Faculty of Sciences, Biology Department, King Abdulaziz University, P. O. Box 12161, Jeddah 21473, Saudi Arabia
Corresponding author email: Fmbokh@kau.edu.sa
Article Publishing History
Received: 05/10/2020
Accepted After Revision: 10/12/2020
Storage fruit at suboptimal conditions would promote the fungal growth and perhaps the production of mycotoxin which probably transfer to apple products such as apple juice and other apple products, such as purees and baby food is consumed by infants which can be dangerous for human consumption. Therefore, the objective of this study was to investigate a samples of fresh and dry apples including (40) fresh apple and (20) dry apple were collected from different markets in Jeddah governorate, Saudi Arabia. In this investigation, mycoflore from apple samples were isolate the by using Potato Dextrose Agar (PDA) medium at a temperature of 28 C. The results of this study include the isolation and molecular identification of 5 genera and 39 different species which were identified as Penicillium, Aspergillus, Rhizopus, Alternaria, and Fusarium. The most dominant genera was Penicillium sp. Then we used thin layer chromatography (TLC) to test about 50 Penicillium isolates for their ability to produce patulin. Moreover, P. expansum and P. chrysogenum were found to be toxigenic fungi with higher probability to produce and secrete patulin. In conclusion, fresh or dried apples may be contaminated by certain strains of fungi that produced toxins, specially patulin which cause may human health problems.
Mycotoxins, Patulin, Apple, TLC, Penicillium, P. expansum, P. chrysogenum
Alzain R. H, Bokhari F. M, Najjar A. Detection of two molecular identified strains of Penicillium and patulin producing from fresh and dried apple in Jeddah, Saudi Arabia. Biosc.Biotech.Res.Comm. 2020;13(4).
Alzain R. H, Bokhari F. M, Najjar A. Detection of two molecular identified strains of Penicillium and patulin producing from fresh and dried apple in Jeddah, Saudi Arabia. Biosc.Biotech.Res.Comm. 2020;13(4). Available from: https://bit.ly/3mySqv0
Copyright © Alzain et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Apples are fruits with high levels of sugars and other nutrients which making it favorable media for fungal growth and production of mycotoxin. Apples can be infected by fungi through a split or other injury of the fruit which breaks the skin of the apple. Then fungi in apart from effects of relative humidity and high temperature can produce some enzymes as pectinase which break down apple pectin then absorb these nutrients (Ruangwises et al., 2013, Ruangwises et al., 2011, Sani et al., 2014). Suboptimal storage conditions of apples would increase fungal growth and mycotoxin production (Singh and Sharma, 2018). Filamentous fungi which occur naturally in fruits produce mycotoxins as secondary products that represent a very large group of different substances and compounds including patulin, ohratoxin , aflatoxins and alternaria toxins that produced by different types of mycotoxigenic species (Henry et al., 2001, Hussain et al., 2008). During the growing season, moulds can infect fruits as apple. These infections can also happen during harvesting, handling, transportation, storage, marketing conditions and by consumer himself. This infection can affect the fruit itself and can affect the products of this fruit as apple juice (Erten et al., 2019).
One of the oldest methods that used for preserving food is drying it producing dried apple in this case however, drying the fruits making it very sensitive to change in color due to drying conditions as temperature and oxygen as well as storing conditions of the packed products. Therefore, polymeric materials that used in packing of dried apples should have appropriate barrier characters of preventing microorganisms, water, oxygen, carbon and nitrogen from entering the package. In general, dried fruits is considered less to spoilage by fungi than fresh apple (Hussain et al., 2008).
There are many fungi that can cause apple spoilage, the most common one is Penicillium expansum and Monilinia fructigena (However, there are other genera of fungi that also can affect the apples and were isolated from apples in previous studies. These genera include Colletotrichum, Xylaria, Botryosphaeria were found in the study of Kamkar et al. (2005). Another fungi genera called Rhizopusoryzae as found in the study of Rouissi (2012) and Aspergillus spp. which is isolated from apples and know for causing fruit infections and cause allergies in human (Ruangwises et al., 2013). Other studies indicated Cladosporium spp. to be an infection species as they found them in stored apples, besides Penicillium, Aureobasidium, Sporobolomyces, Cryptococcus and Alternaria spp. (De Melo et al., 2012).
