Division of Microbial Technology, Post Graduate Research Department of Zoology, Chikkanna Government Arts College, Tirupur – 641602, Tamilnadu, India
Article Publishing History
Received: 09/02/2016
Accepted After Revision: 19/03/2016
The study was carried out for the antibacterial activity of metalloenzyme against Mutans streptococci. The epiphytic bacteria isolated from macroalgae by using traditional culture method were collected from west coast. Fifty epibiotic bacteria were isolated from the macroalgae. The metalloenzyme of the Bacillus spp was isolated and purified. The purified enzyme was tested against Mutans streptococci for their antimicrobial activity. Totally 50 isolates of Mutans streptococci were used for antimicrobial assay. 50µg, 100µg and 150µg of enzyme concentration were tested, among that 100µg of enzyme showed higher activity against mutans streptococci. Based on this study, it is concluded that metalloenzyme would serve as a potential antibacterial agent against Mutans streptococci.
Antimicrobial Activity, Bacillus Spp, Mutans Streptococci, Seaweeds Epibionts
Karikalan S, Mohankumar A. Antibacterial Efficacy of Marine Metalloenzyme Against Mutans Streptococci. Biosc.Biotech.Res.Comm. 2016;9(1).
Karikalan S, Mohankumar A. Antibacterial Efficacy of Marine Metalloenzyme Against Mutans Streptococci. Biosc.Biotech.Res.Comm. 2016;9(1). Available from: https://bit.ly/2qcAYEI
Intoduction
Mutans streptococci (MS) particularly Streptococcus mutans and Streptococcus sobrinus play a significant role in dental decay of the oral cavity and these species found in oral biofilms and Mutans streptococci (Streptococcus mutans and Streptococcus sobrinus) are the common significant bacteria in the pathogenesis of dental caries due to several epidemiological, investigational and animal studies (Who, 1997; Sullivan et al., 1996). This is owing to their ability of hasty lactic acid creation from dietary carbohydrates, mostly sucrose and glucose (Houte et al., 1991). In every individual’s oral cavity there is unrestricted production of microbial biofilms which is especially frequent for the reason that of dissimilar niches and abundant sources of nutrition in it. The individual’s micro flora is enormously complicated and strange group of microorganisms forming different links in the oral cavity thereby residing more than 700 dissimilar species (Metwalli et al., 2013 and Richards, 2015).
Bacteria are significantly the most leading form of microorganisms obtainable in the human oral cavity and little examples are S. sanguis, S. mitis, S. mutans, S. salivarius, S. mitis, L. acidophilus, L. salivarius, L. casei, Staphylococcus spp, Eubacterium spp, Neisseria spp, Actinomyces spp, Peptostreptococcus spp and Micrococcus spp, etc. Among them Streptococci was only form the biggest association in the oral cavity (Bhatia and Ichhpujani, 2003).
Cavities are the most common, chronic disease of early childhood. Oral diseases are infectious, progressive, often painful and expensive to treat. Poor oral health has been linked to diabetes, heart disease and other long-term problems in adults (CSN-SS, 2012). The primary goal of perinatal oral health care, with regard to caries transmission, is to lower the numbers of cariogenic bacteria in an expectant mothers mouth so that Mutans streptococci [MS] colonization of the infant can be delayed as long as possible (AAP, 2013).
Oral health is a significant component of physical condition and well-being. Oral diseases, which range from dental carries (cavities) to more prevalent infections, are problematic for millions of Americans and lead to serious consequences, including complications of major chronic conditions, debilitatins pain, absenteeism from work and school, nutrition issues, loss of teeth, impacts on children’s growth and social development, adverse pregnancy outcomes, inefficient use of emergency department services, and even death (INMRCI, 2011). Fortunately, dental disease and poor oral health can be easily prevented with usual access to dental care and efficient patient education. Specialized prophylaxis and fluoride or sealant purpose are proven intrusions that stop cavities and gum disease. Lacking treatment, gum disease may in the end destroy bone, connective tissue, and teeth, necessitate surgery (CDCP, 2011).
