Department of Mechanical Engineering, Sant Longowal Institute of Engineering and Technology, Longowal, PUNJAB: INDIA -148106
Corresponding author Email: dvijander@gmail.com
Article Publishing History
Received: 06/05/2019
Accepted After Revision: 22/08/2019
By considering economic, environmental and availability aspect, waste cooking oil was selected as a feedstock for production of green diesel. Hydro processing technique was used to obtain green diesel and Na2CO3 is used as a catalyst. The fuel properties such as specific gravity, kinematic viscosity, net calorific value and flash point were measured. The performance characteristics of green diesel were investigated in single cylinder compression ignition engine. Indicated thermal efficiency, break thermal efficiency, break specific fuel consumption (BSFC) and fuel consumption in kg/hour were investigated at different rpm and different load. The maximum indicated thermal efficiency was 87.96% at 1450 rpm and 8.97 kg load whereas maximum break thermal efficiency was 38.14% at 1410 rpm and 16.68 kg load. Minimum BSFC was 0.25 kg/kWh at 1410 rpm and 16.68 kg load whereas minimum fuel consumption was 0.42 kg/hour at 1490 rpm and 0.86 kg load. Physics of combustion of green diesel was analyzed in CI engine and compared with petro diesel. It was found that green diesel has shorter ignition delay which is an indications for good combustion. Green diesel has the high cylinder pressure peak as compared to petro-diesel. The peak pressure in case of green diesel was 72.8 bar at 376° crank angle and for petro-diesel was 72.2 bar at 376° crank angle. The combustion information was obtained from the pressure sensor located in the engine cylinder head
Waste cooking oil, Green diesel, Combustion, Ignition delay.
Kumar V. Green Diesel Production from Waste Cooking Oil: Performance Computation and Combustion Analysis at Different speeds in Single Cylinder CI Engine. Biosc.Biotech.Res.Comm. 2018;11(3).
Kumar V. Green Diesel Production from Waste Cooking Oil: Performance Computation and Combustion Analysis at Different speeds in Single Cylinder CI Engine. Biosc.Biotech.Res.Comm. 2018;11(3). Available from: https://bit.ly/2kf5UBI
Copyright © Kumar, This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
Introduction
Everybody has a desire to live in comfort and pollution free environment. To achieve comfort, human being introduced machinery in modern world such as engine. Generally engines run on fossil fuel which generates harmful emission and are ultimate cause of pollution and global warming (Ramya et al. 2012). In rapidly growing global energy consumption, diesel engine play the main role (Shameer et al. 2018). Although emission of machinery cannot be eliminated completely but a significant reduction is possible by use of green fuel or bio fuels. Green fuels have one more advantage that is these are renewable. Bio fuels have been introduced as a substitute for fossil fuels (Yu et al. 2013). (Deshpande and Kulkarni 2012) derived green fuel from Jatropha oil. Animal fats (TAG) and modified vegetable oils in nature can be used for the production of chemical products such as biofuels (Suarez et al .2007). Due to chemical and thermal instability and high viscosity, vegetable oils forms carbon deposits inside the combustion chamber of diesel engines and hence not used directly in engine (Biswas and Sharma 2013). (Buzetzki et al. 2011) used zeolite catalysts with NaY and clinoptilolite to investigate the cracking of vegetable oils (sunflower, rapeseed, jatropha, waste frying oils and soybean). Most of researcher investigated the influence of biodiesel on engine performance, combustion and emissions (Ozener et al. 2014, Zhang et al. 2013, Qi et al. 2010) but very few on green diesel. Biodiesel has few disadvantages like higher viscosity, higher production cost, restriction on feedstock use, low energy density and higher freezing point. (Agarwal 2007, Knothe et al. 2005). Crude palm oil increases the deposit formation in cylinder as compared to petro diesel (Pipitone and Costanza 2018).