In general, it was found that there are more than 90 species of fungi that can be a causative agent to the infection of apple causing apple decay during storage. Penicillium spp., including Penicillium expansum when infect apple cause production of blue mold which is soft rot on the apple and they are considered that most commonly reported infectious agents. Other species that can cause decay of apple during storage include P. viridicatum, P. brevicompactum, P. solitum and P. expansum which found to produce patulin in apples. Patulin (PAT) is a mutagenic, immunotoxin and neurotoxic mycotoxin that produced by some types of fungi. Therefore, it is very important to detect and ensure the free of apple fruit from P. expansumespecially when these apples are used for juice production (Shim et al., 2004).
Penicillium spp. is a large group that contained at least 150 species, similar in morphology however, there is a great group of variability among many species of them therefore, and we can recognize more than 1,000 different phenotypes. This inherent variability is very important in identifying the type of fungi; however, only 70 to 80 % of the isolate species even when isolated from common sources are readily identifiable. Morphological criteria and characters of Penicillium spp. are the most taxonomic keys to identify Penicillium spp. Therefore, Penicillium spp. can be identifying upon both of micro and macro morphology besides produced color in the mycelium or when diffused into the growth medium.
Moreover, molecular techniques are also used to identify this type of fungi as in recent studies; it is found that they are genotypic characterizations of Penicillium spp. that found to be useful in identifying it. Several methods for detecting DNA polymorphism in fungi have been used to detect intraspecific and interspecific variation in Penicillium sp and other genera (Sebage, 2004, Novotný et al., 2019). Therefore, many isolates were collected and initially characterized using phenotypic and physiological parameters. Moreover, Molecular techniques have been used for the detection of Penicillium sp from isolated samples (Sebage, 2004, Shamsi et al., 2016).
MATERIAL AND METHODS
Apples samples collection: Sixty samples which include 40 samples of fresh apple (Red Delicious apple and Granny Smith apple), 20 samples of dried apple were collected from different markets of Jeddah governorate, Saudi Arabia in August 2018. Samples were collected in sterilized polyethylene bags, transferred into laboratory and preserved in refrigerator until the study time.
Isolation fungi from fresh and dried apple using smear method: Swap from each sample of apple was prepared to inoculate agar plates. The plates were incubated at 25◦C for 7 day. PDA medium was used as a subculture medium to identify the isolates.
Direct plating method for dried apple: Each dried apple sample was cut aseptically into small pieces. Three pieces were transferred to the surface of agar plate containing Potato Dextrose Agar medium (PDA) from OXOID, UK. The plates were incubated at 25°C for 7 days (Pitt et al., 1992). PDA medium was used as a subculture medium to identify the isolates.
Morphological and molecular identification of fungal isolation
Diversity analysis: Culture and morphological characteristics of the isolated fungi of 5-10 days, grown on PDA were assessed by examination under a light microscope. Subsequently, slide preparation of fungi taken from the culture were stained with drop of Lacto phenol cotton blue and examined for spores and mycelia features using light microscope (x 400). Identification of fungi was based on comparison of morphology, color, shapes of spores and colony characters as descript by Domsch et al. (2007) and Watanabe (2010).
Genotypic Identification of two strains isolates: Two strains isolates (P. expansum and P. chrysogenum) were injected in (100) ml of Erlenmeyer flasks containing 20 ml potatoes dextrose broth (PDB) then incubated at 28 °C for 5 days, then was filtration of mycelia using sterilized filters. The fungal mycelium was ground to a powder by sterilized pestle and mortar using liquid nitrogen. Powders were transferred to 1.5 ml Eppendorf tube and stored at -20°C. Thirty mg of frozen mycelium powder were resuspended and lysed in 500 μl of lysis buffer (pH7•8) and incubated in a water bath at 37°C for 60 mins. The lysis buffer was a mixture of 20 mmol/l sodium acetate, 40 mmol/l Tris-acetate, 1 mmol/l EDTA and 1% SDS. Extraction of the DNA was done by Gene JET Genomic DNA extraction kit (Thermo Scientific, USA) and the PCR technique was used to magnify internal transcribed spacer (ITS) region of ribosomal DNA (rDNA) using two primers; ITS1(CTTGGTCATTTAGGGAAGTAA) and ITS4(TCCTCCGCTTATTGATATG) in a thermal cycler (Esco health care, Swift max pro, Malaysia). The reaction mixture (50 μl) included 5 μl each of the primers, 3 μl of templet DNA, 50 μl of nuclease-free water and 25 μl of green PCR mix (Promega, Go Taq ® Green Master Mix, USA).
