1Department of Environmental Science, Arts, Commerce and Science College, Ambad, Maharashtra-431204, India
2ICAR-Indian Institute of Soil Science, Bhopal, Madhya Pradesh–462038, India
Article Publishing History
Received: 10/12/2018
Accepted After Revision: 25/02/2019
The River Godavari serves as a source of water for drinking and domestic use, agricultural irrigation and industries in Ambad stretch. The anthropogenic activities in the stretch caused ecological disturbance through water pollution. In present study, the spatial variation in water quality parameters was studied with respect to the fish population and diversity. The fish diversity and water quality of Godavari River at Ambad Stretch (16 km length) was studied at five identified sampling sites viz. Paithan (R), Balegaon (A), Gandhari (B), Shahagad-A (C) and Shahagad-B (D). The results revealed that the water quality followed the trend: R>D>B>A>C. The fi sh diversity index was observed between 2.35 and 3.03 among different sampling sites with highest at site R. The correlation study showed significant correlation between water quality parameters viz. total dissolved solids, phosphate, nitrate, dissolved oxygen, biological oxygen demand and chemical oxygen demand and fi sh population and diversity. The pollution tolerant fish species Oreochromic mossumbicus population showed significant positive correlation with water quality parameters except dissolved oxygen where it was found negatively correlated.
Godavari River, Fish Diversity, Water Quality, Shannon Diversity Index, Ambad Stretch
Bhutekar D. D, Aher S. B. Biological Monitoring of Riverine Ecosystem and its Correlation With Water Quality. Biosc.Biotech.Res.Comm. 2019;12 (1).
Bhutekar D. D, Aher S. B. Biological Monitoring of Riverine Ecosystem and its Correlation With Water Quality. Biosc.Biotech.Res.Comm. 2019;12(1). Available from: https://bit.ly/2IkJZ3M
Copyright © Bhutekar and Aher et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
Introduction
The water is the most essential and precious resource and is the elixir of life. Water resources comprising of surface water (river and lakes), ground water, and marine and coastal waters support all living things including human beings. Surface Rivers have always been the lifelines of development but the freshwater habitats receiving the highest levels of human disturbance. Recent studies on the status of inland water ecosystems showed that the river catchments in the Indian subcontinent are globally most threatened. Therefore, it is essential to identify, monitor and conserve the riverine ecosystems. Biological assessment of the freshwater habitats aims at char-acterizing and monitoring the conditions of the aquatic resources. The assessments are commonly associated with human impact (Saunders et al., 2002; Resh, 2008; Bhutekar et al., 2015, Bhutekar et al., 2018a).
In India various studies have presented spatial and temporal trends in diversity or biotic index of streams, rivers and lakes (Bhat, 2002). But still relatively little information is available on the correlation between water quality parameters and population and diversity of the aquatic animals (Rai et al., 2013). The river Godavari is considered to be one of the very sacred rivers of India. But being the ultimate sink of anything and everything
drained through surface runoff, the river has been subjected to considerable stress. As a result, the ecology and water quality has suffered. In India some studies have presented spatial and temporal trends in diversity or biotic index of streams, rivers and lakes (Bhat, 2002). The biomonitoring system developed for the temperate streams was tested and found useful. However, biomonitoring can not entirely replace standard physic-chemical
water quality methods. Standard physico-chemical water quality measures provide information on water quality at a particular spatial unit during the time of sampling. It cannot provide historical information on water quality (Bhutekar et al., 2018b). On the other hand, by knowing the ecology of aquatic insect community, biomonitoring tools provide some historic insights into the water quality. Standard physico-chemical water quality methods need to be carried out in conjunction with biomonitoring tools to comprehensively evaluate the health of freshwater ecosystems (Glorian et al., 2018).
River Godavari serves as a source of water for drinking and domestic use, agricultural irrigation and industries in Ambad stretch. Therefore, it was felt appropriate to evaluate, monitor and improve water quality and ecological conditions of river Godavari at Ambad stretch. The attempt was made to study the correlation between the water quality parameters and fi sh population and diversity.
Materials and Methods
Sampling site
Present work was carried out on Godavari River water at Ambad Stretch. The total length of the study stretch was around 16 km. The five different locations were identified and fish diversity was studied by conducting seasonal fi sh sampling. Fishes of this river system were sampled seasonally over a period of two years i.e. July 2012 to May 2014 at 5 sampling sites. The sites were selected on the basis of depth and availability of water. The distance between two sampling sites was approximately 3 km. The five sampling sites were Paithan, Balegaon, Gandhari, Shahagad-A and Shahagad-B (Table 1).
