Immunology and Parasitology Section , Biology Department, Faculty of Sciences and Arts- Alkamel Jeddah University, Saudi Arabia
Corresponding author email: Hodaelsayed71@yahoo.com
Article Publishing History
Received: 05/10/2020
Accepted After Revision: 12/12/2020
In developing countries, people remain infected with protozoan and helminths parasite contributing to death and economic loss. Toll-like receptors (TLRs) family is considered one of the most major pattern recognition receptors found to be concerned in innate immunity and expressed intracellularly as well as on cell surfaces. They play an essential function in the various protozoans and helminths’ ability to activate the host immune system response. These were engaged in producing cytokines and chemokines to promote phagocytosis and the killing of parasites. TLRs have negative regulation strategies to decrease the production of pro-inflammatory cytokines to reduces excessive pathology and reduce tissue damage caused by unregulated TLR activation. The main achievements findings made on the strategies of TLRs negative regulation mediated host response against protozoan and helminthic parasitic infections have been collected using many databases and the research published papers in indexed medical journals. Clarification of the relationship of host-parasites with the TLR pathway would enhance the management method of pathogens immunotherapy. Research in these fields have informed the production of new generation of therapeutics so that science in this field will have a promising future. In this review, we discuss the relationships between most pathogens and the strategies of TLRs to manage infectious diseases in the host immune regulatory network. Furthermore, the promising role that TLRs play in all parasite infections and therapeutic control strategies will also be stressed. Further studies are required to check for interplay strategies that control negative regulation that will provide new guidelines for the therapy of several crosstalk pathogens.
Toll Like Receptors, Parasitic Infections, Protozoan and Helminthic
Elsayed H. M. Toll Like Receptors (Tlrs) Strategies for the Control of Protozoan and Helminthes Parasitic Infections. an Updated Review. Biosc.Biotech.Res.Comm. 2020;13(4).
Elsayed H. M. Toll Like Receptors (Tlrs) Strategies for the Control of Protozoan and Helminthes Parasitic Infections. an Updated Review. Biosc.Biotech.Res.Comm. 2020;13(4). Available from: https://bit.ly/3oHRKW5
Copyright © Elsayed This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
The main concern in today’s world is of the rising number of cases of protozoan and helminth parasite infections with a high mortality rate (Ropert et al. 2008). These parasites have the property of immunomodulation, as they have their own strategies to escape from immune responses to survive inside their host for long periods (Maizels et al. 2004). Toll-like receptors (TLRs) are characteristic cellular receptors that can recognize pathogens and their associated molecules through innate infection responses (Kaye and Aebischer 2011). Thirteen TLRs have been currently identified, of which TLR1–TLR9 are retained within human and mice. Nonfunctional TLR10 found in mice whereas TLR11-TLR13 are detected within mice but are lost from human (Takeda and Akira 2015).In the last decade, the potential role of Toll-like receptors in fighting parasitic infections has attracted much interest. TLRs serve as the front line of defense response in the host against infecting pathogens by initiating an intracellular signaling cascade to activate an early inflammatory and innate immune response (Sudhagar et al. 2020). This review will cover the main conclusion regarding the mediated reaction between TLRs related protozoan and helminths infections.
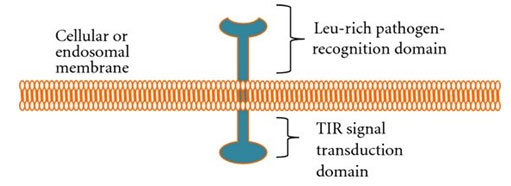
Structure of TLR: All TLRs have two domains: 20–27 extracellular leucine-rich repeats domain that detects pathogen and a Toll-interleukin 1 (TIR-1) domain that interacts with an adapter molecule to set off an immune response (fig. 1). The main differences in TLRs are ligand specificities, signal transduction and subcellular location (El-Zayat et al. 2019).
Figure 1: Schematic diagram of TLR (Muccioli et al. 2012).
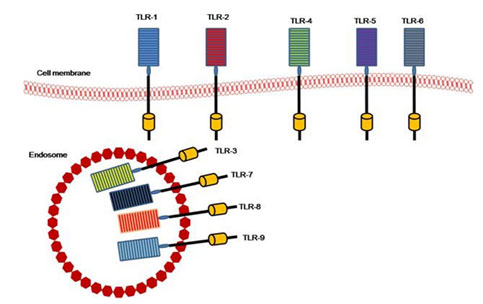
TLRs members: There are 13 mammalian TLR members, 12 in mice, and 10 in humans. The TLR10 mouse gene is not functioning (Ann et al 2016). They are distinguished by repeats of the leucine-rich extracellular domain and an intracellular domain with a retained zone receptor called the Toll/ I L-1 (TIR) domain and the transmembrane domain (Ulevitch 2004). Repeats with Leucine-rich are present in cytoplasmic and transmembrane proteins and participating in the identification, banding, and signal transduction of ligand (Kobe and Deisenhofer 1995). TLRs are functionally subdivided into cell membrane TLRs and Intracellular TLRs or nucleic acids sensors (Fig. 2). All 13 types of TLRs elicit the myeloid differentiation primary response protein 88- (MyD88-) dependent pathways except TLR3 (Ji-Yoon et al. 2020).
Figure 2: TLRS members and their location (El-Zayat et al. 2019).
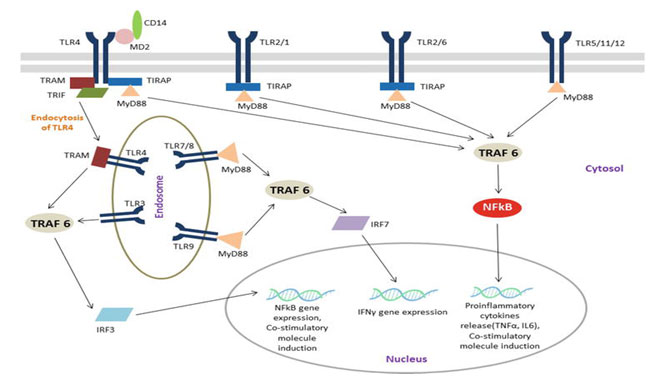
TLRs role of monitoring parasite infection: Specific pathogen-associated molecular patterns (PAMPs) are identified and interacting with pattern recognition receptors (PRRs) expressed in innate and non-immune cells in foreign organisms. PRRs display the critical function, including immune and non-immune cells, of the innate immunological reaction. They also identify released molecules from damaged host cells called damage-associated molecular patterns (DAMPs). TLR and NOD (nucleotide oligomerization domain-like receptor) recognize intracellular PAMPS and activate signals that induce inflammatory cascades of cytokines to be produced (Mariathasan and Monack 2007 & Takeuchi and Akira 2010).PAMPs interactions with PRRs activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-B). Proinflammatory cytokines such as interleukins (e.g. IL6) and tumor necrosis factor α (TNFα) are released into circulation after infection because of NF-B migrate to the nucleus (Fig. 3).