DNA visualization: On to 1.5% agarose gel, the PCR products were tested and run for 45 mines at 130 volts electrophoretic gel, ethidium bromide was using as stained and visualized under UV light. To quantify and identify PCR products was used the DNA marker. Then the samples were sent to Macrogen company, South Korea for purification and sequencing .
DNA sequencing: Using Big Dye terminator cycle sequencing kit (PE-Applied Biosystems, USA) for PCR product from two fungal strain. Using BLAST Genbank general databases from National Center for Biotechnology information (NCBI) database for sequence identities and used Jukes-Cantor model to construct neighbor-joining tree as described by (Najjar et al., 2019 ).The sequencing data were submitted to GenBank and the obtained accession number was recorded for tow fungal strain.
Screening of fungi for mycotoxins production
Growth of fungal isolates : All fungal isolated were plated in duplicate on PDA to test their ability to produce mycotoxins according to Samson and Hoekstra (1988). The center of each plate was inoculated with a plug cut from a plate culture using a flame-sterilized cork-borer (4 mm internal diameter) and the plates were incubated at 25°C for 10 days. The diameter of the fungal colony (mm) was determined.
Preparation and screening of the fungal extracts:All fungi isolated were tested by a rapid screening method for mycotoxin production (Filternborg and Frisvad, 1980). From 10-day old plate culture, 5 agar discs were cut out near the center of the colony with a flame- sterilized stainless steel cork-borer (4 mm) and removed using a flame- sterilized scalpel. Five discs of growth were removed and extracted by mixture of chloroform: methanol (2/1 V/V). The organic layer were collected in small begoes and preserved at 20°C until used.
Detection of patulin-producing strains by thin layer chromatography (TLC):RF was used as standards for the toxins produced by the tested fungi (patulin).
Preparation and development of TLC plates:For routine examination of extracts, aluminum-backed, DC-Alufolien- Kieselgel 25 plates, silica gel matrix, with fluorescent indictor at 254 nm, were cut to 10 x 10 cm and spotted along a line 1.5 cm from the bottom with 10 μl aliquots of extracts or standards. The plates were developed in the solvent system TEF :Toluene, Ethyl acetate 90% and Formic acid (5:4:1 v/v) at the room temperature until the solvent front reached a line marked 1 cm from the top of the plate(Roberts and Patterson, 1975, Bokhari, 1993). After development, the plates were removed from the solvent, and air-dried in a fume cabinet. The plates are sprayed and dried at 100 °C in a hot-air oven, then examined in a Chromato-Vue cabinet (Model UVP Upland, CA USA) under visible light or short wavelength UV light (254 nm) and long wavelength (366 nm) as described by Samson et al., (2000).
Detection and identification of mycotoxin on TLC plates: Development chromatograms were examined under visible light for colored substances, under UV light for fluorescent substance with and without the use of spray reagent. The detection system for particular toxin is shown Table 1. The patulin gives a fluoresced yellow color after exposing the Chromatography plate for 0.5% of 3-methyl-2benzo-thiazolinone hydrazine hydrochloride (MBTH) spray followed by 15 min at 110◦C.fluorescent color, and ochratoxin gives a fluoresced blue color after exposing the Chromatography plate for Ammonia vapor for 10 min and heating at 110°C for 5 min.
Statistical analysis:The means of variable ± SD were recorded and all data was subjected to statistical analysis using SPSS 16, and the differences between mean values as determined by Student’s t-test were considered significant at P < 0.05.