Fish sampling, identification and diversity computation Before starting the sampling, a thematic map was developed denoting the sampling sites where GIS based information was used. On basis of the map a pilot survey was carried out prior to actual sampling. Depending on the suitability and participatory appraisal with local fisherman community the sampling station were decided. Sampling efforts i.e. a single gill net operation or cast net operation for one hour was conducted and a species accumulation curve was obtained which was used to calculate the minimum sampling efforts vs. sampling effort plot.Based on such pilot survey carried out at many sampling sites, a sampling effort for 3 hours by gill net and drag net operation was used as standard for the sampling of fishes at all sites under this study.Fish samples were collected seasonally viz. monsoon, winter and summer for both years at each sampling station using different nets and gears of local fisherman. Sampling was carried out using a variety of fishing nets traditionally used by local fisherman comprising varying mesh sizes of gill, cast nets and drag nets.The fishes were identified and some representative specimens were collected and preserved in (4% formaldehyde solution) in glass jars. The identification of the fish specimens from various stations of the river Godavari was carried out with standard methods given by Mishra (1962), Jayaram (1981; 1999; 2006), Fischer and Bianchi (1984) and Jhingran (1997). In addition to this, electronic database like catalogue of life (2005), fi shebase (2004) and FAO fish identification sheets were also used during fish identification.The identified fishes were grouped as per standard chronology of standard classification and Shannon
Index was computed (Shannon, 1948) as follows:
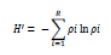
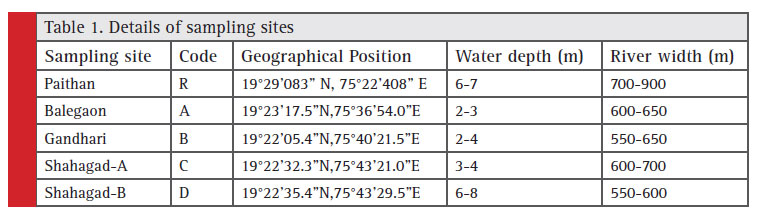 |
Table 1: Details of sampling sites |
Where
H’- Shannon Index
pi – proportion of individuals belonging to the ith species
in the dataset of interest.
Water sampling and analysis
Water samples were collected from each sampling stations (Table 1) in first week of every month during June 2012 to May 2014 in morning hours (between 7.00 to 10.00 am). Water temperature was recorded and dissolved oxygen was fixed at sampling site. The collected water samples were analyzed in laboratory for different physical and chemical parameters as per the standard methods (APHA, 2005; Trivedi and Goel, 1984). The
parameters studied were temperature, turbidity, pH, total dissolved solids, total alkalinity, total hardness, phosphate, nitrate, chlorides, dissolved oxygen, biological oxygen demand and chemical oxygen demand.
Statistical analysis
The collected data was compiled and subjected to statistical analysis for computation of coefficient of determination (r2) and correlation coefficient (r) as follows and the relationship between two parameters was studied.
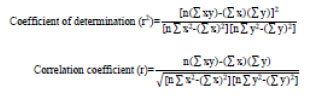
Results and Discussion
Fish diversity and population
During the study period, the fishes belonging to ten different families viz. Notopteridae, Cyprinidae, Bagridae, Siluridae, Heteropneustidae, Mugilidae, Beonidae, Mastacembelidae, Cichlidae and Channidae were observed in Godavari River at Ambad stretch (Table 2). Similarly,
during the study period, total 25 fish species were identified in River Godavari at Ambad stretch at different sampling sites viz., R- Paithan, A-Balegaon, B-Gandhari, C-Sahagad A and D-Sahagad B. The identified fish species were Notopterus notopterus, Chitala chitala, Hypothalmicthys molitrix, Salmostoma novacula, Chela laubuca, Cyprinus carpio carpio, Thynicthys sandkhol, Osteobrama vigorsii, Puntius ticto, Cirrhinus mirgala,
Catla catla, Labeo rohita, Garra mullya, Aorichthys aor, Ompak bimaculatus, Wallago attu, Heteropneustes fossilis, Rhinomugil corsula Xanthodon cansula, Mastacembelus armatus, Mascrognathus pancalus, Oreochromis mossambicus, Channa muralius, Channa straita and Chanda nama.
(Table 2).
