Department of Biotechnology, Gandhi Institute of Engineering and Technology (Autonomous), Gunupur-765022, India
Corresponding author Email: manoja2003@rediffmail.com
Article Publishing History
Received: 28/09/2018
Accepted After Revision: 15/11/2018
Bacillus sp. was isolated from the soil sample of Gunupur, Odisha, India and screened to produce bacitracin at the laboratory. The bacitracin from the bacterium was extracted using butanol-ether solution as a solvent and its antibacterial properties against five bacteria namely Staphylococcus aureus (MTCC 98), Escherichia coli (MTCC 739), Micrococcus luteus (MTCC 106), Salmonella typhimurium (MTCC 1254), and Pseudomonas aeruginosa (MTCC 2453) have been studied in vitro by agar well diffusion method. Simultaneously, the individual, square and combined effect of incubation time, pH and temperature on the bacitracin production was studied using Response Surface Methodology (RSP). Analysis of variance (ANOVA) has shown a coefficient of correlation value of 0.9611) and a quadratic correlation for bacitracin production was derived with 95% confidence level. Incubation time of 26.5 h; pH of 5.67; and temperature of 30.65°C have been found to be as optimum operating conditions for a maximum bacitracin production from Bacillus sp. BSG of 8.72µg/100 ml.
Soil Microorganism; Bacitracin; Antimicrobial Activity; Anova; Response Surface Methodology
Acharya D, Satapathy S, Das M. Extraction of Bacitracin from Bacillus Subtilis BSG and its Optimization Using Response Surface Methodology. Biosc.Biotech.Res.Comm. 2018;11(4).
Kumar V, Garg M. L. Extraction of Bacitracin from Bacillus Subtilis BSG and its Optimization Using Response Surface Methodology. Biosc.Biotech.Res.Comm. 2018;11(4). Available from: https://bit.ly/2mmjFPE
Introduction
The use of antibiotics in clinical biology is a common phenomenon and their uses are rapidly increasing day by day. Most of the antibiotics have been obtained from microorganisms. Antibiotics which have been used today are either natural or semi synthetic or synthetic by nature (Trookman et al., 2011). Semi synthetic antibiotics are prepared by modifying the natural antibiotic, either chemically or enzymatically with an aim to enhance the efficiency of original antibiotics. Antibiotics used in medical practices have been obtained from few groups of microorganisms only. The two genera of bacteria Streptomyces and Bacillus are the maximum contributor of antibiotics (Pavithra et al., 2009). The antibiotics produced by Bacillus species are being recommended for infection caused by gram positive bacteria. Bacillus species produces about 167 antibiotics which are polypeptide by nature (Arias et al., 1999). The two species of Bacillus such as B. subtilis and B. brevis produce 66 and23 polypeptide respectively. The most important peptide antibiotics produced by Bacillus species is Bacitracin. It is commonly used in medical preparations, either alone or with other antibiotics, against all kinds of gram positive bacteria and to some extent gram negative bacteria. The antibiotic interferes with protein synthesis and disrupts the cell of parasitic bacteria. The bacitracin production by Bacillus licheniformis can be enhanced by acetoin reductase activity and the transcription factors Spo0A and AbrB regulate the bacitracin synthesis, (Kasetty et al., 2015 Lagzian
et al., 2018).
Many investigations have been conducted on bacitracin, but the optimization ofits production remains unexplored. The present work aims at the isolation of bacitracin producing the soil inhabitant Bacillus bacterium obtained from the local soil sample with the purpose of establishing its antibacterial activity against five bacteria such as Staphylococcus aureus (MTCC 98), Escherichia coli (MTCC 739), Micrococcus luteus (MTCC 106), Salmonella typhimurium (MTCC 1254), and Pseudomonas aeruginosa (MTCC 2453). Simultaneously, central composite design was used to investigate the effect of incubation time, pH and temperature on the production of bacitracin. The production parameters are optimized in order to maximize the production of bacitracin.
Materials Andmethods
Experimental Procedure
Isolation of Bacillus species
Soil samples were collected by sterilized plastic bags from different parts of Gunupur area. The soil inhabitant microorganism was isolated by following serial dilution (10-4 folds) using nutrient agar plate (Coppuccino et al., 1996) and CFU of the soil microbes’ were recorded.