Such cytokines can regulate the inflammatory reaction through altering vascular endothelial permeability, infiltrate blood cells into inflamed tissues, and develop acute-phase protein production. Certain PRRs monitoring PAMP to recruit IRF3 in addition to IRF7 transcription factors resulting in the development of IFN-β, which play a central role in antiviral response. The innate immune reflex also invokes and induce adaptive immune system activation, which gives the host immune memory towards the invading pathogen (Iwasaki and Medzhitov 2015 & Ji-Yoon et al. 2020).
Figure 3: TLR signaling pathway (Chandrani 2019).
TLRs are mainly expressed in immune-functioning tissues, including spleen and peripheral blood leucocytes as mast cells, macrophages, and dendritic cells. TLRs found on the plasma membrane excluding TLR3, 7, 8, and 9. Such TLRs are found on the endosome and lysosome membranes with no detectable expression on the surface of the cell (Takeda and Akira 2005). The TLRs, especially surface receptor TLR2 and TLR4, have gained immense importance because they are extremely capable of identifying different molecular patterns from invading pathogens. Such (PRRs) function not only as innate sensors but also as innate and adaptive immune responses to form and bridge. They also play an important role in regulating the balance between Th1 and Th2 type responses (Mukherjee 2016).
To control adaptive immune responses, the ancient Toll – IL-1 receptor signaling pathway is important. TLR signals through MyD88 pathway leading to activation of MAPK and induces NF-kB to the nucleus which promotes the induction of proinflammatory cytokines (Franco 2017). Parasites can both activate and negatively regulate TLRs, this mechanism is used by the parasite to suppress the host immune system by interfering with TLRs ‘ expression and function (Venugopal et al 2009). Moreover, TLR-mediated signal transduction mechanisms and their consequences explain the operation of other immune signaling pathways. TLR signaling deficiency leads to various pathological chronic diseases and manipulation of TLR signaling promises to halt their activity (Yibo et al. 2020).
Table 1. Highlights TLR genes distribution across the genome, the number of amino acids, molecular weight, and their adaptor molecule. Table 1: Toll-like receptors (O’Connell et al 2017).
| TLRs | chromosome | Protein a.a | M.W
(kda) |
Ligand(s) | Sources | Adaptor molecule (Vanhoutte et al 2007) |
| TLR1 | 4p14 | 786 | 84 | Triacylated lipopeptides | Gram-positive and negative bacteria | MAL/MyD88 |
| TLR2 | 4q31-32 | 784 | 84 | Bacterial lipoproteins | Gram-positive and negative bacteria | MAL/MyD88 |
| Heat shock protein 70 | Mycoplasma and Host/self | |||||
| TLR3 | 4q35 | 904 | 97 | dsRNA | Viruses and Host/self | TRIF |
| TLR4 | 9q32-33 | 839 | 90 | Lipopolysaccharide | Gram-negative and bacteria | MAL/MyD88 and TRAM/TRIF |
| Heat shock proteins | Host/self | |||||
| TLR5 | 1q33-42 | 858 | 91 | Flagellin | Flagellated bacteria | MyD88 |
| TLR6 | 4p14 | 796 | 91 | Diacylated lipopeptide | Mycoplasma | MAL/MyD88 |
| TLR7 | Xp22 | 1049 | 121 | ssRNA | Virus | MyD88 |
| TLR8 | Xp22 | 1041 | 120 | ssRNA | Virus | MyD88 |
| TLR9 | 3p21 | 116 | Unmethylated CpG DNA | Bacteria and Self | MyD88 | |
| TLR10 | 4p14 | 1032 | 95 | Unknown | Unknown | Unknown |
TLRs distribution in different immune cells: TLRs are receptors expressed by different types of immune cells, a major component of innate immunity. TLR 1,2,4,5,6,7,8 is shown in neutrophils and monocytes/macrophages, and the dendritic myeloid cell expresses TLR 2.3,4,7,8. As well as, B lymphocytes generate TLR 1,3,6,7,9,10 while T lymphocytes (Th1/Th2) express TLR 2, 3, 5, 9. Three types of TLR can express by T lymphocytes (regulatory ) are TLR2,5,8. Expression of TLR2,4,5,7,8,9 usually on mononuclear peripheral blood cells (Chandrani et al 2019).
TLRs and parasite infection: TLRs have potential as therapeutic targets, either alone or in combination with standard immunotherapy and pharmacotherapy. In recent years, TLR antagonists or agonists from their negative regulators have been investigated as vaccine adjuvants to improve tumor, allergy, and infectious disease immune response. TLRs regulate downstream pathways involved in adaptive immune responses by influencing multiple antigen-presenting cell (APC) functions (Kanzler et al 2007). TLRs may be used to differentiate parasites from the self by macrophages and dendritic cells. Furthermore, TLR families are known to be combine with the pathogen and activating downstream signal transduction pathways. Moreover, TLRs have been demonstrated to be important for protozoan parasite resistance and identification of their components (Ghartey et al. 2020)
Infection by L. major in TLR4 mice leads to low parasite replication that correlates with higher levels of inducible nitric oxide synthase. Hence, TLR4 competence could resolve cutaneous lesions and control parasite growth (Kropf et al 2004). TLR2 forms heterodimers with TLR1 or TLR6 and modulates downstream signaling pathways. LPG’s L. major is involved in modulating the expression and function of TLR9. Mice infected with L. major increases the expression of TLR1 and TLR2 but TLR6. The association TLR2-TLR2 increases but the association TLR2-TLR6 decreases. The difference between TLR2 and TLR1 or TLR2 and TLR6 in the development of heterodimers results in a functional duality of TLR2. Pegylated bisacycloxypropylcysteine, a diacylated lipopeptide ligand of TLR6, plays a host-protective role against experimental Leishmania major infection (Pandey et al 2014).