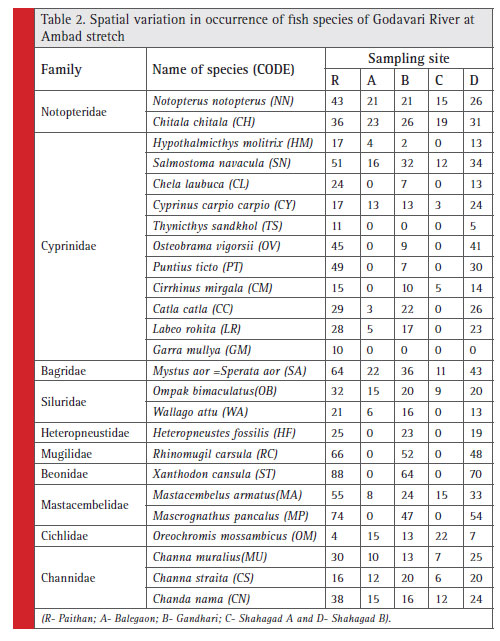 |
Table 2: Spatial variation in occurrence of fish species of Godavari River at Ambad stretch |
The fish population at different sampling sites ranged 133-881 individuals. The highest and lowest fish population was found at site R and site C, respectively. The fish population at study sites followed the trend: R>D>B>A>C (Table 3). The family wise distribution of fish population at different sites revealed that, species from all families were observed at all sites except the species of family Heteropneustidae, Mugilidae and Beonidae
which was not observed at site A and site C (Table 2). The total fish families and species reported at sampling sites R, A, B, C and D was 10 and 25, 7 and 15, 10 and 23, 7 and 12; and 10 and 24, respectively (Table 3). The pollution tolerant fi sh species Oreochromis mossambicus was found dominant at site C followed by site A and B. The species was least dominant at site R and D.
The Shannon diversity index value observed between 2.35 and 3.03 among different sampling sites of Godavari River at Ambad stretch. The lowest value of 2.35 was observed at site C.The results clearly indicated that the maximum Shannon Index value was found at reference and at site D (3.03). The diversity index of site A and B were 2.56 and 2.93 respectively. The higher diversity index at site R might be due to the undisturbed habitat whereas the dilution and self purification process of the river water resulted in higher value of fish diversityat site D. The water quality at site R and D was found suitable for fish growth. The higher degree of the pollution and habitat disturbance made the water quality unsuitable for the growth of the fish community which can be evidenced from lower diversity index value at site C (2.35) and site A (2.56).
Physico-chemical properties of Godavari River water
Temperature, turbidity and pH
The spatial variation in water quality of the Godavari River is presented in Table 4. The average water temperature was observed between 23.3 and 23.6°C among different sampling locations. Temperature plays very important role in the physiological behavior and distribution of aquatic organisms. The variation in river water temperature usually depends on the season, geographic location, ambient air temperature and chemical reaction in a water body (Ahipathi and Puttaiah, 2006). However, no significant difference in site specific temperature was observed during the investigation. Turbidity measures water clarity or the ability of light to pass through water. In present investigation water turbidity was observed between 7.0 and 23.4 NTU among different sampling sites (Table 4). The lowest average turbidity was observed at reference location (R) followed by B and D site. The highest turbidity was recorded for site C (23.4 NTU) followed by site A (17.1 NTU). Surface-runoff, stream fl ow and overland fl ow in natural waters increase the turbidity levels in water. The higher level of pollutants in water also imparts turbidity to water. Yadav and Kumar (2011); Medudhula et al. (2012); Dhawde et al. (2018) and Bhutekar et al. (2018a) reported the similar observations. The pH of the water was found slightly basic at all the sampling sites during the study period (Table 4). The mild alkaline nature of river water attributed to the presence of CO2 in water as bicarbonate (Azeez et al., 2000). The leaching of basic rock material by rainwater and carried by surface runoff to river stream attributed to
the higher values of water pH during rainy season (Lalparmawii, 2007).