The different technologies used a variety of feedstock including lignocelluloses biomass (such as wood and crop residues, dedicated herbaceous or tree energy crops), grains, sugar crops, vegetable oil (soybean, canola/rapeseed, etc.), animal fat (beef tallow, pork lard), and waste cooking greases. These all technologies targeted hydrocarbon products that are similar to petroleum diesel (Milbrandt et al. 2009). (An et al. 2012) computed the combustion and emissions of waste cooking based biodiesel at partial load conditions. (Silitonga et al. 2013) investigated the performance and exhaust emissions of Ceiba pentandra blend biodiesel in diesel engine. Two common techniques are used to produce diesel-type fuel from biomass which are hydro processing and transesterification of triglycerides. Transesterification is used to obtain biodiesel whereas hydro processing is used to synthesize green diesel (Boyás et al. 2012). The cost of feed stock affects the total cost of biofuel significantly. Waste cooking oil (WCO) is more economically viable then vegetable oils, also it is presumed that cooking oil becomes carcinogenic after heating for a long period and it is harmful for health, that is why WCO was selected as a feedstock.
Material and Methods
This experimental work is divided into three parts. The first part of the experiment involves production of green diesel, second part is related to the measurement of key properties of green diesel and third part is associated with performance computation and combustion analysis.
Production of green diesel: For production of green diesel, waste cooking oil was selected as a feedstock where as Na2CO3 is used as a catalyst. In the present study green diesel was produced by hydro processing of waste cooking oil. Hydroprocessing completed in two phases (hydrocracking and biocrude distillation). The steps involves in the production of green diesel are shown below in figure 1 with the help of flowchart.
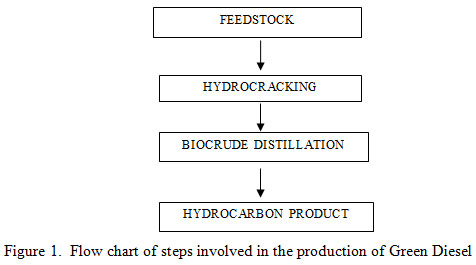 |
Figure 1: Flow chart of steps involved in the production of Green Diesel |
In first step, 1.5 liters waste cooking oil was taken with the help of measuring tube. The quantity of sample has been decided on the basis of capacity of the cylinder of HTHP reactor (2 liters). Na2Co3 (anhydrous) was mixed in the sample of waste cooking oil (WCO) as a catalyst. Sample was filled into the reactor, and hydrogen (H2) was supplied up to 5.5 bar pressure through the inlet vent. Selection of catalyst and optimum pressure of H2 was predefined on the basis of production optimization. About 87 %(by weight) bio crude could be obtained after hydro processing while the balance 8 % escapes as non-condensable gases and 4-5% water at the bottom of the collecting flask. The resultant bio crude was investigated with respect to appropriate standards and distilled as per ASTM D2892 and ASTM D5236 using the TBP distillation unit, (Rakopoulos et al. 2006).
Different hydrocarbon chain lengths have progressively higher boiling points, so they can be separated by distillation. A fractional distillation tower will heat, vaporize, and then condense into different products. In the present experimental investigation, 1200 ml (1100 gm) bio-crude was taken for distillation. After distillation about 110 ml gasoline, 25 ml aviation/kerosene and 240 ml diesel fraction is being produced. Diesel fuel fraction has been produced at 180-380°C temperature range, aviation fuel between 140-180oC and gasoline fuel fraction at 35- 140oC. Different distillates of biocrude oil have different color and different number of carbon. Color of different biocrude oils after biocrude distillation is shown in figure 2. where as carbon atom range shown in Table 1.