Few investigators have reported that it is perhaps non-Mutans streptococci organisms [nonMSO] and Actinomyces that add in the beginning stages of acidification, demineralization of enamel which results in aiding the MS and other oral bacteria to go in for capitalize the environment (Russell, 2009). Good oral health is necessary for eating and talking, and affects physical form and self-esteem (DHSV, 2003). Recent literature has shown that tongue cleanout show the ways to healthy oral environment (Alms et al., 2005).
A variety of compounds of controlling dental caries have been surveyed, such as antibiotics, enzymes, chlorhexidine, organic and inorganic fluoride. Enzymes such as dextranase reduced the amount of actively forming plaque but did not uniformly disperse the plaque already present. Little attention has paid to the elimination of the Streptococci by lytic enzyme and this type of enzymes may not give highly effect the oral and canal flora (Parsons, 1974).Hence, this present study has been carried out to assess the antibacterial activity of metalloenzyme on Mutans streptococci.
Material And Methods
West coast is located at the Kerala state region of Arabian Sea of India (Figure I). Samples were collected and transferred to the laboratory in sterilized translucent container containing Arabian Sea water. Seaweed samples were identified at CMFRI, Mandapam, and Tamilnadu. It was washed thoroughly with marine water and removed the epiphytes by swab method. Isolated the biofilm and serially diluted; only pure colonies were taken for further process of bacterial identification.Swab the epiphytic bacteria on seaweed. Bacteria were taken into the test tubes containing Zobell marine broth used as a transported media. All the test tubes were stored at 37ºC for 24 to 48 h for incubation period. MYP Agar is the suitable media to Bacillus spp. Polymyxin-B and Egg-Yolk Emulsion were additive chemicals for killing other gram positive and gram negative species on the media. Yellow color appearance of colonies and slightly agar surroundings changed to determined colonies of Bacillus spp. which were identified by the Morphological test and Biochemical characterization (Bergey’s Manual of Determinative Bacteriology).
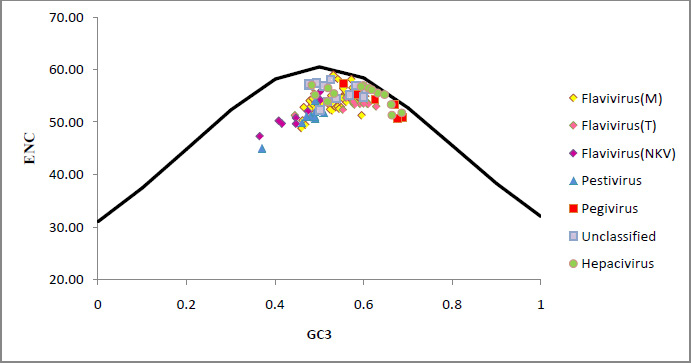 |
Figure 1: The Red seaweed Coralline spp |
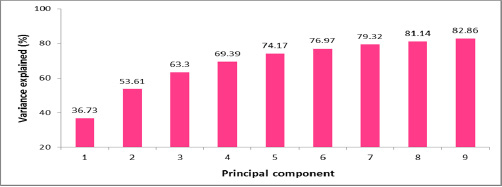 |
Figure 2: The antibacterial activity of
metalloenzyme on Mutans streptococci |
Production Of Metalloenzyme
Production and purification of metalloenzyme was done by the methods of Chakraborty and Paul (2008). The production medium was inoculated at 1.0 % [V/V] with 24 h old [OD = 0.6] culture of Bacillus spp and incubated at [15 ± 1] oC in a shaker at 80 rpm / min for 48 h. The growth cultures were then centrifuged at 4 oC [10,000 × g, 15 min]. The supernatant was used for further purification assay. Metalloprotease enzyme was purified from crude extraction of Bacillus spp by Ammonium sulphate precipitation, Dialysis method and DEAE Cellulose column chromatography.
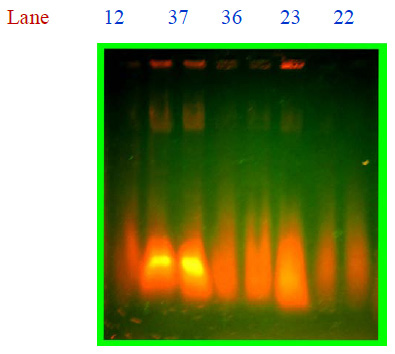 |
Figure 3: Purification of Metalloenzyme profile on DEAE column chromatography |
A specific weight of ammonium sulphate crystals was added to the crude enzyme gradually in an ice bath with continuous stirling for 30 min to get saturation percentage of 40 – 80 % and centrifuged at 10000 rpm at 4 ºC for 20 min. The precipitate was taken and dissolved in minimal amount of 0.1 M phosphate buffer and it was dialyzed against the same buffer for 24 h.