 |
Table 3: Fish population and diversity of Godavari River at various sites |
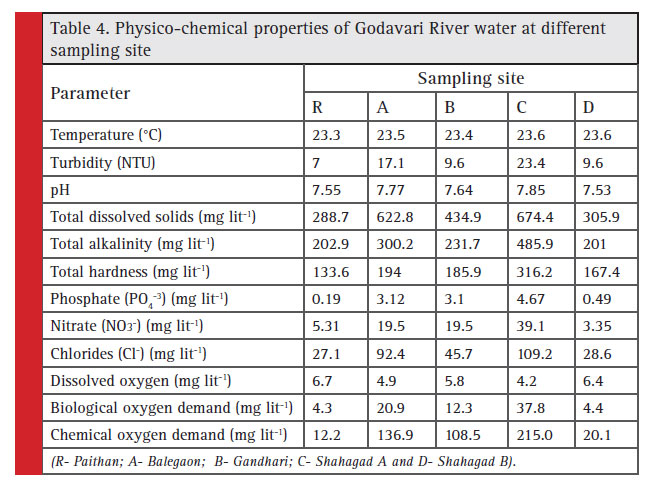 |
Table 4: Physico-chemical properties of Godavari River water at different sampling site |
Dissolved solids, alkalinity and hardness
In the present investigation TDS showed variation at all the sites during study period. The TDS ranged between 288.7 mg l-1 and 674.4 mg l-1 among different sampling locations (Table 4). The highest value of TDS was observed at site C followed by site A (622.8 mg l-1). Total dissolved solids (TDS) are a measure, of the amount of dissolved materials in the water and are mainly contains minerals (Senthilnathan et al., 2011).
Alkalinity is the measure of buffering capacity of the water. It is generally imparted by the salts of carbonates, bicarbonates, phosphate, nitrates etc. (Yellavarthi, 2002). In present investigation the TA ranged from 201.0 mg l-1 (site D) to 485.9 mg l-1 (site C) among different sampling sites (Table 4). The increase in total alkalinity due to various religious activities, domestic waste and especially due to soaps and detergents was earlier reported by Patil (2003); Mithani et al. (2012) and Dhawde et al. (2018).
The total hardness ranged from 133.6 mg l-1 at site R to 316.2 mg l-1 at site C (Table 4). Hardness of water is a measure of its capacity to produce lather with soap (Garnaik et al., 2013). Total Hardness is an important parameter of water quality whether it is used for domestic, industrial or agricultural purposes (Jothivenkatachalam et al., 2010). The cations of calcium, magnesium, iron and manganese contribute to the hardness of water
(Shrivastava and Patil, 2002). The widespread abundance of these metals in rock formations leads often to very considerable hardness levels in surface and ground waters (EPA, 2001). Similarly, the Ca and Mg that enter the water bodies through residues of soaps, detergents and parent bed rock materials made up of Ca, Mg and other metal ions also signifi cantly contributes to total hardness of water (Nanda, 2005).
Phosphate, nitrate and chlorides
The phosphate concentration in Godavari River water at different sampling location was ranged between 0.19 mg l-1 and 4.67 mg l-1 (Table 4). The highest phosphate concentration was found at site C followed by site A and site B. The animal waste, agriculture waste and detergent in domestic wastewater may have contributed towards the observed increment in phosphates (Anda et al., 2001). Sinha et al. (1998) have also reported higher phosphate content in lower stretch of Ganga River. Nitrate concentration in Godavari River water sampled at different locations was ranged between 3.35 mg l-1 and 39.1 mg l-1 with mean value of 17.3 mg l-1 (Table 4). The highest concentration of nitrate was found at site C followed by site A and B (19.5 mg l-1). The lowest concentration of nitrate was observed at site D (3.35 mg l-1). Nitrate in surface water is an important factor for water quality assessment (Jhones and Burt, 1993) which is mainly contributed waste discharges and artificial nitrogenous fertilizers. The chloride in Godavari River water was ranged from 60.6 mg l-1 to 109.2 mg l-1 at various sampling locations (Table 4). The highest concentration of chlorides was observed at site C followed by site A and site B. Venkatesharaju et al. (2010) and Bhutekar et al. (2018a) also observed similar results with respect to chloride
contents of the river water.