Sub culture and characterization of Bacterium
The isolated bacterium was sub cultured in the laboratory using the same nutrient by following agar slant technique, and preserved at the low temperature (4°C) for further use. The characterization of the bacterium was made at the laboratory by morphological observation and biochemical tests. The suspected Bacillus species were identified by molecular techniques. The growth curve of the bacterium was determined by spectrophotometric techniques (Goodfellow et al., 1980).
Extraction, purification and identification of Bacitracin
The batch culture technique was used for the extraction of bacitracin from isolated bacterium. The bacterium was inoculated in the nutrient broth at 37° C at slow agitation condition for 72 hrs and at log phase of growth the extraction was obtained. The purification of the bacitracin in the culture medium was done using butanol-ether solution (pH=4) as a solvent (Murphy
et al., 2007). At acidic condition, the bacitracin remained in lower aqueous layer was collected carefully followed by neutralizing with sodium bicarbonate and was lyophilized to obtain the purified bacitracin. Thin Layer Chromatography (TLC) and GC-MS had been used to identify the obtained bacitracin . The silica phase acts as the solid phase of the TLC and the mobile phase was prepared with chloroform and methanol in the ratio of 9:1. The Rf value is considered for identification of bacitracin (Phillips, 1999). Confirmation of bacitracin was made by GC-MS (MSGC- 11) instrument with the capillary column of HP-3 (50 mm × 0.521mm, film thickness 0.25µm). 1µL of extract was carefully injected into GC-MS for analysis. The chemical compositions were identified by comparing their retention indices (RI) and mass fragmentation pattern.
Standardization of Production of Bacitracin
The production of bacitracin was carried out with solid state fermentation. The fermentation was made in an Erlenmeyer flask (capacity 1 liter) by using wheat bran as substrate. The substrate (100 gm) was added to 100 ml of phosphate buffer. Then the flask along with the substrate had been sterilized in autoclave at 15 psi for 20 minutes. Isolated microorganisms were inoculated carefully into the flask and incubated at 37° C for 24 hrs. The antibiotic was extracted by following standard procedure with little modifications (Rajan et al., 2014), followed by filtration of aliquot and then centrifuging at 10,000 rpm at 4°C for 20 minutes.
Study of Antibacterial activity
Agar well diffusion method was used to investigate the antimicrobial properties of the bacitracin obtained from solid state fermentation. The Muller-Hinton agar (MHA) plates were prepared by using 20ml of the medium and left overnight at room temperature to check for any contamination to the plates. The test bacteria was grown in a nutrient broth and diluted (OD 620 nm = 0.1) to obtain a bacterial suspension of 1×108 CFU/ml before applying onto the agar plate. Agar wells of 5mm diameter were prepared using sterilized steel gel puncher and each well received 10µL of the extracts. The agar plates were incubated in a BOD chamber at 370C for 48 h. The antibacterial activity of the isolated compound against each test organisms was quantified by measuring the zone of inhibition. The average diameter of three replicates for each organism was determined and expressed in millimeter (mm).
Statistical Analysis
The CCD comprises of a full fractional factorial design with center points amplified by a group of ‘star points’. Statistical methodologies such as RSM based on CCD can also be used to maximize the production of a substance by optimization of operational factors. In compare to conventional methods, apart from the individual contribution of process variables, the contributions of interaction among process variables can be investigated using RSM (Dora et al., 2013). Thus, Response Surface Methodology (RSM) based on Central Composite Design (CCD) was used to explore the effect of the different production parameters viz. temperature, pH and time on the production of bacitracin.
Results Anddiscussion
Characterization Of Bacillus Subtilis
The isolated soil microorganisms obtained from different soil samples of the Gunupur area was characterized morphologically and biochemically (Table 1). The cultured bacteria showed irregular colonies in culture media and shape of the bacteria was rod shaped. The gram staining test had shown gram positive. Similarly, glucose fermentation test and VP tests of the isolated bacterium were positive whereas catalase and lactose fermentation test found to be negative. The isolated microorganism was identified by following Bergay’s manual of systematic bacteriology (Sharma et al., 2010) and found as Bacillus subtilis as shown in fig 1. The batch culture of the isolated bacterium retained lag phase up to 4 hrs followed by log phase (Fig. 2). The morphological features of isolated microorganisms presenting the gram + ve, rod shaped, and having endospore confirms the presence of Bacillus subtilis (Das et al., 2012).