 |
Figure 2: Different distillates of bio crude oil |
Table 1: Approximate molecular size of crude oil fractions
Fraction No. of carbons
Gases 1-4
Gasoline 7-11
Aviation fuel/kerosene 9-16
Diesel 12-22
Residue >40
Fuel properties of green diesel: Key fuel properties such as specific gravity, kinematic viscosity, cetane number and flash point were measured. (Pham et al.2018) introduced a correlation to predict kinematic viscosity and density of biodiesel. The principle involves the oscillating U-tube which is a technique to determine the density of liquids and gases based on electronic measurement of the frequency of oscillation, from which the density value is calculated. From density, specific gravity of fuel can be calculated. The engine frequency of this container is influenced by the sample’s mass. The direction of oscillation is normal to the level of the two branches. The oscillator’s eigen frequency is only influenced by the part of the sample that is actually involved in the oscillation. These properties were measured as per ASTM D975.Instrument used for measurement of flash point and fire point follows the ASTM D92 – 12b standard. The kinematic viscosity of the oil was determined as per ASTM D 446 –12 using kinematic viscosity bath. The device was manufactured by Lawler Manufacturing Corporation, USA, with Model 86-17D
Performance computation and combustion analysis: Engine test rig was used for basic measurements, which usually should be undertaken to evaluate the performance of an engine on almost all tests, like engine speed, break power, indicated power, fuel consumption. The test engine and dynamometer were controlled by a microprocessor system equipped with data acquisition and logging. Sensors were fitted to the engine and dynamometer to measure relevant parameters and send the data to the control system. The sensors measured engine load, engine speed, inlet air temperature, exhaust gas temperature, lubrication oil temperature, fuel consumption and the cooling water temperature.
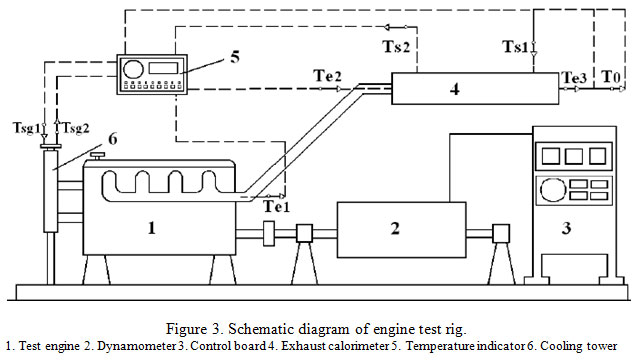 |
Figure 3: Schematic diagram of engine test rig. 1. Test engine 2. Dynamometer 3. Control board 4. Exhaust calorimeter 5. Temperature indicator 6. Cooling tower |
Results and Discussion
The fuel properties help us to explain the behavior of fuel. So, it is necessary to measure the fuel properties. The important fuel properties such as specific gravity, kinematic viscosity, net calorific value and flash point etc. were measured in laboratory. These fuel properties for green diesel are shown in the table 2
Table 2: Green diesel fuel properties
Fuel properties Magnitude
Specfic gravity 0.78
Kinematic viscosity (centi-stoke) 3.0823
Flash point (°C) 114
Cetane Number 84
Net calorific value (KJ/KgK) 38823
From the computational data in table 2 it is concluded that green diesel has low specific gravity and high cetane number. Kinematic viscosity about 3.0 centi-stoke which is satisfactory with past research. The net calorific value is 38823 kJ/kg K which has considerable variation with calorific value calculated by Kennedy et al.(2015). The cause of variation may be due to different feedstock or different production method or may be any device error or due to limited experiment. To overcome this, more experiment need to be performed. The performance parameters: Include indicated thermal efficiency, brake thermal efficiency, specific fuel consumption (kg/kWh), and fuel consumption were calculated at different rpm and at different load condition. Performance data of green diesel observed by this experiment is shown in the table 3 and table 4.
Table 3: Indicated and brake thermal efficiency using green diesel
Speed (rpm) Load IThEff (%) BthEff (%)
1490 0.86 83.82 5.18
1470 4.90 85.76 21.29
1450 8.97 87.96 33.97
1430 12.84 74.23 35.98
1410 16.68 67.98 38.14
1390 20.62 51.37 31.20
From table 3 it is observed that if load is increased and engine speed is decreased then initially indicated thermal efficiency and break thermal efficiency both increases but after that they start decreasing. It can be noticed that indicated thermal efficiency is maximum at 1450 rpm and 8.97 kg load whereas maximum break thermal efficiency is 38.14 % at 1410 rpm and 16.68 kg load. BthEff increases with load because if load increases power also increases.