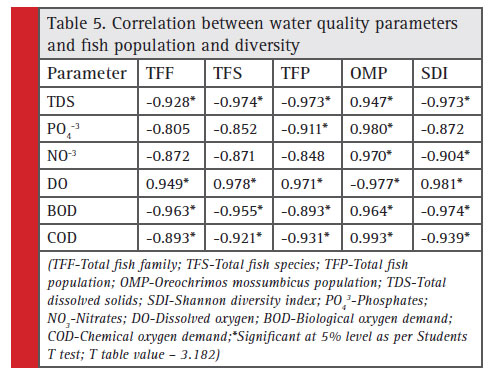 |
Table 5: Correlation between water quality parameters and fish population and diversity |
Dissolved oxygen, Biological oxygen demand and Chemical oxygen demand
The sampling station wise mean dissolved oxygen data showed that, DO ranged between 4.2 mg l-1 to 6.7 mg l-1. The highest DO values was recorded for sampling site R followed by D and B. The mean monthly DO concentration showed that, it was ranged between 4.7 mg l-1 and 7.0 mg l-1 (Table 4). Dissolved oxygen is one of the important parameter in water quality assessment as it regulates and governs metabolic activities and metabolism of the biological community as a whole, respectively and also acts as an indicator of trophic status of the water body (Saksena and Kaushik, 1994). Its presence is essential to maintain variety of forms of biological life in water. Similar observations were also recorded by Singh and Gupta (2010) and Bhutekar et al. (2018a). The biological oxygen demand of Godavari River water was observed between 4.3 mg l-1 and 37.8 mg l-1 among different sampling sites (Table 4). The highest BOD was observed at site C followed by site A (20.9 mg l-1). The lowest BOD was observed at site R (4.3 mg
l-1) followed by site D (4.4 mg l-1). Biodegradation of organic materials exerts oxygen tension in the water and increases the biochemical oxygen demand (Abida, 2008). BOD increases with organic matter addition, wastewater or urban storm water runoff took place at the river water. The higher BOD in polluted water was reported earlier by Garg et al. (2006); Zainudin et al. (2010); Glorian et al. (2018).
The COD is a measure of oxygen equivalent to the organic matter content of the water susceptible to oxidation and thus is an index of organic pollution in river (Khaiwal and Anubha, 2003).In present investigation the COD was observed 12.2 and 215.0 mg l-1 among different sampling sites (Table 4). The highest value of COD was observed at site C followed by site A (136.9 mg l-1) and site B (108.5 mg l-1). The lowest value of COD was observed for site R (12.2 mg l-1) followed by site D (20.1 mg l-1). Highest COD at site C indicated the higher pollution of water while lower level of COD indicated low level of pollution of water at the study area (Waziri and Ogugbuaja, 2010).Similar result was also reported by Bamniya et al. (2011); Bhutekar et al. (2018b) and Dhawde et al. (2018).
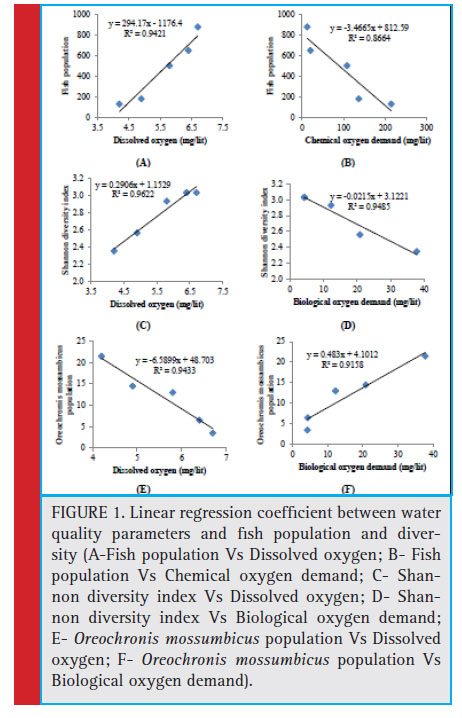 |
Figure 1 |
Correlation between water quality and fish population and diversity
The water quality of a water body is determined by measuring the physic-chemical properties of the water. Among the criteria parameters, dissolved oxygen, biological oxygen demand, total dissolved solids, chemical oxygen demand, nitrate and phosphate are considered as key parameters. The correlation between these parameters and fi sh diversity/population was studied and its significance was interpreted using Student’s T Test (Table 5).