| Table 1: Morphological and biochemical characterstics of microorganism isolated from soil sample of Gunupur area, Odisha | |
| Characters | Observation |
| Shape | Irregular |
| Margin | Lobate |
| Color of the Colony | White |
| Structure | Filamentous |
| Size | Rod Shaped |
| Gram Strian | +ve |
| Carbohydrate fermentation | -ve |
| Mannitol Fermentation | -ve |
| Glucose Fermentation | +ve |
| Catalase fermentation | -ve |
| Nitrate reduction | -ve |
| Methyl | -ve |
| Indole | -ve |
| Litmus | -ve |
| Oxidose | -ve |
| Urase | -ve |
| Lactose Fermentation | -ve |
| Voges proskauer | +ve |
 |
Figure 1: Molecular characterization and phylogenic analysis of the isolated Bacillus subtilis |
 |
Figure 2: Growth curve of the isolated Bacillus subtilis in batch culture with nutrient medium |
Effect of carbon and nitrogen sources on the production of bacitracin
The effect of carbon sources on production of bacitracin is represented in table 2. The extreme production of bacitracin was obtained when maltose is used as the carbon source. Further, minimum production was obtained when fructose is used as the carbon source. To standardize the effect of nitrogen sources on bacitracin production, it was observed that methionine found to be the best nitrogen source among the studied nitrogen compounds (table 3). Earlier report shows that methionine can be used as suitable source of nitrogen for growth of Bacillus species (Kasetty et al., 2015). In the study, least production was observed when NH4Cl is used as the nitrogen source.
| Table 2: Effect of different carbon sources for production of bacitraction by the isolated microorganism | |
| Carbon Sources | Production of bacitracin (μ/100ml of culture) |
| Glucose | 6.0 ± 1.2 |
| Fructose | 3.0 ± 1.6 |
| Sucrose | 6.5 ± 1.4 |
| Mannitol | 8.6 ± 1.2 |
| Sorbitol | 4.8 ± 1.8 |
| Starch | 4.5 ± 2.0 |
| Table 3: Effect of different nitrogen sources for production of bacitracin by the isolated microorganism | |
| Nitrogen Sources | Production of bacitracin (μ/100ml of culture) |
| Asparagine | 7.0 ± 1.5 |
| Tyrosine | 7.5 ± 1.9 |
| Methionine | 8.0 ± 1.2 |
| NH4Cl | 3.0 ± 2.0 |
| NaNO3 | 4.0 ± 1.8 |
| Urea | 4.5 ± 1.6 |
Individual effect of incubation time, pH and temperature on bacitracin production
The production of bacitracin by the isolated bacterium was high at log phase of growth. The initiation of production of bacitracin was observed after 8 hours of inoculation and maximum amount, after 30 hours of
inoculation. The activity continued up to 60 hours of inoculation and then found to decline as depicted in
fig. 3. The effect of pH on bacitracin production is shown in fig. 4. The maximum production of bacitracin was obtained in acidic condition (pH 5.5). The environmental conditions play an important role for the growth and production of metabolites in Bacteria (Kang et al., 2017). There was no production of antibiotic at 20° C by the isolated microorganism. The maximum production of bacitracin was obtained at 35°C. Beyond 350 C, the production of antibiotic decreases as observed in fig. 5. Because Bacillus subtilis is a mesophillic bacterium, the growth shall be optimum at 250C – 350C. Therefore, the production of bacitracin is optimum at this temperature (Deslouches, et al., 2015).
