar | Absent | -ve | E. coli |
| B2S2 | Taif soil | Bacilli | Yellow | Nutrient agar | Absent | -ve | Pseudomonas |
| B2S3 | Abha soil | Bacilli | Pink | Nutrient agar | Present | +ve | Bacillus |
| B2S4 | Abha soil | Bacilli | White | Nutrient agar | Present | +ve | Bacillus |
| B3S5 | Fermented milk | Bacilli | White | MRS agar | Absent | -ve | Lactobacillus |
| B4S3 | Qurayyat soil | Cocci | Yellow | Nutrient agar | Absent | -ve | Proteus vulgaris |
| B5S1 | Fermented milk | Bacilli | White | MRS agar | Absent | -ve | Lactobacillus |
| B5S3 | Fermented milk | Cocci | White | MRS agar | Absent | -ve | Streptococcus |
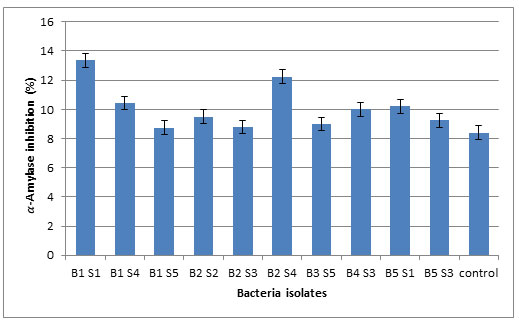
Based on the data presented in Table 2, the filtrates of the ten tested bacterial isolates had considerable potential in producing 𝛼- amylase inhibitor and each isolate of bacteria inhibited enzyme at different rates. Some bacterial filtrates showed good inhibition of enzyme activity like the isolate B1S1 which had the highest inhibition (13.4 ± 0.07) while lower inhibition were recorded for the isolates B1S5 (8.75 ± 2.19), B2S3 (8.8 ± 0.28), B3S5 (9 ± 1.13), B5S3 (9.3± 0.07), B2S2 (9.5 ± 0.42), B4S3(10 ± 1.83), B5S1 (10.2 ± 0.42), B1S4 (10.5 ± 1.06) and B2S4 (12.3 ± 3.04). Based on analysis of variance (ANOVA) results, significant differences between the values of enzyme inhibitory significant differences between the values of enzyme inhibitory for all bacteria isolates compared to the control or isolate B1S1(P<0.05).
Table 2. Inhibition percentage of 𝛼-amylase enzyme by the isolated bacterial filtrates.
Data represent means ± SD; P<0.05 (n=3).
| Bacteria | 𝛼-Amylase inhibition (%) | Bacteria | 𝛼-Amylase inhibition (%) |
| B1S1 | 13.4 ± 0.07* | B2S4 | 12.3 ± 3.04* |
| B1S4 | 10.5 ± 1.06*# | B3S5 | 9.0 ± 1.13*# |
| B1S5 | 8.7 ± 2.19*# | B4S3 | 10.0 ± 1.830*# |
| B2S2 | 9.5 ± 0.42*# | B5S1 | 10.2. ± 0.42*# |
| B2S3 | 8.8 ± 0.28*# | B5S3 | 9.3± 0.07*# |
| Control | 8.4 ± 0.14 | ||
| *: significant results compared to control, #: significant results compared to isolate B1S1 | |||
Figure 2: The percentage of inhibition (%) of α-amylase by the selected bacterial filtrates.
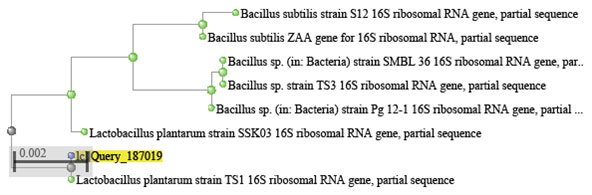
The bacterium that showed the highest inhibitory enzyme activity compared to 9 other isolates was selected and identified. Morphological characterization of the bacterial isolate B1S1was determined. It was gram negative long chain bacilli, microaerophilic and non spore forming bacteria. The identification was confirmed using molecular methods. Bacterial DNA was separated and 16Sr RNA was analyzed and the obtained sequence was compared to related genera at Gene bank data base. The isolate B1S1 was confirmed as species belonging to the genus Lactobacillus. It was identified as L. plantarum B1S1 with similarity level of 95% to strains L. plantarum SSK03 and L. plantarum strain TS1and of 90% to Bacillus subtilis strain S12, Bacillus subtilis ZAA, Bacillus sp. strain Pg 12-1.