Mice deficient in either TLR2, 4, or 9 are more susceptible to L. major infection than TLR9-deficient animals. The TLR9 deficiency inhibits the response of curative Th1 (Abou-Fakher et al 2009). TLR9 seems to play an important role in infection by L. major or L. infantum (Sacramento et al 2015). On the other hand, TLR11 and TLR12 play an important role in a L. major infection by reducing the parasite burden, increasing the IFN-γ level, and decreasing IL-4 production (Shukla et al 2018). TLRs 2, 3, 4, and 9 play an important leishmanicidal role; however, TLRs can facilitate replication of Leishmania and make the recipient more susceptible to infection in the late stages of infection (Dalia 2015). Recent research has shown that TLR4-IRF1 inevitably results in production of IFN-β as a tool to damp the chronic inflammatory process (Sacramento et al. 2020).
Two TLR1 polymorphisms have been evaluated, rs4833095 (Asn248Ser) and rs5743618 (Ser602Ile) for their association with P. falciparum infection and placental malaria susceptibility (Hamann et al 2010). TLR4-Asp299Gly and TLR4-Thr399Ile variants have been found to give a higher risk of severe malaria. TLR4 polymorphisms, infectious diseases, and evolutionary pressure during the migration of modern humans (Ferwerda et al 2007). Only certain adaptive immune responses need to be triggered by TLRs (Barton and Medzhitov 2002).Potential associations between TLR2 and TLR4 mRNA expression and cytokine and nitric oxide (NO) production have been studied with L. chagasi. While in the early stage of infection, the mRNA expression of TLR2, TLR4, IL-17, TNF-α, and TGF-β increases, it decreases in the late stage, correlating with parasite load. At the peak of infection, the mRNA expression of IFN-π and IL-12 declines (Cezario et al 2011). Mice were infected with L. braziliensis showed low levels of cytokine IL-12p40, leading to greater infection by L. braziliensis and a limited expansion of CD4+ T cells that produce IFN-γ and IL-17 during infection. In contrast, TLR2 mice were more immune to infection due to increased IFN-α development. Consequently, TLR2 seems to have a regulatory role in modulating the immune response to L. braziliensi (Vieira et al 2013).
TLR6 S249P’s single-nucleotide polymorphism can be a risk factor for malaria development. (Fabiana et al 2008) TLR5 the single-nucleotide polymorphism S180 L protects against malaria, while the single-nucleotide polymorphism of R392 stop codon increases susceptibility to the same disease (Khor et al 2007).The susceptibility can be modulated by a TLR4 polymorphism rs4986790 toward a P. vivax infection (Rani et al 2018). TLR4 A299G, TLR6 S249, and TLR 9-1486C/T influence the levels of circulating cytokines IL-6, IFN-γ, IL-12, IL-10, and IL-4 during a P. vivax infection. (Costa et al 2018) TLR9 1237C/C correlates with acute parasitemia during a P. vivax infection (Costa et al 2017). A protein resident in the endoplasmic reticulum, UNC93B1, is essential to hosting resistance to T. cruzi and T. gondii (Melo et al 2010). TLR2 blocked Giardia infection and reduced the burden of parasites compared to the control that infected with the same protozoan (Li et al 2017).
When TLR receptors bind to the ligands on the surface of dendritic cells of the S. mansoni, A. lumbricoides, and T. trichiura parasites, expression of co-stimulatory molecules (CD40, CD80, and CD86) and pro-inflammatory mediator synthesis Increase, such as IL-12 and TNF-α, evokes the response of Th1 lymphocytes (McSorley and Maizels 2012). TLR2 is necessary for the priming and expansion of active Regulatory T cells during schistosomiasis (Barton and Medzhitov 2002).In the early stages of infection, mRNA expression of TLR1, TLR2, TLR3, TLR4, and TLR9 is regulated for intestinal parasites. During the adult stage of infection with T. spiralis, TLR1 and TLR4 activate the signaling pathway dependent on MyD88. The expression of TLR2/4 during infection could be closely associated with immune responses mediated by Treg cells and greater expression of cytokines such as IL-10 and TGF-β. (Kane et al 2004) During an S. Mansoni infection, TLR2 and TLR4 restrict immune response activation while that of TLRs 1, 3, 7, and 8 is decreased after deposition of the egg (Dalia 2015).
The deficiency of these TLRs, therefore, facilitates the removal of adult worms (Pasare and Medzhitov 2003). Table 2; contains a list of the major pathogens involving TLRs and cytokines targeting these parasites.
Table 2. Functional immunobiology of different TLRs against array of pathogens.
| Parasite | Targeted receptor | Effector Function |
| Protozoa | ||
| Leishmania major | -TLR2
-TLR2, 4 – TLR9 |
-IFN-γ, TNF-α, and NFκB (Hawn et al 2002)
– IL-12, TNF- α, IFN-γ and NO (Becker et al 2003 & Abu-Dayyeh et al 2010) – IFN-γ (Abou-Fakher et al 2009) |
| Leishmania braziliensis | -TLR4 | -TNF-α and IL-10 (Galdino et al 2016) |
| Leishmania infantum | -TLR9
-TLR4 |
-IFN-α/β and IL-12 (Schleicher et al 2007)
-IFN-β and IRF1 (Sacramento et al 2020) |
| Leishmania Mexicana | -TLR2, 4
-TLR9 -TLR2 |
-TNF-α, IL-1β, IL-12 p40, IL-12 p70, and IL-10 (Rojas et al 2014)
-TNF-α and IL-12 (Martínez et al 2008) -Block IFN mediated (NO) production (Faria et al 2014) |
| Leishmania donovani | -TLR2 | -TNF-α, IL-12, and IFN-γ, NO (Srivastava et al 2013) |
| Leishmania panamensis | -TLR1,2,3, 4 | -TNF-α (Gallego et al 2011) |
| Toxoplasma gondii | -TLR11
– TLR11 – TLR4 |
– IL-2 (Yarovinsky et al 2005)
– IL-12, IFN-γ (Yarovinsky 2014) – TNF-α Reduce IL-10 (Gowda 2007) |
| Trypanosoma cruzi | -TLR2, 6
-TLR2 – TLR4 |
-NF-κB and IL-8, TNF-α, NO (Bott et al 2018)
– (IL-12), (NO) and (TNF) (Aoki et al 2012) – IL-12, TNF-α NO (Coelho et al 2002) |
| Plasmodium falciparum | -TLR2, 4
-TLR9 |
-TNF-α NF-κB, and IFN-γ (Hisaeda et al 2008)
-FN-γ (Matthew et al 2007) |
| Entamoeba histolytica | -TLR9
-TLR2 ,4 -TLR2, 4 |
-TNF-α (Ivory et al 2008)
-NF‐κB ,(IL)‐10, IL‐12, (TNF)‐α, and IL‐8 (Maldonado et al 2005) |
| Giardia lamblia | -TLR2 | -IL-12 p40, TNF-α, and IL-6, IFN-γ (Li et al 2017) |
| Helminths | ||
| Ascaris lumbricoides | -TLR2, 4 | -IL-10 and TGF-β (Kane et al 2004)
– IL-10 (Magdalena et al 2019) |
| Brugia malayi | -TLR2,4, 9 | -TNF-α, IL-6, and IL-10 (Faria et al 2014) |