RESULTS AND DISCUSSION
This investigation involve an inclusive survey of the mycoflora associated with apple fruits in 60 samples collected from different markets in Jeddah governorate, Saudi Arabia. The tested samples embrace fresh and dry apple (40 samples for fresh apple and 20 samples for dried apple). In this respect, Akinmusire (2011) and Chukwuka et al. (2010) mentioned that wide range of microorganisms can affected fruits such as fungi which have a severe menace to production of fruits. Spoilage impute to any change in the condition of food making it less agreeable, or even toxic. Furthermore, presence of mycotoxin in fruits is an ongoing global concern. Mycotoxin contamination is considered an unavoidable and unpredictable problem, even where perfect agricultural, storage, and processing practices are implemented, posing a difficult challenge to fruits safety (Patriarca, 2019). Additionally, during food processing many mycotoxins are not easily eliminated because of their stability against heat, chemical, and physical treatments (Barkai-Golan, 2001, Battacone et al., 2005, Baert et al., 2007, Bilandzic et al., 2010, Basson et al., 2019).
Fruits contamination can also pose an extra hazard for food safety due to the possible relocate of mycotoxins to fruits products such as juices and other products, such as purees and baby food is consumed by infants which can be dangerous for human consumption. Therefore, there is a growing and continuous interest in the study of the basic biology and genetics of toxigenic Penicillia because of their natural occurrence in fruit and fruit products and of the toxic effects of their secondary metabolites on humans (Puel et al., 2010, Frizzell et al., 2014; Patriarca et al., 2019).
PAT is the most mycotoxin found in apples and in their products which discompose a serious risk to the health of consumers. Its presence is related primarily to contamination by P. expansum in apples post-harvest (Paterson et al., 2006; Pianzzolla et al., 2004). Acute PAT exposure may lead to vomiting, nausea and other gastrointestinal symptoms such as intestinal haemorrhages, gastric ulcers, and lesions in the duodenum, as well as alterations in the intestinal barrier function accompanied with kidney damage (Speijers et al., 1988, Mahfoud et al.,2002, De Melo et al., 2012). Arnau et al., (2019) have reported that these alterations were observed in rats after a dose of 1 mg/kg bw in rats.
During the first part of this investigation, it was isolate 39 species belonging to 5 fungal genera which were identified as Penicillium, Aspergillus, Rhizopus, Alternaria, and Fusarium (Table 1, 2). The findings of this study showed that, Penicillium was the genus most frequently isolated from the fresh and dried apple. Among the 20 species of Penicillium, P. chrysogenumr and P. expansum were the most common species and was identified using molecular identification (Table 3) wherefore, dried apples was less contaminated by fungi, since dried apple contain very low moisture content compared to fresh apples, which make them less exposed infection by fungi.
Table 1. Total colony (TC per g), number of appearance out of 40 samples of fresh apple and frequency occurrence (FC)of fungal species from samples at 28C for 7-10 days on PDA cultural medium.
|
Fungal species |
Total count (TC) |
Total count% (TC %) |
Frequency of occurrence in 60 samples (FC) |
Frequency % (FC%) |
| Penicillium sp | 1716 | 64.7 | 84 | 44.2 |
| P. expansum | 618 | 23.3 | 30 | 15.8 |
| P. chrysogenum | 186 | 7.0 | 21 | 11.1 |
| Alternaria sp. | 38 | 1.4 | 12 | 6.3 |
| Aspergillus sp.
|
32 | 1.2 | 10 | 5.3 |
| Aspergillus flavus | 11 | 0.5 | 5 | 2.5 |
| Aspergillus niger | 21 | 0.8 | 14 | 7.4 |
| Rhizopus sp. | 8 | 0.3 | 3 | 1.6 |
| Fusarium sp.
|
21 | 0.8 | 11 | 5.8 |
| Total counts per
sample |
2651 | 100 | 190 | 100 |
Table 2. Total colony (TC per g), number of appearance out of 20 samples of dried apple and frequency occurrence (FC) of fungal species from samples at 28C for 7-10 days on PDA cultural medium.