Figure 3: The growth of the isolate B1S1 on MES agar medium, D: examined
under electron microscope, C: After Gram staining

Table 3. The biochemical characters of the isolate B1S1
| Tests | Substrate degradation | Results |
| ADH | Araginine | + |
| LDC | Lysine | – |
| ODC | Ornithine | – |
| CIT | Citrate | – |
| H2S | Na Thiosulfate | – |
| URE | Urea | – |
| TDA | Tryptophan | – |
| VP | Na Pyruvate | – |
| GEL | Charcoal gelatin | + |
| INO | Inositol | – |
| AMY | Amygdalin | + |
| ARA | Arabinose | – |
| OX | Oxidase | – |
| IAA | Indole production | – |
| +: positive results, -: negative results | ||
Figure 4 : The polygenetic tree of the isolate B1S1 and the closely related strains.
The bacterial filtrates were evaluated as inhibitors of α-amylase. From bacteria, Lactobacillus isolate showed the highest inhibitory enzyme compared to other isolates. The two isolates, Bacillus sp. and E. coli had the lowest inhibitory enzyme activity. Lactobacillus belong to Gram positive, microaerophilic, long rod, non spore forming bacteria.
Genus Lactobacillus had more than 25 taxonomic genera and 250 species. Lactobacillus is the main agent of human and animal microbiota as it was recorded in the female genital organ like vagina in addition to digestive system. It is a probiotic bacteria can be isolated from yogurt, cheese and fermented milk and has varied application where it protects human body against pathogens and treat diarrhea, vaginal infections, and skin disorders (Allam et al., 2016; Al-Zahrani et al., 2019).
The utilization of microbes in the production of metabolites to cure diabetes is urgent and necessary. The potentially used microbes, in this case, are some fungi responsible in the biosynthesis of secondary metabolites that inhibit alpha-amylase. They used ethyl acetate extract of these fungi to inhibit alpha-amylase activity and three of them had high inhibition percentages (14.385%, 12.849%, and 39.246%).
In another study, α amylase inhibitory assay showed that whey of both cow milk and buffalo milk fermented by L. delbeurkii had higher antidiabetic activity than L. lactis (Vankudre et al., 2015). Moreover, eleven different α-amylase inhibitors with maximum activity of 72% were recorded (Sri Pujiyanto et al., 2018; Hidayati et al., 2018). They added that presence of starch or lactose in growth medium or adjusting the initial pH at pH 5.0 or 6.0 affect significantly the production of α-amylase inhibitors (P <0.05).
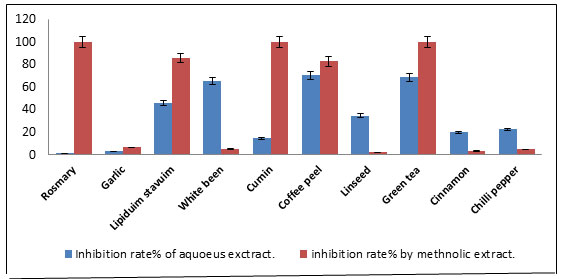
The used plants for amylase inhibition, their common names, scientific names and used parts were summarized in Table 4. The plants were extracted with boiling water or with methanol. The inhibition rates of the two extracts of the tested plants for α-amylase have been determined. Inhibition of α-Amylase is expressed in percentage (%). Based on Figure 5, the results of amylase inhibition indicated that all aqueous extracts of the tested plants showed that α -amylase was inhibited in different proportions and the values were in the range of 1.19 % to 70.23%. Also, it was found that the aqueous extract of coffee peel had the highest inhibitory enzyme activity (70.23%) compared to the other plant extracts and it is followed by green tea (68.45%) and white beans (65.47%).