| Toxocara canis | -TLR2 | -IL-4, IL-5, and IL-13 (Ludwig et al 2012) |
| Acanthocheilonema viteae | -TLR4 | -Block of IL-12 and TNF-a Production (Diaz and Allen 2007) |
| Taenia crassiceps | -TLR2, 4 | -IL-6 (Correale and Farez 2009) |
| Schistosoma mansoni | -TLR2
-TLR2, 4 |
-IL-1β, IL-6, IL-12, and TNF-α (Du et al 2014)
-IL-10, TGF-β (Kane et al 2004) |
| Fasciola hepatica | -TLR3 | -IL-10 and IL-4 (Carranza et al 2012) |
| Trichuris trichiura | -TLR4 | -TNF-α (Donnelly et al 2010) |
| Trichinella spiralis | -TLR4, 9 | -IL-10 and TGF-β (Sin et al 2015) |
| Trichomonas vaginalis | -TLR4 | -NF-κB (Im et al 2016) |
| Naegleria fowleri | -TLR4 | -IL-8, TNF-α, IL-1β (Martínez et al 2018) |
TLRs Negative regulation: TLRs down-regulation is a strategy that protozoa use to avoid immune responses. It has been shown that protozoan parasites such as Trypanosome spp and Entamoeba histolytica inhibit the immune response by reducing the expression of TLR2. Similarly, B. malayi significantly reduced the mRNA spreading of TLR3,4,5 and 7 from DCs that derived from monocyte (Maldonado et al 2000). The expression of TLR1,2,4 and 9 in B cells has decreased in filarial infected individuals. (Babu et al 2005). During TLR signaling, T cells play an important function as T cells release many of the TLRs. Upon induction by B cells and monocytes, T cells express lower levels of TLR1, 3, and 4 during lymphatically infected patients (Babu et al 2006).L. donovani has developed strategies for survival that suppress the pro-inflammatory reaction created by TLRs by suppression of TLR2, 4, and activation of IL-12p40 and IL-10 production (Chandra and Naik 2008). G. lamblia helps to reduce the development of TLR3 signaling pro-inflammatory cytokine. S. mansoni and F. hepatica have a restrained effect on the ripeness of DCs induced by TLR ligands to generate IL-12 less than those that are only activated with TLR4 ligand (Rodríguez et al 2015).
mansonistimulates IL-12 development or increases CD80, CD86 in addition to MHC class II surface expression in DCs. It decreases the development of IL-12, IL-6, and TNF-α, as well as the release of CD80/86 molecules, which can repress the Th1 response and enhances the Th2 response evolved by LPS (79). TLR2 and TLR3 are needed to shape the immune response during murine schistosomiasis and to increase the Th1/Th2 immune response balance (Vanhoutte et al 2007).Low T. cruzivirulence strain induces relatively high TLR4 expression and elevated pro-inflammatory cytokine rates such as IL-12 and TNF-α. On the other hand, it maintains low TLR4 expression and decreases TNF-α development (da Costa et al 2014). In addition, the expression of TLR2 and TLR4 in the brain of the mice was increased after Acanthamoeba sp infection in contrast to animal control (Wojtkowiak et al 2016). Acanthamoeba sp, on the other hand, generates high mRNA expression and high levels of TLR2 in animal brains at 2, 4, 8, 16, and 30 days after infection compared to uninfected mice (Wojtkowiak et al 2018).
Negative TLR regulation may reduce pro-inflammatory cytokine production to control the balance of Th1/2 cells and to modulate the expression of TLR4 that can protect the host from autoimmune development (Terrazas et al 2013). Most helminth pathogens operate the downregulating TLRs to improve the immune system and interfere with multiple gene expression linked to the transduction pathway. Several studies show that continued response to helminth antigens leads to control the response of cells to PAMPs from these parasites and suggesting a poor immune response in helminth infected individuals (Kane et al 2004).Moreover, latest results suggest that TLR4 obstructs the IRF1 and IFN-β immune response to avoid immunopathological disease (Sacramento et al. 2020)
Current treatment design considerations and Conclusion
Recent studies have shown that TLRs are likely to succeed in combating various parasites by activating signals pathways and their inhibition strategies offer great promise as an entirely new class of biologics for the treatment of inflammatory diseases (Shepherd et al 2015 and Chandrani et al 2019). Moreover, TLR ligands essential for vaccine adjuvant activity which should anticipate adjuvants that may improve the effectiveness of vaccines or have the effects of antiparasitic. The strong interest of biological researchers in TLRs will surely lead to desperately needed parasite therapies.
TLRs have many types of actions that can play a major role in the pathogen immune reaction. Every TLR plays a crucial role in this response. The inborn immune sensors encoded by germline which identify preserved parasite structures and induce signaling cascades that increase the development of inflammatory mediators by macrophages, neutrophils, dendritic cells, and other types of cells. TLRs are also able to trigger inflammatory cytokine production, which in many diseases plays a pathogenic role. Such processes activate mechanisms of innate host defense immediately, as well as trigger and organize the adaptive immune response.
Studies on infectious diseases identified by TLRs helped to understand the fundamental mechanisms of innate immune responses to most parasites. Parasites interfere with the immune response in order to promote their host survival. TLRs polymorphisms play an important role in rising host parasitemia and ligands of TLRs come from PAMPS and host DAMP.Ligand-mediated toll-like receptor activation (TLRs) not only causes inflammation but also suppress immunity, which is a new area of research. Uncontrolled activation of TLR can lead to severe inflammatory conditions and tissue damage. Through developing strategies for stabilizing negative regulation molecules it is possible to reduce the incidence of infectious diseases. Additional signaling channels of the innate immune system affect the negative regulation of TLR signaling. Consequently, TLRs negative regulation results in a lower output of pro-inflammatory cytokine and limits excessive pathology. Additional studies are necessary to describe the control in this system through these interactions. This upcoming research will offer new guidelines for the therapeutic treatment of several pathogenic infections.