The fish population, fish diversity, number of fish families observed, number of fish species observed and the population of pollution tolerant fish species Oreochromis mossumbicus was studied in relation with the water quality parameters. The data showed significant negative correlation between total fish families observed and total dissolved solids (r=-0.928), biological oxygen demand (r= -0.963) and chemical oxygen demand (r=
-0.893) whereas significant positive correlation with dissolved oxygen (r=0.949). The similar type of relationship was also existed with total fi sh species observed and total fish population recorded. The total fish population showed significant positive correlation with dissolved oxygen (r= 0.971; Fig. 1-A) and significant negative correlation with biological oxygen demand (r= -0.893) and chemical oxygen demand (r= -0.993; Fig. 1-B). The
nitrate content did not show significant correlation with total fish families, species and population. The phosphate content also did not shown correlation with TFF and TFS but showed significant negative correlation with total fish population. It is interesting to note that, the pollution tolerant fi sh species Oreochromis mossumbicus showed significant positive correlation with TDS (r= 0.947); phosphate (r= 0.980); nitrate (r= 0.970); BOD (r= 0.964) (Fig. 1-F) and COD (r= 0.993). The Oreochromis mossumbicus showed significant negative correlation with dissolved oxygen (r= -0.977) (Fig.1-E). The Shannon diversity index showed signifi cant positive correlation with dissolved oxygen (r= 0.981; Fig. 1- C)
whereas significant negative correlation with TDS (r= -0.973); nitrate (r= -0.904); BOD (r= -0.974; Fig. 1-D) and COD (r= -0.939).
The aquatic biodiversity of world is changing and getting depleted alarmingly fast as a result of extinctions caused by habitat loss, pollution, introduction of exotic species, over exploitation and other anthropogenic activities. Fishes are the keystone species which determine the distribution and abundance of other organisms in the ecosystem they represent and are good indicators of water quality and aquatic ecosystem. The fresh water
fi shes are one of the most threatened taxonomic groups (Darwall and Vie 2005) because of their high sensitivity to the quantitative and qualitative alteration of aquatic habits (Laffaille et al., 2005; Kang et al., 2009; Sarkar et al., 2008). Therefore, fishes are often used as bioindicator for the assessment of water quality, river network connectivity or flow regime (Chovance et al., 2003; Matta et al., 2018). Present investigation, clearly indicated a significant correlation of water quality parameters and fish diversity. The higher population of Oreochromis mossambicus at site C and B indicated it is a pollution tolerant species and favors such habitat (Murugan and Prabaharan, 2012). The healthy population of remaining species at site R, site D and site B may be attributed to the favorable water quality as evidenced from the data on water quality parameters (Table 4). The observed dominant species at site R and D are fresh water fishes (Munshi and Srivastava, 2006; Teta et al., 2017).
Conclusion
The results of the present investigation revealed that, the water quality followed the trend: R>D>B>A>C. The fish diversity index was observed between 2.35 and 3.03 among different sampling sites with highest at site R. The correlation study showed significant correlation between water quality parameters viz. total dissolved solids, phosphate, nitrate, dissolved oxygen, biological oxygen demand and chemical oxygen demand and
fish population and diversity. The pollution tolerant fish species Oreochromic mossumbicus population showed significant positive correlation with water quality parameters except dissolved oxygen where it was found negatively correlated.
References
Abida, B. (2008). Study on the quality of water in some streams of Cauvery River. e-Journal of Chemistry, 5(2): 377-384.
Ahipathi, M.V. and Puttaiah, E.T. (2006). Ecological characteristics of Vrishabhavathi river in Bangalore (India). Environmental Geology, 49: 1217-1222.
Anda, J., Shear, H., Maniak, U. and Riedel, G. (2001). Lakes & Reservoirs: Research and Management, 6: 313–321.
APHA. (2005). Standard Methods of Water and Wastewater. 21st Edn., American Public Health Association, Washington, DC. 2-61.
Azeez, P.A., Nadarajan, N.R. and Mittal, D.D. (2000). The impact of monsoonal wetland on ground water chemistry. Pollution Research, 19(2): 249–255.
Bamniya, B.R., Mahecha, G.S., Kapoor, C.S. and Kapasya, V. (2011). Water quality status of two water reservoirs of Thar desert city, Jaisalmer. Indian Journal Environment and Eco planning , 18(1): 113-116.
Bhat, A. (2002). A study of the diversity and ecology of the freshwater fi shes of four river systems of Uttara Kannada District, Karnataka, India. Ph.D. Thesis submitted to Indian Institute of Science, Bangalore, Pp. 178.
Bhutekar, D.D., Aher, S.B. and Babare, M.G. (2018a) Spatial and seasonal variation in physico-chemical properties of Godavari River water at Ambad region, Maharashtra. Journal of Environment and Biosciences, 32(1):15-23.
Bhutekar, D.D., Aher, S.B. and Babare, M.G. (2018b) Status of water quality and strategies for conservation of Pushkarni reservoir, Ambad. International Journal of Recent Trends in Science and Technology, Special Issue, ACAEE: 2018, 110-113.