Rosemary was found to have lower inhibitory enzyme activity compared to other plant extracts (1.19 %). Furthermore, Lepidium and Linseed extracts showed partial inhibition. The results in Figure 6 showed that the methanol extract had results completely different as it was found that three plant extracts showed complete inhibition of the enzyme (100%), so that there was no apparent hydrolysis of the starch.
They were rosemary, cumin and green tea. Linseed and cinnamon had lower inhibitory enzyme activity compared to the other plant methanol extracts (2.38%) and (3.57%). α-Amylase inhibition by water extract and methanol extracts were compared. The data from methanolic extracts of the tested plants have a strong inhibitory effect than aqueous extract (Table 5. Figure 7).
Also, it was noticed that generally methanolic extracts were more active and extracts of rosemary and cumin showed the highest activities while water extracts of Green tea, Coffee peel and White beans showed excellent α-Amylase inhibition. It was found that coffee peel in the aqueous extract was the best compared to other extracts (70.23%), while in methanol extract it was not the best. Rosemary, cumin, and green tea were the most enzyme inhibitors in methanol extract (100%). Moreover, rosemary is lowest inhibitory in the aqueous extract (1.19%).
The inhibition rate of green tea in both extracts was high (in methanol extract was 100% while in aqueous extract was 68.45%). Based on the results of the analysis of variance (ANOVA), significant differences were indicated between the values of enzyme inhibitory for all water or methanolic plant extracts (P<0.05(. Also, significant differences were found between water and methanol extracts of all tested plants. Nowadays, people still believe that plants can be used as alternative medicines as bay leaves cure diabetes.
Also, many researcher and nutritionist are extremely interested to fabricate a novel nutritional approach to perfectly control the postprandial glycaemia without inducing negative circumstances on the digestive system. Thus, medicinal plants were gained special importance for the treatment of diabetes (Gislin et al., 2018). In many third world countries, plant extracts are traditionally used and have been accepted by the users for the treatment of diabetes (Hidayati et al., 2018, Orhan and Akincioglu, 2020).
In this study, the 10 plants extracts either aqueous and methanol extracts were screened using fluorescence-based in vitro enzyme assays to provide plants with potential α-amylase inhibitory activities. It was found that coffee peel had the highest inhibitory for enzyme in aqueous extract (70.23%), while in methanol extracts rosemary, green tea and cumin gave complete inhibition of the enzyme (100%). As it is well known, the most popular and consumed beverage in the world is coffee.
In 2014, amount consumed approximately 17.8 billion packages of coffee bought in common food stores. Coffee peel or coffee husk is one of the main components of coffee which is a thin layer that directly covers the coffee seeds. Coffee peel composed high amount of dietary fiber which contain mainly of cellulose, and hemicellulose and the last composed mainly of xylose. It is also rich in protein and minerals such as potassium, magnesium and calcium and many other components (Bessada et al., 2018).
Coffee peels contributed significantly to the inhibitory potential of the extract due to the presence of bioactive compounds such as: chlorogenic acids (1-6%), caffeine (0.8-1.25%), melanoid (17-23%) and other antioxidants. The presence of the bioactive compounds was proven vital activity in prevention skin aging. Moreover it considered anti-microbial, anti-inflammatory, anti-hair loss activities and protection against UV damage. There is another use for coffee peel, as in 2014 there was a study of an antioxidant beverage for body weight control made on a powdered coffee peel which consists of 1.37% caffeine and 1.1-3.0% chlorogenic acids with low concentrations of reducing sugars and high fiber and also it had low glycemic index. This beverage was suitable for obese and diabetic people (Bondesson, 2015; Bessada, et al., 2018).