REFERENCES
Abou-Fakher FH, Rachinel N, Klimczak M, Louis J, Doyen N. (2009): TLR9-dependent activation of dendritic cells by DNA from Leishmania major favors Th1 cell development and the resolution of lesions. Journal of Immunology.;182(3):1386-1396. Available from: DOI: 10.4049/jimmunol.182.3.1386
Abu-Dayyeh I, Hassani K, Westra ER, Mottram JC, Olivier M. (2010): Comparative study of the ability of Leishmania mexicana promastigotes and amastigotes to alter macrophage signaling and functions. Infect Immun. ;78:2438–45.DOI: 10.1128/IAI.00812-09
Ann Vu , Andrew Calzadilla, Sanaz Gidfar, Rafael Calderon-Candelario, Mehdi Mirsaeidi (2016): Toll-like receptors in mycobacterial infection. European Journal of Pharmacology. Volume 808, 5 August 2017, Pages 1-7. DOI: 10.1016/j.ejphar.2016.10.018
Aoki MP, Carrera-Silva EA, Cuervo H, Fresno M, Girones N, Gea S. (2012): Nonimmune cells contribute to crosstalk between immune cells and inflammatory mediators in the innate response to Trypanosoma cruzi infection. J Parasitol Res.2012;2012:737324. doi: 10.1155/2012/737324
Babu S., Blauvelt C. P., Kumaraswami V., Nutman T. B. (2005): Diminished expression and function of TLR in lymphatic filariasis: a novel mechanism of immune dysregulation. The Journal of Immunology.;175(2):1170–1176. doi: 10.4049/jimmunol.175.2.1170.
Babu S., Blauvelt C. P., Kumaraswami V., Nutman T. B. (2006): Cutting edge: diminished T cell TLR expression and function modulates the immune response in human filarial infection. The Journal of Immunology.;176(7):3885–3889. doi: 10.4049/jimmunol.176.7.3885.
Barton GM, Medzhitov R (2002): Toll-like receptors and their ligands. Curr Top Microbiol Immunol. 2002; 270():81-92. DOI: 10.1007/978-3-642-59430-4_5
Becker I, Salaiza N, Aguirre M, Delgado J, Carrillo-Carrasco N, Kobeh LG, et al. (2003): Leishmania lipophosphoglycan (LPG) activates NK cells through toll-like receptor-2. Molecular and Biochemical Parasitology.;130(2):65-74. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12946842
Bott E, Carneiro AB, Gimenez G, Lopez MG, Lammel EM, Atella GC, et al. (2018): Lipids from Trypanosoma cruzi Amastigotes of RA and K98 strains generate a pro-inflammatory response via TLR2/6. Frontiers in Cellular and Infection Microbiology.;8:151. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29868507
Carranza F, Falcón CR, Nuñez N, Knubel C, Correa SG, Bianco I, et al. (2012): Helminth antigens enable CpG-activated dendritic cells to inhibit the symptoms of collagen-induced arthritis through Foxp3+ regulatory T cells. PLoS One.;7(7):e40356. Available from: https://dx.plos.org/10.1371/journal.pone.0040356
Cezario GA, de Oliveira LR, Peresi E, Nicolete VC, Polettini J, de Lima CR, et al. (2011): Analysis of the expression of toll-like receptors 2 and 4 and cytokine production during experimental Leishmania chagasi infection. Memórias do Instituto Oswaldo Cruz.;106(5):573-583. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21894379
Chandra D, Naik S. (2008): Leishmania donovani infection down-regulates TLR2-stimulated IL-12p40 and activates IL-10 in cells of macrophage/monocytic lineage by modulating MAPK pathways through a contact-dependent mechanism. Clinical and Experimental Immunology.;154(2):224-234. Available from: http://doi.wiley.com/10.1111/j.1365-2249.2008.03741.x)
Chandrani Fouzder, Alpana Mukhuty, Snehasis Das and Dipanjan Chattopadhyay (2019): TLR Signaling on Protozoan and Helminthic Parasite Infection [Online First], IntechOpen, DOI: 10.5772/intechopen.84711. Available from: https://www.intechopen.com/online-first/tlr-signaling-on-protozoan-and-helminthic-parasite-infection
Coelho PS, Klein A, Talvani A, et al. (2002): Glycosylphosphatidylinositol anchored mucin-like glycoproteins isolated from Trypanosoma cruzi trypomastigotes induce in vivo leukocyte recruitment dependent on MCP-1 production by IFN-gamma-primed-macrophages. J Leukoc Biol. ;71:83744.