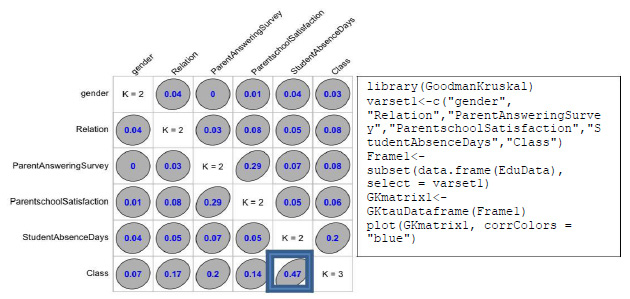 |
Figure 3: Production of bacitracin by isolated Bacillus subtilis in batch culture with respect to incubation time |
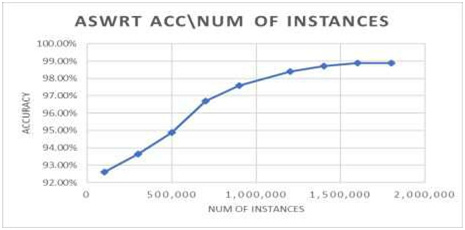 |
Figure 4: Effect of pH on production of bacitracin by isolated Bacillus subtilis |
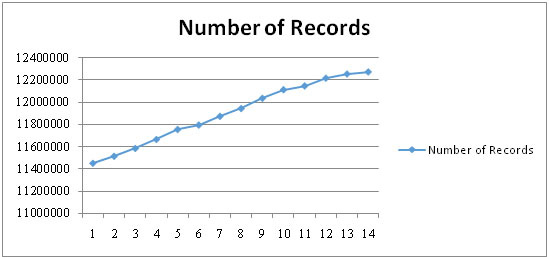 |
Figure 5: Effect of temperature on production of bacitracin by isolated Bacillus subtilis |
The chromatogram of GC-MS of extract is represented as fig. 6. It is observed that, there are two major peaks having the retention time of 10.478 and 14.617 and the area occupied are 69.27% and 2.51% respectively. The molecular weight of the bacitracin was measured as 1.6 k Da. The lyophilized antibiotic is faint yellow in color, amorphous and water soluble. The zone of inhibition of isolated bacitracin against five pathogens namely Staphylococcus aureus, Escherichia coli, Micrococcus luteus, Salmonella typhimurium and Pseudomonas aeruginosa is represented in the table 4. The maximum zone of inhibition was observed against Staphylococcus aureus (23 mm) and the minimum zone of inhibition was observed against Salmonella typhimurium (12 mm).
| Table 4: Zone of Inhibition (in mm) against five pathogens by the bacitracin extracted from the isolated microorganism. | ||
| Test Organism | Zone of Inhibition (in mm) | |
| After 24 hours | After 48 hours | |
| Staphylococcus aureus | 16.6 ± 1.4 | 23.0 ± 1.2 |
| Escherichia coli | 12.4 ± 1.3 | 15.6 ± 1.6 |
| Micrococcus luteus | 14.5 ± 1.2 | 18.2 ± 1.4 |
| Salmonella typhimureum | 8.3 ± 1.6 | 12.0 ± 1.4 |
| Pseudomonas aureginosa | 11.4 ± 1.7 | 17.6 ± 1.5 |
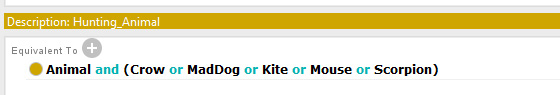 |
Figure 6: Chromatogram of GC-MS analysis of Bacitracin by the isolated Bacillus subtilis |
Statistical analysis
From the analysis of experimental data, it was found that the production of bacitracin is influenced by three process parameters viz. incubation time, pH and temperature. Thus the RSM based on CCD is employed in order to estimate the combined effect of the operating parameters on the bacitracin production. The range of the process parameters and the complete design of the matrix along with the experimental data are presented as tables 5 and 6 respectively. Table 6 is planned to obtain a quadratic correlation for the production of bacitracin consisting 23 numbers of axial runs, 6 numbers of star configurations and 5 numbers of center points. The runs 15-19 around the center points in table 6 are repeated in order to determine experimental errors. The correlation obtained for the production of bacitracin in terms of coded factor is presented as eq. 1.