|
Fungal species |
Total count (TC) |
Total count (TC) % |
Frequency of occurrence in 60 samples (FC) |
Frequency % (FC) |
|
Penicillium sp. |
27 |
49.1 |
84 |
44.2 |
|
P. expansum |
8 |
14.5 |
30 |
15.8 |
|
P. chrysogenum |
1 |
1.8 |
21 |
11.1 |
|
Alternaria sp. |
3 |
5.5 |
12 |
6.3 |
|
Aspergillus sp.
|
7 |
12.7 |
10 |
5.3 |
|
A. niger |
7 |
12.7 |
14 |
7.4 |
|
Fusarium sp.
|
2 |
3.7 |
11 |
5.8 |
| Total counts per
sample |
55 |
100 |
190 |
100 |
Table 3. Molecular identification of fungal strains recovered in the present study, their GenBank accession No. and % identity with closely related strains
| Fungal strains isolated
in the current study |
Closely related strains
accessed from GenBank |
|||||
|
Fungal species |
Strain No. |
Accession No. |
Fungal species |
GenBank No. |
Coverage (%) |
Identity (%) |
| P. expansum | RZ1 | GU561988.1 | P. expansum | GU561988.1 | 100% | 100% |
| P. chrysogenum | RZ2 | KT601570.1 | P. chrysogenum | KT601570.1 | 91% | 99.94% |
However, fruit drying is one of the oldest methods for preserving Fruits for centuries. Fruit may be dried as whole fruit e.g., grapes, in sliced form e.g., banana, mango, papaya, kiwi, apple etc.), in puree form e.g., mango, apricot (Ratti and Mujumdar, 2004). The drying of fruits allows for their better preservation by reducing water content, thus inhibiting microbial growth and enzymatic modifications. These products are thought to be resistant to microbial spoilage because of their low water activity, high acidity and sugar content, as a consequence of drying process.
In a recent study, Chalupowicz et al.,(2020) have reported that Penicillium appeared to be the most fungal genus infected apple. In addition, Barkai-Golan (2001) and Paola et al., (2008) reported that many Penicillium species were the causative agent of post-harvest diseases in which they infect a wide range of crop. DovPrusky et al., (2010) found that P. expansum responsible for rotting apple fruits, in which it lead pH to decreases from 3.95 to 4.31 in the healthy mesocarp to values ranging from 3.64 to 3.88 in the rotting tissue. The growth rate and PAT production by P. expansum are also largely influenced by environmental and endogenous factors of the substrate (Baert et al., 2007).
Since blue mould is a problem principally occurring during storage, temperature and atmosphere composition are the most important factors determining mould growth and mycotoxin production. Apples are usually stored under controlled conditions: low temperature (0.5–3.5◦C) in combination with reduced O2 (1–3%) and elevated CO2 (0.8–3%) (McCallum et al., 2002). PAT production has been observed at all temperatures allowing P. expansum and P. chrysogenum growth, encompassing an approximate range of 4–30◦C (Sommer et al., 1974).
Additional reports on fungi isolated from fresh and dried apple were reviewed by many authors (Sani et al., 2014, Shamsi et al., 2016, Lee et al., 2017, Basson et al., 2019). In Riyadh, Alwakeel (2013) isolated the fungi associated with apple spoilage which included P. chrysogenum, P. adametzii, P. steckii, and Aspergillus oryzae. The authors demonstrated the P. chrysogenum was the most frequent isolate.During the second part of this investigation, we were tested Penicillium isolates for ability to produce PAT in conditions in vitro using TLC method (thin-layer chromatography) (Table 4). TLC is a chromatography technique used to separate non-volatile mixtures (Harry et al., 1989).