Different secondary plant metabolites had potential inhibition activity for α-amylase and hence maybe used as therapeutic drugs. Some species of the genus Teucrium showed excellent α-amylase inhibitory activities compared to the standard inhibitor, Acarbose. The inhibitory activities of α amylase was higher for Teucrium polium extract compared to T. oliverianum extract which showed more inhibition actively than T. Orientale. The IC50 of the alcoholic extracts of T. polium, T. oliverianum and T. orientale were 3.63, 3.86 and 13.93 mg/ml, respectively.
Moreover, the organic plant extracts had strong inhibitory activity on the α-amylase activity. However, the dichloromethane extract showed the lowest inhibitory strength. Thus, α-amylase inhibition by these plant extracts could provide a successful use of these plants as useful drugs. Also, Hussain et al. (2018) concluded that aqueous extracts of Amomum subulatum and Amomum tsako dry fruit constitutes (seeds and rind) have the significant inhibitory activity of α-amylase due to the occurrence of plant secondary products like polyphenol, flavonoids, alkaloids, saponins, and tannins.
Table 4. The used plants for amylase inhibition, their common and scientific names and the used parts.
| Common name | Scientific name | Used part |
| Rosemary | Salvia Rosmarinus | Leaves |
| Garlic | Allium sativum | Powder |
| Lepidium | Lepidium sativum | Seeds |
| White beans | Phaseolus vulgaris | Seeds |
| Cumin | Cuminum cyminum | Seeds |
| Coffee peel | Coffea arabica | Peel of the seeds |
| Linseed | Linum usitatissimum | Seeds |
| Green tea | Camellia sinensis | Leaves |
| Cinnamon | Cinnamomum zeylanicum | Park |
| Chili pepper | Capsicum annuum | Dried fruit |
Figure 5: Inhibition of α amylase enzyme by some aqueous plant extracts.
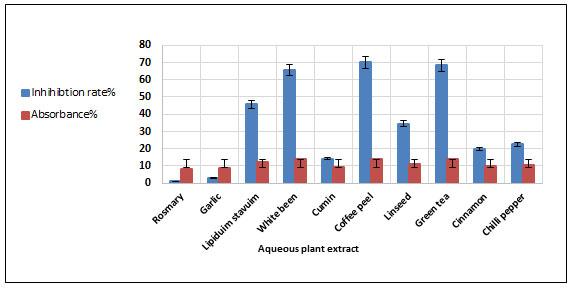
Figure 6: Inhibition of amylase enzyme by some methanolic plant extracts
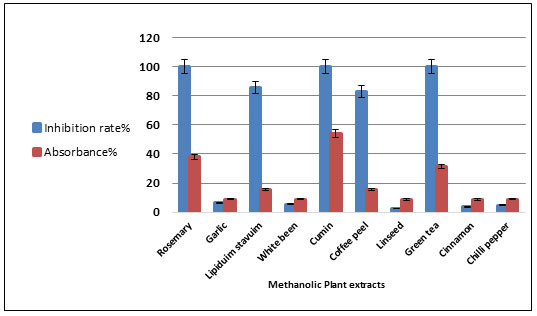
Table 5. Percentage of inhibition of 𝛼-amylase (%) by the tested aqueous and
methanolic plant extract, data represent means ± SD.
|
Medicinal plants |
Aqueous extract (control)
|
Methanol extract | ||
| Absorbance (%)
|
α-Amylase
inhibition (%) |
Absorbance (%) | α-Amylase
inhibition (%) |
|
| Rosemary | 8.5± 2.68 | 1.19 | 37.95 ± 1.35 | 100* |
| Garlic | 8.65 ± 1.48 | 2.97 | 8.95 ± 0.85 | 6.54* |
| Lepidium sativum | 12.25 ± 0.77 | 45.83 | 15.6 ± 2 | 85.71* |
| White beans | 13.9 ± 0.70 | 65.47 | 8.85 ± 0.95 | 5.35* |
| Cumin | 9.6 ± 0.98 | 14.28 | 54.85 ± 6.25 | 100* |
| Coffee peel | 14.3 ± 0.28 | 70.23 | 15.35 ± 1.85 | 82.73* |
| Linseed | 11.3 ± 3.25 | 34.52 | 8.6 ± 0.9 | 2.38* |
| Green tea | 14.15 ± 2.89 | 68.45 | 31.45 ± 3.25 | 100* |
| Cinnamon | 10.05 ± 1.21 | 19.64 | 8.7± 0.2 | 3.57* |
| Chili pepper | 10.3 ± 3.81 | 22.61 | 8.8 ± 1.5 | 4.67* |
* indicates significant difference at P <0.05 compared to the control.