After the purification of dialyzed sample, it was subjected to purification by DEAE Cellulose column chromatography. 4 g of DEAE Cellulose was suspended in 25 mM buffer and kept overnight for equilibration. The column was carefully packed and equilibrated with the buffer at a constant flow rate of 30 ml / h. 3 ml of the dialyzed enzyme extract was diluted to 15 ml and loaded onto the column. The flow through was collected as a single fraction and tested for protease activity. The column was washed with approximately 100 ml of the equilibration buffer. The washings were collected as 3 ml fractions and tested for absorption at 600 nm. Fractions were collected till the absorption at 600 nm is zero. The washings were checked for protease activity. The equilibration buffer with 0.1M NaCl was used for elution. The column was eluted at a flow rate of 30 ml / h and 3 ml fractions were collected. The absorbance at 600 nm was recorded. The fractions with a high absorption at 600 nm were tested for protease activity. The fractions with the highest protease activity were pooled followed by equilibration and elution.
The agar well diffusion assay was used for antibacterial susceptibility test. Spread plates were prepared with proper concentration of inocula. 50µg, 100µg and 150µg of enzyme solution were prepared and placed in the well of agar plates. The plates were incubated for 24 hr at 37˚C. The inhibition zone was measured from the edge of the well to the inner margin of the surrounding bacterial growth. The bioassay was carried out under sterile condition in laboratory.
Results And Discussion
50 isolates of marine epiphytic bacteria were isolated from seaweeds species of Green algae, Red algae and Brown algae. These bacteria could acquire the necessary nutrition such as vitamin, polysaccharide and fatty acid from their animal or plant hosts; while on the other, they could excrete products such as amino acid, antibiotic and toxin propitious for the development of the hosts, or to chemical defense capability of the hosts (Armstrong et al., 2001).
Among the microbial sources, bacterial lipases, proteases and lytic enzymes are the most widely used class of enzymes in therapeutic and pharmaceutical applications, due to their high resistance and to high temperature (Jaeger and Eggert, 2002).
Metalloenzyme was purified by column chromatography, and totally 12 fractions were collected for enzyme activity. High values of metalloenzyme activity were observed (8th and 6th fraction) at 600nm in spectrophotometer 0.370 and 0.420 and low value was observed 0.08 at 12th fraction.Antibacterial activity was determined by using Mueller-Hinton Agar and all the chemicals were purchased from Hi Media Ltd., Mumbai. Lowest activity 9mm was observed in crude extract of Bacillus spp, in all concentrations of the enzyme injected (Table I). No activity was observed in the well with 150µg. Highest activity 17mm was observed in 100µg. Slight activity was observed in 50µg, 10mm. there was a significant differences observed in statistical interpretation. Only 100µg showed high rate of inhibitory effect than the other two concentrations. All the isolates of the Mutans streptococci were inhibited by 100µg concentration compared than 50µg and 150µg. These findings of the results were observed earlier against on clinical pathogens and lytic activity of lytic enzymes derived from soil Bacillus spp, particularly on Mutans streptococci (Kim
et al., 1999; Kim et al., 2003).
| Table 1: Antibacterial activity of the purified metalloenzyme on Mutans streptococci | ||||
| S. No | Name of the Algae | Name of the Bacillus strains | Enzyme concentration µg | Zone of Inhibition mm |
| 1. | Green seaweed | BL01 | 100 µg | 17mm |
| 2. | Red seaweed | BL05 | 50 µg | 10mm |
| 3. | Brown seaweed | BL09 | 150 µg | Nil |
Metalloenzyme could be valuable for the prevention of pathogenic biofilm of Mutans streptococci and considering that metalloenzyme was showed strong killing activity Bacillus spp BL01 can produce a high yield of enzyme. Therefore, the presence of metalloenzyme of this bacterial isolate could protect the human being against the infection by pathogenic Mutans streptococci and it might be utilized a good source for therapeutic agents production in pharmaceutical industries.