Table 4. Screening of two selected isolates for the production of mycotoxins by TLC
|
Type of apple |
Toxin |
Tested Fungi |
Production of Patulin |
Characteristics |
| Fresh
apple |
Patulin |
Penicillium sp. (20)
P. chrysogenum (1) P. expansum (1) |
–
+ +
|
Yellow fluorescence after
exposure to 0.5% of 3-methyl-2 benzo-thiazolinone hydrazine hydrochloride (MBTH) spray followed by 15 min at 110C. |
| Dried
apple |
Patulin |
Penicillium sp (8)
P. chrysogenum (1) P. expansum (1) |
–
– –
|
The capacity of the two tested strains (P. expansum and P. chrysogenum) were producing PAT in conditions in vitro, the results were determined by the distance traveled by the substance being considered is divided by the total distance traveled by the mobile phase. (The mobile phase must not be allowed to reach the end of the stationary phase.) This ratio is called the retardation factor (Rf). In general, a substance whose structure resembles the stationary phase will have low Rf, while one that has a similar structure to the mobile phase will have high retardation factor. Retardation factors are characteristic, but will change depending on the exact condition of the mobile and stationary phase. For this reason, chemists usually apply a sample of a known compound to the sheet before running the experiment.
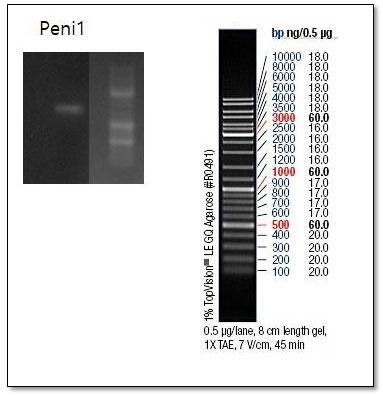
Figure 1. Gel electrophoresis of PCR product from P. chrysogenum strains. Data shown was obtained with ITS1/4 primer.
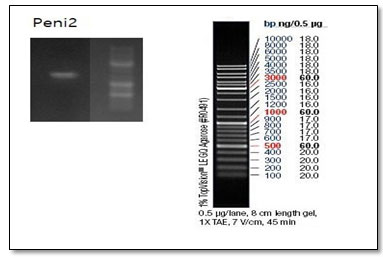
Figure 2. Gel electrophoresis of PCR product from P. expansum strains (Data shown was obtained with ITS1/4 primer).
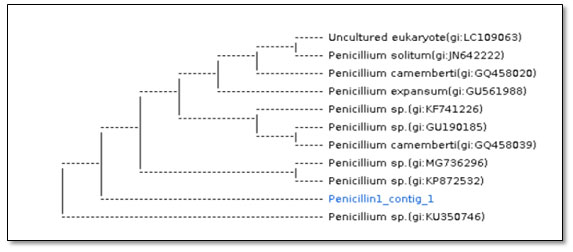
Figure 3: Dendrogram showing phylogenetic analysis based on the ITS region and NCBI GenBank database for P. chrysogenum strains (Sample name :Penicillin1_contig_1).
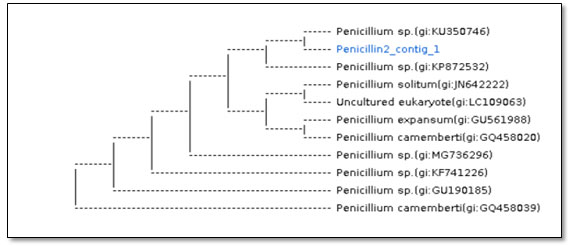
Figure 4: Dendrogram showing phylogenetic analysis based on the ITS region and NCBI GenBank database for P. expansum species (Sample name: Penicillin2_contig_1).
CONCLUSION
For human safety, food laws should be set up in Saudi Arabia for the occurrence of toxigenic fungi and mycotoxins, in fresh and dried apple even though the dried apple are exposed to heat before consumption due to the heat stability of mycotoxins.
REFERENCES
Alwakeel S (2013). Molecular identification of isolated fungi from stored apples in Riyadh, Saudi Arabia. Saudi Journal of Biological Sciences, 20(4), 311-317
Arnau Vidal, Ouhibi, S., Ghali, R., Hedhili, A., De Saeger, S., & De Boevre, M. (2019). The mycotoxin patulin: An updated short review on occurrence, toxicity and analytical challenges. Food and Chemical Toxicology, 129, 249-256.
Akinmusire, O.O., 2011. Fungal Species Associated with the Spoilage of Some Edible Fruits in Maiduguri Northern Eastern Nigeria. Advances in Environmental Biology, 2011; 5(1): 157-161.