Figure 7: Inhibition of α-amylase enzyme by aqueous and methanol plant extracts.
Numerous α-amylase inhibitors were purified from some medicinal plants and are used as alternative drugs with good activity and less side effects compared to synthetic products. Active compounds like phenolic substances derived from the medicinal plants are a source of α-amylase and α-glycosidase inhibitors. The seeds of the terrestrial herb, Amomum which belongs to family Zingiberaceae and found in tropical area are used in traditional medicine.
The methanol extract of Amomum tsaoko had markedly influence on plasma glucose and thiobarbituric acid reactive substances and antioxidant potential. So, some plant extracts and some types of bacteria are considered useful factors for preventing or treating obesity. However, more studies are needed to develop new plant and bacterial resources to treat obesity with minimal side effects (Matsuda et al., 2002; Longguan 2008; Cicero et al., 2013; Pujiyanto et al., 2018; Moses et al., 2020).
Inhibition of α-amylase by natural resources is a successful method in preventing obesity and its associated diseases more than synthetic compounds and slimming drugs. Therefore, traditional medicines, including herbal medicine, have a great way in this regard (Mahmood, 2016, Buchholz and Melzig, 2016). In summary, it could be stated that the advantage of carbohydrate digestive enzyme inhibitors by plant extracts consists in not causing severe side effects compared to synthetic compounds and slimming drugs.
So, it may be beneficial in weight loss in individuals consuming large amounts of starch, or had overweight, obesity or diabetes (Mahmood, 2016). Based on the results of Gulati et al., (2018), natural products are particularly best compared to the other oral drug or anti-diabetes agents currently available which recorded many side effects. In this study, Lactobacillus and/or some water or organic extracts of some medicinal plants showed excelled inhibition of α-amylase, thus they must be chosen for more detail studies and detection of the active product which can be used safely to treated obesity.
CONCLUSION
The metabolic disorder, Diabetes mellitus is associated with high level of blood glucose which is obtained due to insufficiency of insulin production or action. Inhibition of α-amylase can be an essential strategy in the management of postprandial blood glucose levels in patients with type II diabetes. Inhibition of α-amylase using some plant extracts increases levels of undigested starch in the colon and prevents the degradation of polysaccharides which decreases the blood sugar. These data may be beneficial in further exploring natural sources having pharmaceutical applications, as natural products have the advantage of being safe in their activity and have lower side effects.
Conflict of Interest: There is No Conflict of Interest.
Data Availability Statement: The database generated and /or analysed during the current study are not publicly available due to privacy, but are available from the corresponding author on reasonable request.
REFERENCES
Al-Haik, W, Abu Zinadah, GM. S, Bokhari, F M., Aly, M M. (2017a). Inhibition of Fungal Growth and Mycotoxin Productions Using Lactic Acid Bacteria Isolated From Milk and Fermented Food. IOSR Journal of Pharmacy and Biological Sciences Vol.12 (4) 122-129.
Al-Haik, W., Bawazir A., Aly M.M, Al-Haddad, A.M. and Shantaram, M. (2017b). Antimicrobial Activity of Lactic Acid Bacteria against Toxigenic Fungi. International Journal of Current Research and Academic Review, Vol. 5 (11): 12-18.