| Table 5: Level of independent variables | ||||||
| Variables | Symbol | –á | -1 | 0 | +1 | +á |
| Incubation Time, h | A | 2 | 8 | 18 | 28 | 34 |
| pH | B | 4 | 5 | 6.5 | 7.5 | 8.5 |
| Temperature, °C | C | 20 | 28 | 37.5 | 48 | 55 |
| Table 6: Experimental design matrix and response | |||||||
| Run | Coded variables | Actual variables | Response | ||||
| A | B | C | A | B | C | (Y) | |
| 1 | -1 | -1 | -1 | 8 | 5 | 28 | 1.2 |
| 2 | +1 | -1 | -1 | 28 | 5 | 28 | 7.5 |
| 3 | -1 | +1 | -1 | 8 | 7.5 | 28 | 2 |
| 4 | +1 | +1 | -1 | 28 | 7.5 | 28 | 6.5 |
| 5 | -1 | -1 | +1 | 8 | 5 | 48 | 2.5 |
| 6 | +1 | -1 | +1 | 28 | 5 | 48 | 6.2 |
| 7 | -1 | +1 | +1 | 8 | 7.5 | 48 | 3.3 |
| 8 | +1 | +1 | +1 | 28 | 7.5 | 48 | 4.5 |
| 9 | -á | 0 | 0 | 2 | 6.5 | 37.5 | 1.1 |
| 10 | +á | 0 | 0 | 34 | 6.5 | 37.5 | 7.8 |
| 11 | 0 | -á | 0 | 18 | 4 | 37.5 | 3.7 |
| 12 | 0 | +á | 0 | 18 | 8.5 | 37.5 | 5.1 |
| 13 | 0 | 0 | -á | 18 | 6.5 | 20 | 5.8 |
| 14 | 0 | 0 | +á | 18 | 6.5 | 55 | 4.5 |
| 15 | 0 | 0 | 0 | 18 | 6.5 | 37.5 | 8.22 |
| 16 | 0 | 0 | 0 | 18 | 6.5 | 37.5 | 8.36 |
| 17 | 0 | 0 | 0 | 18 | 6.5 | 37.5 | 8.3 |
| 18 | 0 | 0 | 0 | 18 | 6.5 | 37.5 | 8.42 |
| 19 | 0 | 0 | 0 | 18 | 6.5 | 37.5 | 8.33 |
| 20 | 0 | 0 | 0 | 18 | 6.5 | 37.5 | 8.34 |
Y=8.33+1.97A+0.092B−0.211C−0.53AB−0.73AC−0.087BC
−1.42A2−1.43B2− 1.172C2 (1)
The positive and negative signs against each factor in eq. 1 show the synergistic and antagonistic effect of those respective terms on bacitracin production. The calculated values of bacitracin production from eq. (1) are compared with the experimental ones and represented as fig. 7. The values of coefficient of correlation (R2), adjusted R2 (Radj2), predicted R2 (Rpred2) are found to be 0.986, 0.973 and 0.901 respectively, which shows that the calculated values are analogous with the experimental ones. The adequate precision, which measures the signal to noise ratio, is found to be 24.21, indicates that the correlation developed can be used to navigate the design space. The Analysis of variance (ANOVA) for the production of bacitracin is represented as table 7. The F-value” of 75.47 indicates that the model is significant and there is only 0.01% chance of failure of this correlation. The significance of the model terms can be estimated through the “P-value”. The terms A, B, C, AB, AC, A2, B2 and C2 are significant, because the “P-value” for these terms are less than 0.05.
| Table 7: ANOVA for bacitracin production | ||||||
| Source | Sum of Squares | Degree of freedom | Mean Square | F Value | p-value Prob > F | Remarks |
| Model | 118.5209 | 9 | 13.16898 | 75.4718 | 2.13265E-07 | significant |
| A | 53.25338 | 1 | 53.25338 | 305.1967 | 2.98275E-08 | significant |
| B | 0.115238 | 1 | 0.11523 | 0.6604 | 0.037374471 | significant |
| C | 0.610016 | 1 | 0.61001 | 3.4960 | 0.034333927 | significant |
| AB | 2.31125 | 1 | 2.31125 | 13.2458 | 0.005405229 | significant |
| AC | 4.35125 | 1 | 4.35125 | 24.9371 | 0.000745341 | significant |
| BC | 0.06125 | 1 | 0.06125 | 0.3510 | 0.568118214 | |
| A2 | 27.5197 | 1 | 27.51969 | 157.7162 | 5.22146E-07 | significant |
| B2 | 28.20921 | 1 | 28.20920 | 161.6678 | 4.69653E-07 | significant |
| C2 | 18.76233 | 1 | 18.76232 | 107.5274 | 2.64328E-06 | significant |
| Residual | 1.570399 | 9 | 0.17448 | |||
| Lack of fit | 1.548479 | 5 | 0.30969 | 56.5138 | 0.000840525 | |
| Pure Error | 0.02192 | 4 | 0.00548 | |||
| Cor Total | 120.0913 | 18 | ||||
 |
Figure 7: Comparison of calculated values of bacitracin production from eq. 1 with the experimental ones |
Combined effect of process parameters on the bacitracin production