Table 4: SFC & Fuel consumption for green diesel
Speed (rpm) Load (kg) SFC(kg/kWh) Fuel (kg/h)
1490 0.86 1.79 0.42
1470 4.90 0.43 0.56
1450 8.97 0.27 0.69
1430 12.84 0.26 0.97
1410 16.68 0.25 1.48
1390 20.62 0.30 1.67
Specific fuel consumption in kg/kWh decrease with increase in load but it starts increasing when load is increased up to 20.62 kg. This is due to initial increase in brake power with load but after certain value of load, brake power also decreases. The fuel consumed in kg/h increases with load. This is because power requirements increases with load and ultimately consumption of fuel increases
In combustion analysis, the behavior of fuel – air mixture inside the cylinder is analyzed to find out the physics behind the performance and emissions characteristics of the fuel. During the combustion of green diesel, the variation in pressure, temperature, net heat release, and fraction of fuel mass burned were investigated with crank angle and then compared to petro diesel. Combustion information was obtained from the pressure sensor located in the engine cylinder head. The in-cylinder pressure plot reflects slightly late combustion and larger ignition delay for the green diesel fuels when compared to petro-diesel fuel. In the initial phase of combustion, the pressure rise for green diesel was less because it does not contain smaller length hydrocarbons that can mix quickly with air. Diesel fuel is composed of a wide range of hydrocarbons with varying chain lengths in the range of C-8 to C-21. Green diesel consists of primarily C-11 to C-17 alkenes (Yoon and Lee 2011). The green diesel contains a narrow range (i.e. 11 to 17) which leads to high intensity combustion occurs with. The high pressure values of green diesel fuel may be a result of better combustion near top dead centre (TDC). Green diesel has the high cylinder pressure peak as compared to petro-diesel. The peak pressure in case of green diesel was 72.8 bar at 376° crank angle and for petro-diesel was 72.2 bar at 376° crank angle. Figure 4 shows the plots of the pressure rise with crank angle for both fuels for complete cycle where as figure 5 show this variation at top dead centre.
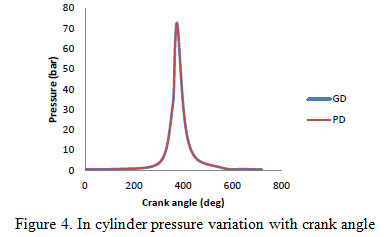 |
Figure 4: In cylinder pressure variation with crank angle |
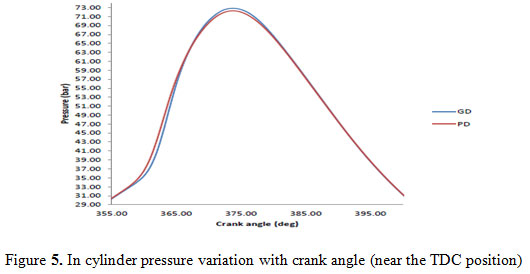 |
Figure 5: In cylinder pressure variation with crank angle (near the TDC position) |
The rate of pressure rise for the green diesel fuels is also noticeably large as compared to petro-diesel fuel. This mainly depends on the fuel’s chemical and physical properties, which affect the mixing and ignition process. Higher increasing rate of in-cylinder pressure generally leads to more combustion noise. Figure 6 show the rate of pressure with crank angle.
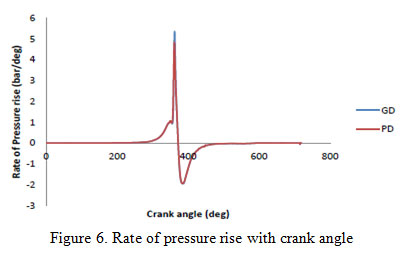 |
Figure 6: Rate of pressure rise with crank angle |
Figure 7 shows the plots of the net heat release rate for both fuels. The heat release rate curve for both the fuels follow a similar pattern. Both have a pre-mixed combustion phase and a diffusion combustion phase. The green diesel fuels have shorter ignition delays (Dolanimi et al. 2015) and higher heat release rates as compared to petro diesel. The previous literature reported the shorter ignition delays experienced by green diesel fuels and may be due to thermal cracking which tends to break up longer chain hydrocarbons to form smaller hydrocarbons and gaseous compounds which are liable to ignite much faster than their longer chain hydrocarbon counterparts. (Yoon and, Lee 2011). But in present study the combustion results show that the green diesel might not contain smaller hydrocarbons and gaseous compounds. The combustion results of green diesel are somehow different from the previous publications. This might be due to the specific conditions for the current green diesel production process and experimental setup. More experiments are required by altering the production parameters.