Acknowledgements
The authors wish to thank Dr. K. Shanmugasundharam, Principal, Chikkanna Government Arts College, and Tirupur for providing facilities to conduct the research.
References
Alms K., Al-Sanawi E. and Al- Shahrani B. (2005). The effect of tongue scraper on Mutans streptococci and Lactobacilli in patients with caries and periodontal disease. Odontostomatol Trop. Mar. 28(109):5-10
American Academy of Pediatrics (2003). Policy on oral health risk assessment timing and establishment of the dental home. Pediatrics 111(5Pt1):1113-6.
Armstrong E., Yan L., Boyd K.G., Wright P.C. and Burgess J.G. (2001). The symbiotic role of marine microbes on living surfaces. Hydrobiol 461; 37-40.
Bergey’s Manual of Determinative Bacteriology, www.uiweb.uidaho.edu/micro_biology/250/IDFlowcharts.pdf.
Bhatia R. and Ichhpujani R.L. (2003). Microbiology for Dental Students. 3rd ed. Jaypee Brothers.
Centers for Disease Control and Prevention (2011). Oral Health: Preventing Cavities, Gum Disease, Tooth Loss, and Oral Cancers: Ata Glance, http://www.cdc.gov/chronicdisease/resources/publications/aag/doh.htm.
Chakraborty K. and Paul R.R. (2008). Enrichment of eicosapentaenoic acid from sardine oil with D5-olefinic bond specific lipase from Bacillus licheniformis MTCC 6824. Journal of gricultural and Food Chemistry 56:1428–1433.
Child health news spring summer (2012). Why Oral Health Important. http://www.durham.ca/departments/health/dental.
Dental Health Services Victoria (2003). Clinical Analysis and Evaluation Unit, unpublished data. Institute of Medicine and National Research Council, Improving Access to Oral Health Care for Vulnerable and Underserved Populations (Washington, DC: The National Academies Press, 2011), http://www.hrsa.gov/publichealth/clinical/oralhealth/improvingaccess.
Institute of Medicine and National Research Council, Improving Access to Oral Health Care for Vulnerable and Underserved Populations (Washington, DC: The National Academies Press, 2011), http://www.hrsa.gov/publichealth/clinical/oralhealth/improvingaccess.
Jaeger K.E. and Eggert T. (2002). Lipases for biotechnology. Current Opinion of Biotechnology, 13; 390–397.
Kim K.J., Yang Y.J. and Kim J.G. (2003). Purification and characterization of chitinase from Streptomyces sp. M-20. J. Biochem. Mol. Biol. 36 (2): 185-189.
Kim S.Y., Shohk B.D.H. and Yu J.H. (1999). Purification and properties of Bacteriolytic enzymes from Bacillus licheniformis YS-1005 against Streptococcus mutans. Biosci.Biotechnol.Biochem. 63(1); 73-77.
Metwalli K.H., Khan S.A., Krom B.P. and Jabra-Rizk M.A. (2013). Streptococcus mutans, Candida albicans and the Human Mouth: A Sticky Situation. PLOS, Pathogens 9(10):1-4.
Parsons J.C. (1974). Chemotherapy of dental plaque- A review. J. Periodontal., 45: 177-186.
Richards DJW (2015) Molecular basis of Streptococcus mutans sortase A Inhibition by the flavonoid natural product trans-chalcone Chem Comm.51 10483-10485
Russell R.R. (2009). Changing concepts in caries microbiology. Am J Dent., 22 (5): 304-310.
Sullivan A., Borgstrom M., Granath L. and Nilsson G. (1996). Number of Mutans streptococci or Lactobacilli in a total dental plaque sample does not explain the variation in caries better than stimulated whole saliva. Comm. Dent. Oral. Epid. 24: 159-163.
Van Houte J., Sanson C., Joshipura K. and Kent R. (1991). Mutans streptococci and non Mutans streptococci acidogenic at low pH, and in vitro acidogenic potential of dental plaque in two different areas of the human dentition. J. Dent. Res. 70: 1503-1507.
WHO (1997). Oral health surveys. Basic method. 4th ed. Geneva. www.ncbi.nlm.nih.gov/pubmed/26029850 July 4: 51 (52)