Fig. 8 shows the combined effect of pH and incubation time on the bacitracin production at a temperature of 37.5°C. Bacitracin production increases with the increase in pH up to 6.5, beyond which it decreases, which may be due to the effect on the rate of nutrient transport and consumption from the medium to bacterial body. Again, the bacitracin production increases exponentially with the increase in incubation time up to 22.75 h, then start decreasing. This may be due to decrease in essential nutrients in the medium. A maximum bacitracin production of 8.3 µg/100ml is found as depicted in fig.7. Similarly, the combined effect of temperature and incubation time on the bacitracin production at pH of 6.25 is presented as fig. 9. It was observed that the bacitracin production increases up to a temperature of 38°C and after that the production decreases. The increase may be due to proper functions of enzymes involved in the bacitracin synthesis pathway of microorganisms. Beyond 38°C what happens so that it decreases, it may be due to the inhibitory effect of enzymes involved in bacitracin synthesis pathway also write down. Fig. 10 shows the combined effect of temperature and pH on the bacitracin production is 18h of incubation time. A maximum bacitracin of production of 7.2 µg/100mlis found (fig. 11).
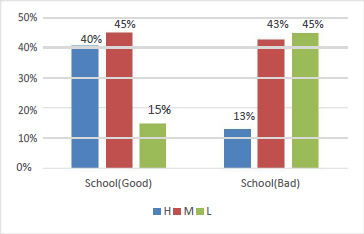 |
Figure 8: Combined effect of pH and incubation time on the production of bacitracin at a temperature of 37.5°C |
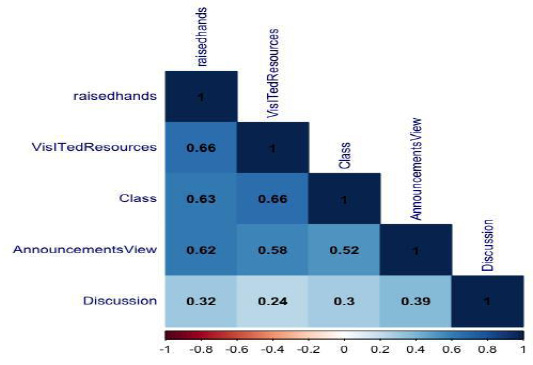 |
Figure 9: Combined effect of temperature and incubation time on the bacitracin production at a pH of 6.25 |
 |
Figure 10: Combined effect of temperature and pH on the bacitracin production at 18h of incubation time. |
 |
Figure 11: Optimization region of pH and incubation time for maximum production of bacitracin. |
Optimization of the production of bacitracin
The focal objective of this study is to optimize the process parameters for the production of bacitracin using the developed correlation (eq 1). The quadratic correlations are optimized using quadratic programing to maximize the production of bacitracin (Bodroth et al., 2012). The optimum region of pH and time for the maximum production of bacitracin is presented as fig. 11.The optimized conditions are found to be incubation time of 26.5 h; pH of 5.67 and temperature of 30.65°C for a maximum production of bacitracin of 8.9 µg/100ml as shown in table 8.
| Table 8: Optimized conditions for bacitracin production | ||||
| Incubation time, h (A) | pH (B) | Temperature, °C (C) | Production of bacitracin (μg/100ml of culture) (Y) | |
| observed | predicted | |||
| 26.5 | 5.67 | 30.65 | 8.72 | 8.90663 |
Conclusion
The Bacillus subtilis BSG isolated from Gunupur soil is a gram +ve, rod shaped bacteria has the capabilities to produce bacitracin, a peptide antibiotic. The bacitracin production was optimized in different parameter and found the suitable production point. The bacitracin production will be affected by imbalanced carbon and nitrogen sources. The present work will give a milestone for the production of antibiotics in pilot plant scale. Bacitracin also has potential antimicrobial properties with maximum inhibition for pathogenic bacteria. Simultaneously, RSM based CCD was used to estimate the individual, combined and the square effect of all incubation time, pH and temperature on the bacitracin production. Using ANOVA results a quadratic correlation was developed with a coefficient of correlation of 0.986. Again, the process parameters are optimized so as to maximize the bacitracin production. The optimized conditions is found of incubation time of 26.5h; pH of 5.67 and temperature of 30.65°C for a maximum production of bacitracin of 8.9 µg/100ml. Therefore, the correlation developed and the data reported may provide convenient information for the economic production of bacitracin within ranges of the operating parameters investigated.