Chovance, A., Hoffer, R. and Schiemer, F. (2003). Fish as bioindicators. In: Market, B.A., Breure, A.M. and Zechmeiser, H.G. (eds) Bioindicatos and biomonitors, Pp. 639–675.
Darwall, W.R.T. and Vie, J.C. (2005). Identifying important sites for conservation of freshwater biodiversity: extending the species- based approach. Fisheries Management Ecology, 12: 287-293.
Dhawde, R., Surve, N., Macaden, R., Wennberg, A., Seifert- Dähnn, I., Ghadge, A. and Birdi, T. (2018). Physicochemical and Bacteriological Analysis of Water Quality in Drought Prone Areas of Pune and Satara Districts of Maharashtra., India. Environments, 5(61): 1-20.
EPA (2001). Parameters of Water Quality, Interpretation and Standards, Environmental Protection Agency, Ireland. Fischer, W. and Bianchi, G. (1984). FAO Species Identifi cation Sheets for Fishery Purposes: Western Indian Ocean Fishing Area. Food and Agriculture Organisation of the United Nations, Rome. Pp. 51
Garg, R.K., Saksena, D.N. and Rao, R.J. (2006). Assessment of physic-chemical water quality of Harsi Reservoir, district Gwalior, Madhya Pradesh. Journal of Ecophysiology and Occupational Health, 6: 33-40.
Garnaik, B., Panda, S., Tripathy, S. and Tripathy, U. (2013). Seasonal Variation of Nagavali River Water Quality at the Vicinity of Paper Mill near Jaykaypur, Odisha, India. International Research Journal of Environment Sciences, 2(5): 46-52.
Glorian, H., Börnick, H., Sandhu, C. and Grischek, T. (2018). Water Quality Monitoring in Northern India for an Evaluation of the Efficiency of Bank Filtration Sites. Water, 10 (1804):1-15.
Jayaram, K.C. (1981). The freshwater Fishes of India, Pakistan, Bangladesh, Burma and Sri Lanka. Hand Book of Zoological Survey of India, Calcutta, Pp. 475.
Jayaram, K.C. (1999). The fresh water fi shes of the Indian region. Narendra Publishing House, Delhi, Pp. 551. Jayaram, K.C. (2006). Catfi shes of India. Narendra Publishing House, Delhi, Pp.386.
Jhingran, V.G. (1997). Fish and Fisheries of India, 3rd edition, corrected reprint. Hindustan Publishing Corporation, Delhi, India. Pp. 727.
Jones, P.J. and Burt, P.T. (1993). Nitrate in Surface Waters. In: Burt, T.P., Heathwaite, A.L. and Trudgill, S.T. eds. Nitrate: Processes, Patterns and Management. John Wiley & Sons, Chichester, 269-317.
Jothivenkatachalam, K., Nithya, A. and Chandra, M.S. (2010). Correlation analysis of drinking water quality in and around Perur block of Coimbatore district, Tamil Nadu, India. Rasayan Journal of Chemistry, 3(4): 649-654.
Kang, B., He, D., Perrett, L., Wang, H., Hu, W., Deng, W. and Wu, Y. (2009). Fish and fisheries in the Upper Mekong: current assessment of the fish community, threats and conservation. Reviews in Fish Biology and Fisheries, 19: 465–480.
Khaiwal, R. and Anubha, K. (2003). Seasonal variations in physicochemical characteristics of River Yamuna in Haryana and its ecological best-designated use. Journal of Environmental Monitoring, 5: 419-426.
Laffaille, P., Acou, A., Guillouet, J. and Legult, A. (2005). Temporal change in European eel, Anguilla anguilla, stock in a small catchment after installation of fish passes. Fisheries Management Ecology, 12: 123–129.
Lalparmawii, S. (2007). Analysis of water quality and Biomonitoring of Tuirial River in vicinity of the Hydel Project in Mizoram. Ph.D Thesis. Mizoram University, Aizawl. Matta, G., Gjyli, L., Kumar, A. and Machel, J. (2018). Hydrochemical characteristics and planktonic composition assessment of River Henwal in Himalayan Region of Uttarakhand using CPI, Simpson’s and Shannon-Weaver Index. Journal of Chemical and Pharmaceutical Sciences, 11(1): 122-130.
Medudhula, T., Samatha, C. and Sammaiah, C. (2012). Analysis of water quality using physico-chemical parameters in lower manair reservoir of Karimnagar district, Andhra Pradesh. International Journal of Environmental Sciences, 3(1): 22-25.