Allam, R.F., Aly M.M., El-zhrany, K.M., Shafei, M.S. (2016). Production of β-Galactosidase enzyme from Lactobacillus acidophilus RK isolated from different sources of milk and dairy products . International Journal of ChemTech Research (IJCRGG), Vol.9, No.10, pp 218-231
Al-Zahrani, K., Allam R, Amasha R., Jastaniah S. and Aly M. (2019). Isolation and identification of Lactobacillus plantarum KM5 from camelmilk for β-galactosidase production. IOSR Journal Of Pharmacy And Biological Sciences (IOSR-JPBS) Volume 14: PP 27-35
Barrett, M.L. and Udani, J.K. (2011). A proprietary alpha-amylase inhibitor from white bean (Phaseolus vulgaris): a review of clinical studies on weight loss and glycemic control. Nutr J,. 10: p. 24.
Bessada, S.M.F., Alves R. C., and Oliveira M.B. (2018). Coffee Silverskin: A Review on Potential Cosmetic Applications.. 5(1): p. 5.
Bondesson, E. 2015A nutritional analysis on the by-product coffee husk and its potential utilization in food production
Brain-Isasi, S., Álvarez-Lueje A., and Higgins T.J. (2017). Heterologous expression of an α-amylase inhibitor from common bean (Phaseolus vulgaris) in Kluyveromyces lactis and Saccharomyces cerevisiae. 16(1): p. 110.
Buchholz, T. and Melzig, M.F. (2016). Medicinal plants traditionally used for treatment of obesity and diabetes mellitus–screening for pancreatic lipase and α‐Amylase inhibition.. 30(2): 260-266.
Carrim, A.J.I. (2006). Enzymatic activity of endophytic bacterial isolates of Jacaranda decurrens Cham. (Carobinha-do-campo).. 49(3): p. 353-359
Chaineau, C.H., Rougeux G, Yepremian C, and Oudot J. (2005). Effect of Nutrient concentration on the biodegradation of crude oil and associated microbial Populations in the soil. Soil Biology and biochemistry, 1-8.
Cicero, L.T, Yenshou, L., Arlene, P.B., Yi-Chin, C., ShaoChih, C., Wen-Chin,Y. (2013). Herbal Therapies for Type 2 Diabetes Mellitus: Chemistry, Biology and Potential Application of Selected Plants and Compounds, 43: 1-33.
Etxeberria, U., de la Garza, A.L., Campión, J., Martínez, J.A., Milagro, F.I. (2012). Etxeberria, U., et al., Antidiabetic effects of natural plant extracts via inhibition of carbohydrate hydrolysis enzymes with emphasis on pancreatic alpha amylase.. 16(3): p. 269-297.
Feng, Z.H., Wang, YS. and Zheng, Y.G. (2011). A new microtiter plate-based screening method for microorganisms producing Alpha-amylase inhibitors. Biotechnol Bioproc E 16, 894.
Gislin, D., Sudarsanam D., Raj G.A. Baskar K. (2018). Antibacterial activity of soil bacteria isolated from Kochi, India and their molecular identification, Journal of Genetic Engineering and Biotechnology, Volume 16 (2): 287-294,
Gowsalya, A., Ponnusami V. and Sugumaran K.J. (2014). Isolation of bacteria from soil sample for exo-polysaccharide production. 6(5): p. 2925-2928.
Gulati, V., Harding, I.H., Palombo, E.A. (2012). Enzyme inhibitory and antioxidant activities of traditional medicinal plants: potential application in the management of hyperglycemia, 12(1): p. 77.
Hidayati, W., Padillah, A., Maharadingga, HN., Prastiwi, R., Pratiwi, A., Farahmayuni, L., Syahputra, R. and Fahrul, M. (2018). The Alpha-Amylase Inhibition Potential of Endophytic Fungi from Indonesian Bay Leaves (Eugenia polyantha WIGHT.). In Proceedings of the 1st Muhammadiyah International Conference on Health and Pharmaceutical Development (MICH- Ph. D), pages 107-111.
Hussain, S.A, Hameed, A., Fu, J., Xiao, H., Liu, Q., Song, Y. (2018). Comparative in vitro analysis of anti-diabetic activity of IndoPak black cardamom (Amomum subulatum Roxb.) and Chinese black cardamom (Amomum tsaoko Crevost et Lemaire). Progress in Nutrition; Vol. 20, N. 3: 403-414.