Acknowledgements
The authors wish to acknowledge the management of GIET for encouragement and providing laboratory facility to carry out the investigation.
References
Arias, R.S., Sagardoy, M.A. and Vanvuurde, J.W.L. (1999). Spatio-temporal distribution of naturally occurring Bacillus spp. and other bacteria on the phylloplane of soybean under field conditions. J Basic Microbiol. 39, 283–292.
Bodroth, R.P. and Das, M. (2012). Phytochemical Screening and antimicrobial activity of ethanol and chloroform extract of Zizyphus nummularis Wt. & Arm. African J Biotechn. 11, 4929-4933.
Coppuccino, J.G. and Sherman. N, (1996). A Laboratory Manual in General Microbiology, fourth ed. Benjamin Commius Publication Company, California.
Das, M. and Kumar, S. (2012). Characterization of â lactamase producing bacterium from soil of Mahendragiri region, Orissa. Asian Jr. Microbiol. Biotech. Env. Sc. 14 (2), 229- 232.
Deslouches, B., Steckbeck, J. D., Craigo, J. K., Doi, Y., Burns, J. L., and Montelaro, R. C. (2015). Engineered cationic antimicrobial peptides to overcome multidrug resistance by ESKAPE pathogens. Antimicrob. Agents Chemother. 59, 1329–1333. doi: 10.1128/AAC.03937-14.
Dora, D.T.K., Mohanty, Y.K. and Roy, G.K. (2013). Hydrodynamics of three-phase fluidization of a homogeneous ternary mixture of regular particles – Experimental and statistical analysis. Pow Techn. 237, 594 – 601.
Goodfellow, M. and Board, R.G. (1980). Microbiological Classification and Identification, Aca Press New York 152
Kang, H. K., Kim, C., Seo, C. H., and Park, Y. (2017). The therapeutic applications of antimicrobial peptides (AMPs): a patent review. J. Microbiol. 55, 1–12. doi: 10.1007/s12275-0176452-1
Kasetty, G., Kalle, M., Morgelin, M., Brune, J. C., and Schmidtchen, A. (2015). Anti-endotoxic and antibacterial effects of a dermal substitute coated with host defense peptides. Biomat. 53, 415–425. doi: 10.1016/j.biomaterials.2015.02.111
Lagzian, M., Shahraki, A., Besharatian, M. and Asoodeh, A. (2018). A thermostable alkaliphilic protein-disulfide isomerase from Bacillus subtilis DR8806: cloning, expression, biochemical characterization and molecular dynamics simulation. Internat J Biomacromol. 107, 703-712.
Murphy, T., Roy, I., Harrop, A., Dixon K. and Keshavarz, T. (2007). Effect of oligosaccharide elicitors on bacitracin a production and evidence of transcriptional level control. J biotechn 131, 397–403.
Pavithra, P.S., Sreevidya, N. and Verma, R.S. (2009). Antibacterial activity and chemical composition of essential oil of Pamburusmissionis. J. Ethnopharmaco. 124, 151-153.
Phillips, I. (1999). The use of bacitracin as a growth promoter in animals produces no risk to human health. J Antimicrob Chemother. 44(6),.725−728.
Rajan, B.M. and Kannabiran, K. (2014). Extraction and identification of antibacterial secondary metabolites from marine Streptomyces sp. VITBRK2. Int J Mol Cell Med. 3,130–137.
Sharma, D. and Das, M. (2010). Efficacy of antimicrobial metabolites of Pseudomonas fluroscenes PFG against phytopathogenic fungi. Pro Nat Aca. 1, 68-71.
Trookman, N.S., Rizer, R.L. and Weber, T. (2011). Treatment of minor wounds from dermatologic procedures: A comparison of three topical wound care ointments using a laser wound model. J. Am. Acad. Dermatol. 64, 08–15.