Correale J, and Farez M. (2009): Helminth antigens modulate immune responses in cells from multiple sclerosis patients through TLR2-dependent mechanisms. Journal of Immunology.;183(9):5999-6012. Available from: DOI: 10.4049/jimmunol.0900897
Costa AG, Ramasawmy R, Ibiapina HNS, Sampaio VS, Xabregas LA, Brasil LW, et al. (2017): Association of TLR variants with susceptibility to Plasmodium vivax malaria and parasitemia in the Amazon region of Brazil. PLoS One.;12(8):e0183840. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28850598
Costa AG, Ramasawmy R, Val FFA, Ibiapina HNS, Oliveira AC, Tarragô AM, et al. (2018): Polymorphisms in TLRs influence circulating cytokines production in Plasmodium vivax malaria. Cytokine.;110:374-380. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29656958
da Costa TA, Silva MV, Mendes MT, et al.(2014): Immunomodulation by Trypanosoma cruzi: toward understanding the association of dendritic cells with infecting TcI and TcII populations. Journal of Immunology Research. 2014 ;2014:962047. DOI: 10.1155/2014/962047
Dalia S Ashour (2015): Toll-like receptor signaling in parasitic infections, Expert Review of Clinical Immunology, 11:6, 771-780, DOI: 10.1586/1744666X.2015.1037286
Diaz A, and Allen JE. (2007): Mapping immune response profiles the emerging scenario from helminth immunology. Eur J Immunol. ;37:331926. DOI: 10.1002/eji.200737765
Donnelly S, O’Neill SM, Stack CM, Robinson MW, Turnbull L, Whitchurch C, et al. (2010): Helminth cysteine proteases inhibit TRIF-dependent activation of macrophages via degradation of TLR3. The Journal of Biological Chemistry.;285(5):3383-3392. Available from: http://www.jbc.org/lookup/doi/10.1074/jbc.M109.060368
Du L, Liu L, Yu Y, Shan H, Li L.( 2014): Trichinella spiralis excretory-secretory products protect against polymicrobial sepsis by suppressing MyD88 via mannose receptor. BioMed Research International.;2014:898646. Available from: https://www.hindawi.com/journals/bmri/2014/898646/
El-Zayat, S.R., Sibaii, H. & Mannaa, F.A. Toll-like receptors activation, signaling, and targeting: an overview. Bull Natl Res Cent 43, 187 (2019). https://doi.org/10.1186/s42269-019-0227-2
Fabiana M. S. Leoratti, Lilian Farias, Fabiana P. Alves, Martha C. Suarez-Mútis, José R. Coura, Jorge Kalil, Erney P. Camargo, Sandra L Moraes, Rajendranath Ramasawmy,. (2008): Variants in the Toll-Like Receptor Signaling Pathway and Clinical Outcomes of Malaria, The Journal of Infectious Diseases, Volume 198, Issue 5, 1 September 2008, Pages 772–780, https://doi.org/10.1086/590440
Faria MS, Calegari-Silva TC, de Carvalho Vivarini A, Mottram JC, Lopes UG, Lima AP. (2014): Role of protein kinase R in the killing of Leishmania major by macrophages in response to neutrophil elastase and TLR4 via TNFalpha and IFNbeta. FASEB J.;28:3050–63. doi: 10.1096/fj.13-245126
Ferwerda B, McCall MB, Alonso S, Giamarellos-Bourboulis EJ, Mouktaroudi M, Izagirre N, et al. (2007): TLR4 polymorphisms, infectious diseases, and evolutionary pressure during migration of modern humans. Proc Natl Acad Sci U S A.;104:16645–50. DOI: 10.1073/pnas.0704828104
Franco L. H., Fleuri A. K., Pellison N. C., et al. (2017): Autophagy downstream of endosomal Toll-like receptor signaling in macrophages is a key mechanism for resistance to Leishmania major infection. The Journal of Biological Chemistry. 292(32):13087–13096. DOI: 10.1074/jbc.M117.780981
Galdino H Jr, Saar Gomes R, Dos Santos JC, Pessoni LL, Maldaner AE, Marques SM, et al. (2016): Leishmania (Viannia) braziliensis amastigotes induces the expression of TNFalpha and IL-10 by human peripheral blood mononuclear cells in vitro in a TLR4-dependent manner. Cytokine.;88:184-192. Available from: DOI: 10.1016/j.cyto.2016.09.009
Gallego C, Golenbock D, Gomez MA, Saravia NG. (2011): Toll-like receptors participate in macrophage activation and intracellular control of Leishmania (Viannia) panamensis. Infection and Immunity.;79(7):2871-2879. Available from: http://iai.asm.org/lookup/doi/10.1128/IAI.01388-10
Ghartey-Kwansah, G., Adu-Nti, F., Aboagye, B. et al. (2020): Autophagy in the control and pathogenesis of parasitic infections. Cell Biosci 10, 101. https://doi.org/10.1186/s13578-020-00464-6
Gowda DC. (2007): TLR-mediated cell signaling by malaria GPIs. Trends Parasitol. ;23:596–604. DOI: 10.1016/j.pt.2007.09.003
Hamann L, Bedu-Addo G, Eggelte TA, Schumann RR, Mockenhaupt FP. (2010): The toll-like receptor 1 variant S248N influences placental malaria. Infection, Genetics and Evolution.;10(6):785-789. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20478407
Hawn TR, Ozinsky A, Underhill DM, Buckner FS, Akira S, Aderem A. (2002): Leishmania major activates IL-1 alpha expression in macrophages through a MyD88-dependent pathway. Microbes and Infection.;4(8):763-771. Available from: DOI: 10.1016/s1286-4579(02)01596-4
Hisaeda H, Tetsutani K, Imai T, Moriya C, Tu L, Hamano S, et al. (2008): Malaria parasites require TLR9 signaling for immune evasion by activating regulatory T cells. Journal of Immunology.;180(4):2496-2503. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18250459
Im SJ, Han IH, Kim JH, Gu NY, Seo MY, Chung YH, et al. (2016): Inflammatory response of a prostate stromal cell line induced by Trichomonas vaginalis. Parasite Immunology.;38(4):218-227. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26832322
Ivory CP, Prystajecky M, Jobin C, Chadee K. (2008): Toll-like receptor 9-dependent macrophage activation by Entamoeba histolytica DNA. Infection and Immunity.;76(1):289-297. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17984204
Iwasaki A and Medzhitov R (2015): Control of adaptive immunity by the innate immune system. Nature Immunology 16: 343–353. DOI: 10.1038/ni.3123
Ji-Yoon Noh, Suk Ran Yoon, Tae-Don Kim, Inpyo Choi, Haiyoung Jung, Toll-Like Receptors in Natural Killer Cells and Their Application for Immunotherapy, Journal of Immunology Research, vol. 2020, Article ID 2045860, 9 pages, 2020. https://doi.org/10.1155/2020/2045860
Kane CM, Cervi L, Sun J, McKee AS, Masek KS, Shapira S, et al. (2004): Helminth antigens modulate TLR-initiated dendritic cell activation. Journal of Immunology.;173(12):7454-7461. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15585871
Kanzler H, Barrat FJ, Hessel EM, Coffman RL. (2007): Therapeutic targeting of innate immunity with toll-like receptor agonists and antagonists. Nature Medicine.;13(5):552-559. Available from: DOI: 10.1038/nm1589
Kaye P. M. and Aebischer T.(2011): Visceral leishmaniasis: immunology and prospects for a vaccine, Clinical Microbiology and Infection, vol. 17, no. 10, pp. 1462–1470, 2011. doi.org/10.1111/j.1469-0691.2011.03610.x.