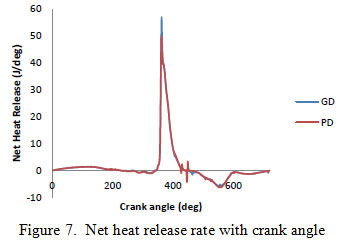 |
Figure 7: Net heat release rate with crank angle |
Figure 8 shows the fraction of mass burned with crank angle for both green diesel and petro diesel. From the graph it is observed that at initial stage when the fuel is injected, the fraction of petrol diesel consumed is more than green diesel, at the middle stage it is equal and in later stage fraction consumed of green diesel is more as compared to petro diesel. This is due to high cetane number of green diesel. It will burn at low temperature as compared to petro diesel which have low cetnane number. If a tinny drop of green diesel is injected inside the cylinder it starts burning immediately but when tiny drop of petro diesel is injected in cylinder it will not burn at this temperature and pressure. Meanwhile another drop injected at same time and piston moves more upward which create more compression. Due to this, temperature will increases which in turn increases the rate of fuel burn.
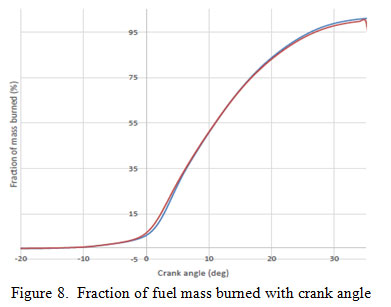 |
Figure 8: Fraction of fuel mass burned with crank angle |
Figure 9 show variation of gas temperature inside the cylinder with crank angle near the top dead centre. From the figure 9 it is concluded that the temperature rise in initial phase is more for green diesel than petro diesel. The reason behind this is the shorter ignition delay of green diesel due to which fuel burn rate is high in this phase as compared to petro diesel which lead to high temperature. In later stage temperature generated is slightly more for petro diesel. Petro diesel have higher ignition delay which causes long lasting of burning fuel as compared to green diesel
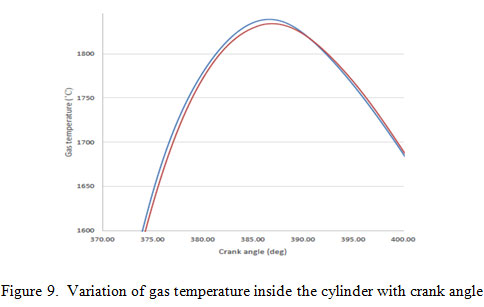 |
Figure 9: Variation of gas temperature inside the cylinder with crank angle |
Conclusion and Recommendations
Green diesel has quality attributes comparable to those of syndiesel, including complete compatibility with petroleum diesel, high energy density, low specific gravity (0.78), excellent storage stability. Results from combustion show shorter ignition delays for green diesel which can overcome the problem of knocking in CI engine. Green diesel has the high cylinder pressure peak as compared to petro-diesel. The peak pressure in case of green diesel was 72.8 bar at 376° crank angle and for petro-diesel was 72.2 bar at 376° crank angle. In India due the poor economic condition and lack of awareness, used oil is not dumped, and is reused for cooking purpose. If the government introduces some program related to awareness of the harmful effect of reusing the used oil and also provides proper waste oil collection system, then there is huge scope to produce bio fuel from waste oil in India also.
Acknowledgement
The author extends his gratitude to all the people who contributed directly or indirectly in bringing out this research to final form, a special thanks to Prof. Rakesh Kumar.
References
Agarwal AK. (2007). Biofuels (alcohols and biodiesel) applications as fuels for internal combustion engines. Prog Energy Combust Sci;33:233–71.
An H, Yang WM, Chou SK, Chua KJ.(2012). Combustion and emissions characteristics of diesel engine fueled by biodiesel at partial load conditions. Appl Energy ;99:363–71
Biswas D.K. Shelly, Sharma.(2013). Studies on cracking of Jatropha oil, J. Anal. Appl.Pyrolysis 99 (January) 122–129.