Baert, K., Devlieghere, F., Flyps, H., Oosterlinck, M., Ahmed, M. M., Rajković, A., …& De Meulenaer, B. (2007). Influence of storage conditions of apples on growth and patulin production by Penicillium expansum. International Journal of Food Microbiology, 119(3), 170-181.
Barkai-Golan, R., 2001. Post-harvest diseases of fruits and vegetables, Development and Control. Amsterdam: Elsevier.
Basson, E., Meitz-Hopkins, J. C., & Lennox, C. L. (2019). Morphological and molecular identification of fungi associated with South African apple core rot. European journal of plant pathology, 153(3), 849-868.Chemosphere (CHEMOSPHERE)
Battacone G, Nudda A, Palomba M, Pascale M, Nicolussi P, Pulina G. (2005). Transfer of aflatoxin B1 from feed to milk and from milk to curd and whey in dairy sheep fed artificially contaminated concentrates. J. Dairy Sci.; 88:3063–3069.
Bilandzic, N., Varenina, I and Solomun, B. (2010). Aflatoxin M1 in raw milk in Croatia. Food Control, No. 21, pp:1279–1281.
Chalupowicz, D., Veltman, B., Droby, S., &Eltzov, E. (2020). Evaluating the use of biosensors for monitoring of Penicillium digitatum infection in citrus fruit. Sensors and Actuators B: Chemical, 311, 127896.
Chukwuka, K.S., Okonko, I.O., Adekunle, A.A., 2010. Microbial ecology of organisms causing pawpaw (Carica papaya L.) Fruit decay in Oyo State, Nigeria. American-Eurasian J. Toxicol. Sci., 2 (1): 43-50.
De Melo, F.T., de Oliveira, I.M., Greggio, S., Dacosta, J.C., Guecheva, T.N., Saffi, J., Henriques, J.A.P., Rosa, R.M., 2012. DNA damage in organs of mice treated acutely with patulin, a known mycotoxin. Food Chem. Toxicol. 50, 3548–3555.
DovPrusky, N.A., Itay, M., Shiri, B., Maayan, D., Droby S., 2010. Improving quality and safety of fresh fruits and vegetables after harvest by the use of biocontrol agents and natural materials. Acta Horticulture, 709: 45–51.
Erten, H., Agirman, B., Boyaci-Gunduz, C. P., Carsanba, E., &Leventdurur, S. (2019). Natural Microflora of Different Types of Foods. In Health and Safety Aspects of Food Processing Technologies (pp. 51-93). Springer, Cham.
Frizzell, C., Elliott, C. T., & Connolly, L. (2014). Effects of the mycotoxin patulin at the level of nuclear receptor transcriptional activity and steroidogenesis in vitro. Toxicology letters, 229(2), 366-373.
Harry W. Lewis & Christopher J. Moody. (1989). Experimental Organic Chemistry: Principles and Practice (Illustrated ed.). WileyBlackwell. pp. 159–173. ISBN 978-0-632-020171.
Henry SH, Whitaker T, Rabbini I, Bowers J, Park D, Price WD, Bosch FX, Pennington J, Verger P, Yoshizawa T, et al. (2001). Aflatoxin M1. In: Safety evaluation of certain mycotoxins in food. Prepared by the Fifty-sixth Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). FAO Food and Nutrition Paper No. 74. Rome (Italy): Food and Agriculture Organization of the United Nations.
Hussain I, Anwar J, Munawar MA, Asi M R (2008). Variation of levels of aflatoxin M1 in raw milk from different localities in the central areas of Punjab, Pakistan. Food Control; 19(12):1126-1129.
Kamkar, A., (2005). A study on the occurrence of aflatoxin M1 in raw milk produced in Sarab city of Iran. Food Control, 16: 593-9
Lee, S. Y., Park, S. J., Lee, J. J., Back, C. G., Ten, L. N., Kang, I. K., & Jung, H. Y. (2017). First report of fruit rot caused by Fusarium decemcellulare in apples in Korea. The Korean Journal of Mycology, 45(1), 54-62.