Khor CC, Chapman SJ, Vannberg FO, Dunne A, Murphy C, Ling EY, et al. (2007): A Mal functional variant is associated with protection against invasive pneumococcal disease, bacteremia, malaria and tuberculosis. Nature Genetics.;39(4):523-528. Available from: http://www.nature.com/articles/ng1976
Kobe, B. and Deisenhofer, J.(1995): Proteins with leucine-rich repeats. Curr. Opin. Struct. Biol. 5, 409–416. https://doi.org/10.1016/0959-440X(95)80105-7
Kropf P, Freudenberg N, Kalis C, Modolell M, Herath S, Galanos C, et al. (2004): Infection of C57BL/10ScCr and C57BL/10ScNCr mice with Leishmania major reveals a role for toll-like receptor 4 in the control of parasite replicationJournal of Leukocyte Biology.;76(1):48-57. Available from: http://doi.wiley.com/10.1189/jlb.1003484)
Li X, Zhang X, Gong P, Xia F, Li L, Yang Z, et al. (2017): TLR2(−/−) mice display decreased severity of giardiasis via enhanced proinflammatory cytokines production dependent on AKT signal pathway. Frontiers in Immunology.;8:1186. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28979269
Ludwig-Portugall I, Layland LE. (2012): TLRs, Treg, and B cells, an interplay of regulation during helminth infection. Frontiers in Immunology.;3:8. Available from: http://journal.frontiersin.org/article/10.3389/fimmu.2012.00008/abstract
Magdalena M. Aguirre-García, Araceli Rojas-Bernabé, A. Pamela Gómez-García and Alma R. Escalona-Montaño (2019): TLR-Mediated Host Immune Response to Parasitic Infectious Diseases [Online First], IntechOpen, DOI: 10.5772/intechopen.84679.
Maizels, R. M., A. Balic, N. Gomez-Escobar, M. Nair, M. D. Taylor, and J. E. Allen. (2004): Helminth parasites—masters of regulation. Immunol. Rev. 201: 89–116. doi.org/10.1111/j.0105-2896.2004.00191.x
Maldonado C., Trejo W., Ramirez A., et al.(2000): Lipophosphopeptidoglycan of Entamoeba histolytica induces an antiinflammatory innate immune response and downregulation of toll-like receptor 2 (TLR-2) gene expression in human monocytes. Archives of Medical Research.;31(4):71–73. DOI: 10.1016/s0188-4409(00)00199-5
Maldonado‐Bernal C. , Kirschning C. J., Rosenstein Y. et al. (2005): The innate immune response to Entamoeba histolytica lipopeptidophosphoglycan is mediated by toll‐like receptors 2 and 4. https://doi.org/10.1111/j.1365-3024.2005.00754.x
Mariathasan S, and Monack DM. (2007): Inflammasome adaptors and sensors: intracellular regulators of infection and inflammation. Nat Rev Immunol. 2007 Jan; 7(1):31-40. DOI: 10.1038/nri1997
Martínez-Castillo M, Santos-Argumedo L, Galván-Moroyoqui JM, Serrano-Luna J, Shibayama M. ( 2018): Toll-like receptors participate in Naegleria fowleri recognition. Parasitology Research.;117(1):75-87. Available from: . https://doi.org/10.1007/s00436-017-5666-9
Martínez-Salazar B, Berzunza-Cruz M, Becker I. (2008): Leishmania mexicana DNA activates murine macrophages and increases their TLR9 expression. Gaceta Médica de México.;144(2):99-104. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18590029
Matthew B. B. McCall, Mihai G. Netea, Cornelus C. Hermsen, Trees Jansen, Liesbeth Jacobs, Douglas Golenbock, André J. A. M. van der Ven, Robert W. Sauerwein. (2007): Plasmodium falciparum Infection Causes Proinflammatory Priming of Human TLR Responses. The Journal of Immunology July 1, 2007, 179 (1) 162-171; DOI: 10.4049/jimmunol.179.1.162
McSorley HJ, and Maizels RM. (2012) Helminth infections and host immune regulation. Clinical Microbiology Reviews.;25(4):585-608. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23034321)
Melo MB, Kasperkovitz P, Cerny A, Konen-Waisman S, Kurt-Jones EA, Lien E, et al. (2010): UNC93B1 mediates host resistance to infection with Toxoplasma gondii. PLoS Pathogens.;6(8):e1001071. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20865117)
Muccioli M, Sprague L, Nandigam H, Pate M, Benencia F. (2012): Toll-like receptors as novel therapeutic targets for ovarian cancer. ISRN Oncol.;2012:642141. Epub 2012 Mar 4. PMID: 22530148; PMCID: PMC3317035. doi: 10.5402/2012/642141.
Mukherjee S, Karmakar S, Babu SP. (2016): TLR2 and TLR4 mediated host immune responses in major infectious diseases: a review. Braz J Infect Dis.2016 Mar-Apr;20(2):193-204.DOI: 10.1016/j.bjid.2015.10.011
O’Connell, Sarah and McKernan, Declan P (2017): Toll-like Receptors. In: eLS. John Wiley & Sons, Ltd: Chichester. https://doi.org/10.1002/9780470015902.a0004028.pub3
Pandey SP, Chandel HS, Srivastava S, Selvaraj S, Jha MK, Shukla D, Ebensen T, Guzman CA , Saha B (2014): Pegylated Bisacycloxypropylcysteine, a Diacylated Lipopeptide Ligand of TLR6, Plays a Host-Protective Role against Experimental Leishmania major Infection. The Journal of Immunology October 1, 2014, 193 (7) 3632-3643; DOI: 10.4049/jimmunol.1400672
Pasare C and Medzhitov R. (2003): Toll pathway-dependent blockade of CD4+CD25+ T cell-mediated suppression by dendritic cells. Science.;299(5609):1033-1036. Available from: http://www.sciencemag.org/cgi/doi/10.1126/science.1078231)
Rani A, Nawaz SK, Arshad M, Irfan S. (2018): Role of rs4986790 polymorphism of TLR4 gene in susceptibility towards malaria infection in the Pakistani population. Iranian Journal of Public Health.;47(5):735-741. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29922617
Rodríguez E, Noya V, Cervi L, Chiribao ML, Brossard N, Chiale C, et al. (2015): Glycans from Fasciola hepatica modulate the host immune response and TLR-induced maturation of dendritic cells. PLoS Neglected Tropical Diseases.;9(12):e0004234. Available from: https://dx.plos.org/10.1371/journal.pntd.0004234)
Rojas-Bernabe A, Garcia-Hernandez O, Maldonado-Bernal C, Delegado-Dominguez J, Ortega E, Gutierrez-Kobeh L, et al. (2014): Leishmania Mexicana lipophosphoglycan activates ERK and p38 MAP kinase and induces production of proinflammatory cytokines in human macrophages through TLR2 and TLR4. Parasitology.;141(6):788-800. Available from: DOI: 10.1017/S0031182013002187