Kellogg, J., Grace, M. H. and Lila, M. A. (2014). Phlorotannins from Alaskan Seaweed. Carbolytic Enzyme Activity, 5277–5294.
Liu L, Cen Y, Liu, F, Yu J, Jiang X and Chen X (2015). Analysis of α-amylase inhibitor from cornifructus by coupling magnetic cross-linked enzyme aggregates of α-amylase with HPLC–MS. Journal of Chromatography B, Volumes 995–996: 64-69
Longguan, YU ( 2008). Effect of Lipid Extracted from Tsao-ko (Amomum tsao-ko Crevost et Lemaire) on Digestive Enzyme Activity, Antioxidant Activity, Plasma and Liver Lipids, and Blood Glucose Levels of Mice. Journal of Nutritional Science and Vitaminology, Vol. 54 (No. 5) P: 378-383.
Mahmood, N.J. (2016). A review of α-amylase inhibitors on weight loss and glycemic control in pathological state such as obesity and diabetes.. 25(6): p. 1253-1264.
Matsuda, H., Morikawa T., Yoshikawa M. (2002). Antidiabetogenic constituents from several natural medicines. Pure and Applied Chemistry, vol. 74, no. 7, pp. 1301-1308.
Moses, Z.Z., Sunday H.G., Jude, O.O., Jeremiah D.G. and Hassan S. M. (2020). Evaluation of alpha amylase inhibitory activity of the ethanol extract of some locally sourced plants with known hypoglycaemic properties. Journal of Biologics Research and Development Vol.1 No. 1, 351-3
Obiro, W.C., Zhang, T. and Jiang B.J. (2008). The nutraceutical role of the Phaseolus vulgaris α-amylase inhibitor. 100(1): p. 1-12.
Orhan, F. and Akincioglu H.J. (2020). Determination of carbonic anhydrase enzyme activity in halophilic/halotolerant bacteria.. 155: p. 103650
Prakash, O., Jaiswal N.J. (2010), α-Amylase: an ideal representative of thermostable enzymes.. 160(8): p. 2401-2414.
Pujiyanto, S, Resdiani M, Raharja B, Ferniah RS (2018). α-Amylase inhibitor activity of endophytic bacteria isolated from Annona muricata. J Phys Conf Ser.111-119.
Rasouli, H, Hosseini-Ghazvini, SM, Adibi, H, Khodarahmi R. (2017). Differential α-amylase/α-glucosidase inhibitory activities of plant-derived phenolic compounds: a virtual screening perspective for the treatment of obesity and diabetes. Food Funct. May 24;8(5):1942-1954
Sales, P.M. (2012).α-Amylase inhibitors: a review of raw material and isolated compounds from plant source. 15(1): p. 141-183.
Saufi, B. (2016). Antioxidative and Amylase Inhibitor Peptides from Basil Seeds. 22(1): p. 3.
Siow, H.L. and Gan, C.Y (2017). Optimization study in extracting antioxidative and α amylase inhibitor peptides from Cumin seeds (Cuminum cyminum). 41(1): p. e12280.
Sun, Z., Lu, W., Liu, P. (2015). Isolation and characterization of a proteinaceous α-amylase inhibitor AAI-CC5 from Streptomyces sp. CC5, and its gene cloning and expression. Antonie van Leeuwenhoek 107, 345–356.
Vankudre. M , Balpande A and Athale M (2015). a-amylase inhibition and Comparative analysis of antioxidant activity of whey from cow and buffalo milk fermented with Lactobacillus species. Biosci. Biotech. Res. Comm. 8(1): 25-28.
Xu, F., Gu, D., Wang, M., Zhu, L., Chu, T., Cui, Y., Tian, J., Wang, Y., Yang, Y. (2017). Screening of the potential α-amylase inhibitor in essential oil from Cedrus deodara cones. Industrial Crops and Products, Volume 103:251-256.