Buzetzki E., K. Sidorová, Z. Cvengroˇsová, J. Cvengroˇs.(2011). Effects of oil type on products obtained by cracking of oils and fats, Fuel Process. Technol. 92 (10) 2041–2047
Deshpande P., K. Kulkarni. (2012). Production and evaluation of biodiesel from palm oil and ghee (clarified butter), Chem. Process Eng. Res. 2 :33–42, ISSN2224-7467 (Paper) ISSN 2225-0913
Dolanimi Ogunkoya, William L. Roberts, Tiegang Fang , Nirajan Thapaliya. (2015).Investigation of the effects of renewable diesel fuels on engine performance, combustion, and emissions, Fuel 140 541–554
Knothe G, Van Gerpen J, Krahl J. (2005). The biodiesel handbook, Illinois. Champaign: AOCS Press.
Milbrandt A., C. Kinchin, and R. McCormick. (2011). The Feasibility of Producing and Using Biomass-Based Dieseland Jet Fuel in the United States, Technical Report NREL/TP-6A20-58015
Ozener O, Yuksek L, Ergenc AT, Ozkan M. (2014).Effect of soybean biodiesel on a DI diesel engine performance, emission and combustion characteristics. Fuel;115:875–83.
Pipitone, E., Costanza, A.,( 2018). An experimental investigation on the long-term compatibility of preheated crude palm oil in a large compression ignition diesel engine. Biofuel Res. J. 5(4), 900-908.
Pham, M.T., Hoang, A.T., Le, A.T., Al-Tawaha, A., Dong, V.H., Le, V.V., (2018). Measurement and prediction of the density and viscosity of biodiesel blends. Int. J. Technol. 9(5), 1015-1026.
Qi DH, Chen H, Geng LM, Bian YZ. (2010). Experimental studies of the combustion characteristics and performance of a direct injection fueled with biodiesel/ diesel blends. Energy Convers Manage;51:2985–92.
Rakopoulos CD, Antonopoulos KA, Rakopulos DC, Giakoumis EG. ( 2006). Study of combustion in a divided chamber turbocharged diesel engine by experimental heat release analysis in its chambers. Appl ThermEng; 26:1611–20.
Ramya G., R. Sudhakar, J. Amala Infant Joice, R. Ramakrishnan, T. Sivakumar. (2012) Liquid hydrocarbon fuels from Jatropha oil through catalytic cracking technol-ogy using AlMCM-41/ZSM-5 composite catalysts, Appl. Catal. A: Gen. 433-434(August (8))170–178.
Shameer, P.M., Ramesh, K.,( 2018). Assessment on the consequences of injection timing and injection pressure on combustion characteristics of sustainable biodiesel fuelled engine. Renew. Sust. Energy Rev. 81, 45-61
Silitonga AS, Masjuki HH, Mahlia TMI, Ong HC, Chong WT. (2013). Experimental study on performance and exhaust emissions of a diesel engine fuelled with Ceiba pentandra biodiesel blends. Energy Convers Manage;76:828–36.
Suarez A.Z., S.M.P. Meneghetti, M.R. Meneghetti, C.R. Wolf. (2007). Transformac¸ ão detriglicerídeos em combustíveis, materiais poliméricos e insumos químicos:algumas aplicac¸ ões da catálise na óleo-química, Quim. Nova 30 (3) 667–676
Yoon SH, Lee CS. Experimental investigation on the combustion and exhaust emission characteristics of biogas–biodiesel dual – fuel combustion in a CI engine. Fuel Process Technol 2011; 92:992–1000
Yu F., L. Gao, W. Wang, G. Zhang, J. Ji.(2013). Bio-fuel production from the catalytic pyrolysis of soybean oil over Me-Al-MCM-41 (Me = La, Ni or Fe) mesoporousmaterials, J. Anal. Appl. Pyrolysis 104 (November) 325–329
Zhang J, Jing W, Roberts WL, Fang T. (2013). Soot temperature and KL factor for biodiesel and diesel spray combustion in a constant volume combustion chamber. Appl Energy;107:52–65.