Ropert C, Franklin BS, Gazzinelli RT (2008): Role of TLRs/MyD88 in host resistance and pathogenesis during protozoan infection: lessons from malaria. Semin Immunopathol. 2008 Feb;30(1):41-51. doi: 10.1007/s00281-007-0103-2
Sacramento L, Trevelin SC, Nascimento MS, Lima-Junior DS, Costa DL, Almeida RP, et al. (2015): Toll-like receptor 9 signaling in dendritic cells regulates neutrophil recruitment to inflammatory foci following Leishmania infantum infection. Infection and Immunity.;83(12):4604-4616. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26371124)
Sacramento, L. A., Benevides, L., Maruyama, S. R., Tavares, L., Fukutani, K. F., Francozo, M., Sparwasser, T., Cunha, F. Q., Almeida, R. P., da Silva, J. S., & Carregaro, V. (2020). TLR4 abrogates the Th1 immune response through IRF1 and IFN-β to prevent immunopathology during L. infantum infection. PLoS pathogens, 16(3), e1008435. https://doi.org/10.1371/journal.ppat.1008435
Schleicher U, Liese J, Knippertz I, Kurzmann C, Hesse A, Heit A, et al. (2007): NK cell activation in visceral leishmaniasis requires TLR9, myeloid DCs, and IL-12, but is independent of plasmacytoid DCs. The Journal of Experimental Medicine.;204(4):893-906. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17389237
Shepherd C, Navarro S, Wangchuk P, Wilson D, Daly NL, Loukas A. (2015): Identifying the immunomodulatory components of helminths. Parasite Immunology.;37(6):293-303. Available from: http://doi.wiley.com/10.1111/pim.12192
Shukla D, Chandel HS, Srivastava S, Chauhan P, Pandey SP, Patidar A, et al. (2018): TLR11 or TLR12 silencing reduces Leishmania major infection. Cytokine.;104:110-113. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29017773)
Sin Kim, Mi Kyung Park, and Hak Sun Yu. (2015): Toll-Like Receptor Gene Expression during Trichinella spiralis Infection 2015 Aug 25. doi: 10.3347/kjp.2015.53.4.431
Srivastava S, Pandey SP, Jha MK, Chandel HS, Saha B. (2013): Leishmania expressed lipophosphoglycan interacts with Toll-like receptor (TLR)-2 to decrease TLR-9 expression and reduce anti-leishmanial responses. Clinical and Experimental Immunology.;172(3):403-409. Available from: DOI: 10.1111/cei.12074
Sudhagar, A., El-Matbouli, M., & Kumar, G. (2020): Identification and Expression Profiling of Toll-Like Receptors of Brown Trout (Salmo trutta) during Proliferative Kidney Disease. International journal of molecular sciences, 21(11), 3755. https://doi.org/10.3390/ijms21113755
Takeda K. and Akira S. (2015): Toll-like receptors, in Current Protocols Immunology, vol. 109, 14 edition, 2015. doi.org/10.1002/0471142735.im1412s109
Takeda K. and Akira, S. (2005): Toll-like receptors in innate immunity. Int. Immunol. 17, 1–14.). https://doi.org/10.1093/intimm/dxh186
Takeuchi O and Akira S (2010): Pattern recognition receptors and inflammation. Cell 140: 805–820. https://doi.org/10.1016/j.cell.2010.01.022
Terrazas CA, Alcántara-Hernández M, Bonifaz L, Terrazas LI, Satoskar AR. (2013): Helminth-excreted/secreted products are recognized by multiple receptors on DCs to block the TLR response and bias Th2 polarization in a cRAF dependent pathway. The FASEB Journal.;27(11):4547-4560. Available from: http://www.fasebj.org/doi/10.1096/fj.13-228932)
Ulevitch R. (2004): Therapeutics targeting the innate immune system. Nat Rev Immunol 4, 512–520 doi:10.1038/nri1396
Vanhoutte F, Breuilh L, Fontaine J, Zouain CS, Mallevaey T, Vasseur V, Capron M, Goriely S, Faveeuw C, Ryffel B, Trottein F. (2007): Toll-like receptor (TLR)2 and TLR3 sensing is required for dendritic cell activation, but dispensable to control Schistosoma mansoni infection and pathology. Microbes Infect. 2007 Nov-Dec;9(14-15):1606-13. Epub 2007 Sep 26. DOI:
10.1016/j.micinf.2007.09.013Venugopal P. G., Nutman T. B., Semnani R. T.(2009): Activation and regulation of Toll-Like Receptors (TLRs) by helminth parasites. Immunologic Research.;43(1–3):252–263. doi: 10.1007/s12026-008-8079-0.
Vieira ÉLM, Keesen TSL, Machado PR, Guimarães LH, Carvalho EM, Dutra WO, et al. (2013): Immunoregulatory profile of monocytes from cutaneous leishmaniasis patients and association with lesion size. Parasite Immunology.;35(2):65-72. Available from: http://doi.wiley.com/10.1111/pim.12012)
Wojtkowiak-Giera A, Derda M, Kolasa-Wołosiuk A, Hadaś E, Kosik-Bogacka D, Solarczyk P, et al. (2016): Toll-like receptors in the brain of mice following infection with Acanthamoeba spp. Parasitology Research.;115(11):4335-4344. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27511368
Wojtkowiak-Giera A, Derda M, Kosik-Bogacka D, Kolasa-Wołosiuk A, Solarczyk P, Cholewiński M, et al. (2018): Influence of Artemisia annua L. on toll-like receptor expression in brain of mice infected with Acanthamoeba sp. Experimental Parasitology.;185:17-22. Available from: https://linkinghub.elsevier.com/retrieve/pii/S001448941730365X
Wong-Baeza I, Alcantara-Hernandez M, Mancilla-Herrera I, et al. (2010): The role of lipopeptidophosphoglycan in the immune response to Entamoeba histolytica . J Biomed Biotechnol.;2010:254521. doi: 10.1155/2010/254521
Yarovinsky F, Zhang D, Andersen JF, Bannenberg GL, Serhan CN, Hayden MS, et al. (2005): TLR11 activation of dendritic cells by a protozoan profilin-like protein. Science.;308(5728):1626-1629. Available from: http://www.sciencemag.org/cgi/doi/10.1126/science.1109893
Yarovinsky, F. (2014): Innate immunity to Toxoplasma gondii infection. Nat Rev Immunol 14, 109–121. doi:10.1038/nri3598
Yibo Wang, Shuting Zhang, Hongyuan Li, Hongshuang Wang, et al. (2020): Small-Molecule Modulators of Toll-like Receptors. Accounts of Chemical Research, 53 (5), 1046-1055 DOI: 10.1021/acs.accounts.9b00631