Mishra, K.S. (1962). An aid to the identifi cation of the common commercial fi shes of India and Pakistan. Records of the Indian Museum, 57(1-4): 320.
Mithani, I., Dahegaonkar, N.R., Shinde, J.S. and Tummawar, S.D. (2012). Preliminary studies on physico-chemical parameters of river Wardha, District Chandrapur, Maharashtra. Journal of Research in Ecology, 1: 14-18.
Munshi, D. and Srivastava, J.S. (2006). Natural History of Fishes and Systematics of Freshwater Fishes of India. Narandra Publishing House, Delhi. Pp. 423.
Nanda, S.P. (2005). Environmental management plan (EMP) for Bhubaneswar, India.
Patil, D.S. (2003). Ecological studies in some lentic water bodies of Kolhapur city. Ph.D. Thesis, Shivaji University, Kolhapur. Rai, A.K., Diba, F. and Paul, B. (2013). A study on the seasonal variations of different physic-chemical water quality parameters of Indrapuri Dam Rohtas District Bihar. International Journal of Environmental Sciences, 2(3): 125-129.
Resh, V.H. (2008). Which group is best? Attributes of different biological assemblages used in freshwater biomonitoring programs. Environmental Monitoring and Assessment, 138:131-138.
Saksena, D.N. and Kaushik, S. (1994). Trophic status and habitat ecology of entomo fauna of three water bodies at Gwalior, Madhya Pradesh. In: Agrawal, O.P. Eds, Perspective in entomological research Scientifi c Publishers, Jodhpur.
Sarkar, U.K., Pathak, A.K. and Lakra, W.S. (2008). Conservation of freshwater fish resources of India: new approaches, assessment and challenges. Biodiversity Conservation, 17: 2495–2511.
Saunders, D.L., Meeuwig, J.J. and Vincent, A.C.J. (2002). Freshwater protected areas: Strategies for Conservation. Conservation Biology, 16(1): 30-41.
Senthilnathan, T., Parvathavarthini, K.V. and George, S.M. (2011). Assessment of ground water quality of Maraimalai Nagar town near Chennai, India. Journal of Environmental Research and Development, 5(4): 943-955.
Shannon, C.E. (1948). A mathematical theory of communication. The Bell System Technical Journal, 27: 379–423.
Shrivastava, V.S. and Patil, P.R. (2002). Tapti river water pollution by industrial wastes: A statistical approach. Nature, Environment, Pollution and Technology, 1: 279-283.
Singh, M.R. and Gupta, A. (2010). Seasonal variation in certain physicochemical parameters of Imphal, Iril and Thoubal rivers from Manipur river system, India. Ecology, Environment and Conservation, 16(2): 197-207.
Sinha, M., De, D.K. and Jha, B.C. (1998). The Ganga environment and fisheries. Central Inland fisheries Institute, Barrackpore. pp.142
Teta, C., Ncube, M. and Naik, Y. S. (2017). Heavy metal contamination of water and fi sh in peri-urban dams around Bulawayo, Zimbabwe. African Journal of Aquatic Science, 42(4): 351-358.
Trivedy, R.K. and Goel, P.K. (1984). Chemical and biological methods for water pollution studies, Environmental publication Media, Karad, India. 1-251.
Venkatesharaju, K., Ravikumar, P., Somashekar, R.K. and Prakash, K.L. (2010). Physicochemical and bacteriological investigation on the river Cauvery of Kollegal stretch in Karnataka. Karnataka University Journal of Science, Engineering and Technology, 6(1): 50-59.
Waziri, M. and Ogugbuaja, V.O. (2010). Interrelationship between physicochemical water pollution indicators: A case study of River Yobe-Nigeria. American Journal of Science and Industrial Research,1: 76-80.
Yadav, S.S. and Kumar, R. (2011). Monitoring Water quality of Kosi River in Rampur District, Uttar Pradesh, India. Advances in Applied Science Research, 2(2): 197-201.
Yellavarthi, E. (2002). Hydrobiological studies of Red hills Reservoir, North Chennai, Tamilnadu. Journal of Aquatic Biology, 17(1): 13-16.
Zainudin, Z., Rahman, N.A., Abdullah, N. and Mazlan, N.F. (2010). Development of water quality model for Sungai tebrau using QUAL2K. Journal of Applied Sciences, 10: 2748-2750.