Mahfoud, R., Maresca, M., Garmy, N., &Fantini, J. (2002). The mycotoxin patulin alters the barrier function of the intestinal epithelium: mechanism of action of the toxin and protective effects of glutathione. Toxicology and applied pharmacology, 181(3), 209-218.
McCallum, J. L., Tsao, R., & Zhou, T. (2002). Factors affecting patulin production by Penicillium expansum. Journal of Food Protection, 65(12), 1937-1942.
Novotný, D., Lukáš, J., Brožová, J., & Růžičková, P. (2019). Comparison of the occurrence of fungi causing postharvest diseases of apples grown in organic and integrated production systems in orchards in the Czech Republic.
Paola, B., Carlo, B., Antonio, L., 2008. Risk assessment and safety evaluation of mycotoxins in fruits. Cited in: Mycototin production in fruit and vegetable. (eds: Rivka Barkai-Golan and NachmanPaster), Elsevier.
Patriarca, A. (2019). Fungi and mycotoxin problems in the apple industry. Current Opinion in Food Science.
Patriarca, A., da Cruz Cabral, L., Pavicich, M. A., Nielsen, K. F., & Andersen, B. (2019). Secondary metabolite profiles of small-spored Alternaria support the new phylogenetic organization of the genus. International journal of food microbiology, 291, 135-143.
Pitt, J.I., Hocking, A.D., 2009. Fungi and Food Spoilage. Springer Science and Business Media, Dordrecht Heidelberg.
Puel, O., Galtier, P., & Oswald, I. P. (2010). Biosynthesis and toxicological effects of patulin. Toxins, 2(4), 613-631.
Ratti, C. and Mujumdar A.S. (2004). Drying of Fruits. In: Processing Fruits Science and Technology (D.M. Barrett , L.P. Somogyi, Ramaswamy H.S., (ed.), 2nd ed., CRC Press, New York, USA, 127–161.
Rouissi, W. (2012). Study of thiabendazole resistance and volatile organic compounds production of Penicillium expansum strains (Doctoral dissertation, alma).
Ruangwises S , Saipan P and Ruangwises N (2013).Occurrence of Aflatoxin M1 in Raw and Pasteurized Goat Milk in Thailand http://dx.doi.org/10.5772/52723
Ruangwises, N., Saipan, P., &Ruangwises, S. (2011). Estimated Daily Intake of Afla‐ toxin M1 in Thailand. In: Guevara-González RG (ed.). Aflatoxins- Biochemistry and Occurrence of Aflatoxin M1 in Raw and Pasteurized Goat Milk in Thailand http://dx.doi.org/10.5772/52723 217.
Sani, I., Kutama A. S. and Nuhu A. A. 2014). “Isolation and Identification of Rot Fungi Associated with Fruits Sold in Some Markets within Kano Metropolis, Nigeria.” Asian J. of Adv. Basic Sci 3.1: 194-199.
Sebage. K. (2004) Mango Mountains, Burkino Faso. Tropical Whole Food Itdg.
Shamsi, S., Hosen, S., Al-Mamun, M., & Begum, M. (2016). Mycoflora associated with infected fruits of Momordicaco chinchinensis (Lour.) Spreng. Bangladesh Journal of Plant Taxonomy, 23(2), 181-188.
Shim W. B., Kolosova A.Y., Kim Y. J., Yang Z. Y., Park S. J., Eremin S.A., Lee I.S., and Chung D.H. (2004). Fluorescence polarization immunoassay based on a monoclonal antibody for the detection of OTA. International Journal of Food Science and Technology, 39: 829-837.
Singh, D., & Sharma, R. R. (2018). Postharvest diseases of fruits and vegetables and their management. In Postharvest Disinfection of Fruits and Vegetables (pp. 1-52).
Sommer, N.F, Buchanan J R, Fortlage R J. (1974). Production of patulin by Production of patulin by Penicillium expansum. Appl. Microbiol. 28:589-593.
Speijers, G.J.A., Franken, M.A.M., van Leeuwen, F.X.R., 1988. Subacute toxicity study of patulin in the rat: effects on the kidney and the gastro-intestinal tract. Food Chem. Toxicol. 26, 23–30.