Centre for Drug Discovery and Development, Sathyabama Institute of Science and
Technology (Deemed to be University), Chennai, Tamil Nadu – 600 119. India
Corresponding author email: mrkactinos@gmail.com
Article Publishing History
Received: 15/07/2020
Accepted After Revision: 10/09/2020
Public health threat due to drug resistant pathogens like Mycobacterium tuberculosis and others necessitates the discovery of novel antibiotics. Actinobacteria from understudied sources is emerging as promising source for novel molecules effective against drug resistant pathogens. The present study was undertaken for bioprospecting of endophytic actinobacteria from the mangrove plant Rhizophora apiculata. Different actinobacterial cultures isolated from Rhizophora apiculata leaves were screened for antimicrobial activity by agar plug method against a wide range of bacterial and fungal pathogens. Antimicrobial metabolites from potential strain SACC4 was produced by agar surface fermentation and extracted using different organic solvents and tested against bacterial pathogens. Antitubercular activity of strain SACC4 was also tested by adopting luciferase reporter phage (LRP) assay. Strain SACC4 was identified based on their phenotypic and molecular characteristics. About 28 distinct actinobacterial strains were isolated from the mangrove leaves in which 50% of the strains showed antimicrobial activity. Notably strain SACC 4 showed broad spectrum activity against bacterial pathogens.
Based on the studied phenotypic and molecular characteristics, strain SACC4 was identified as Streptomyces species. Among the different solvents tested, methanol extract showed a maximum antimicrobial activity against S. aureus in disc diffusion method at 100µg/ml concentration. The methanol extract showed more than 60 percentage inhibition against the standard strain M. tuberculosis H37Rv, drug sensitive and MDR M. tuberculosis strains at both 100µg/ml and 500µg/ml concentrations. The results of the present investigation revealed that the endophytic Streptomyces from mangrove plants are the potent source of novel bioactive metabolites effective against M. tuberculosis and other drug resistant pathogens.
Actinobacteria, Streptomyces, antimicrobial, anti TB, taxonomy
Ayswarya S, Manigundan K, Vijayalashmi G, Vignesh A, Gopikrishnan V, Anbarasu S, Joseph J, Radhakrishnan M. Antimicrobial and Antitubercular Activity of Endophytic Actinobacterium, Streptomyces Sp. SACC 4 Isolated from the Mangrove Plant Rhizophora apiculata. Biosc.Biotech.Res.Comm. 2020;13(3).
Ayswarya S, Manigundan K, Vijayalashmi G, Vignesh A, Gopikrishnan V, Anbarasu S, Joseph J, Radhakrishnan M. Antimicrobial and Antitubercular Activity of Endophytic Actinobacterium, Streptomyces Sp. SACC 4 Isolated from the Mangrove Plant Rhizophora apiculata. Biosc.Biotech.Res.Comm. 2020;13(3). Available from: https://bit.ly/2ZDHbZv
Copyright © Ayswarya et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Antimicrobial resistance (AMR) is a global health crisis. Multi drug resistant pathogens like Mycobacterium tuberculosis pose serious threat to public health. Hence, the critical component of AMR solution is the development of truly novel antibiotic. Natural products and their derivatives are mainstays of our antibiotic drugs (Wright, 2017) in which members of the bacterial phylum actinobacteria in particular the genus Streptomyces are the known produces of numerous novel secondary metabolites especially antibiotics. However, in recent years, searching of routine sources like terrestrial soil results in the isolation of known actinobacteria which in turn produce known bioactive metabolites (Berdy, 2012).
Instead, exploring rare sources like plants may results in the isolation of novel actinobacterial strains with promising bioactive potential. Actinobacteria reside within the healthy plants as endophytes are the promising source for bioactive metabolites to fight against drug resistant pathogens (Golinska et al., 2015, Simpkin et al., 2017). Limited reports on endophytic actinobacetria from plants revealed their enormous bioprospecting potential for clinical, environmental and agricultural applications, (Golinska et al., 2015; Singh et al., 2018).
The present study reports the isolation of endophytic actinobacteria from the mangrove plant Rhizophora apiculata and their antimicrobial and antitubercular properties.
MATERIAL AND METHODS
Leaves sample of the mangrove plant Rhizophora apiculata was collected from Parangipettai mangrove zone (11.50°N, 79.75°E), Tamil Nadu, India. Endophytic actinobacteria from the leaves were isolated by adopting the method described by Golinska et al., (2015). Morphologically distinct actinobacterial strains were recovered and preserved on ISP2 agar slants at 4°C. All the actinobacterial cultures were screened for antimicrobial activity by agar plug method against clinical strains of Staphylococcus aureus MTCC96, clinical strains of Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, Candida albicans and Aspergillus fumigates (Radhakrishnan et al., 2014). Actinobacterial strain SACC 4 which showed promising activity was selected as potential strain.
Phenotypic and molecular characteristics of strain SACC4 was studied by the methods described by Shirling and Gottileb (1966) and Radhakrishnan et al., (2013). The 16S rRNA gene of SACC4 was amplified by using the primer pairs: 27F 5´AGAGTTTGATCMTGGCTCAG3´ (forward) and 1492R 5´TACGGYTACCTTGTTACGACTT3´ (reverse) and sequencing was carried out at Eurofins Genomics India Pvt. Ltd., Bangalore. The identification of phylogenetic neighbors and calculation of pair wise 16SrRNA gene sequence similarities were achieved using the MEGA version 6 (Tamura et al., 2013) and BLAST analysis (http://blast.ncbi.nlm. nihgov/Blast.cgi). The obtained 16SrRNA sequence was submitted to GenBank to get the accession number.
Effect of solid-state and submerged fermentation on bioactive metabolite production by the strain SACC4 was investigated against S. aureus by agar well diffusion method. Effect of solvents on the extraction of bioactive metabolites from the cell free supernatant of strain SACC4 was studied using different solvents such as n-hexane, chloroform and ethyl acetate at 1:2 ratio for 24 hours. The antibacterial activity of different solvent extracts was tested against S. aureus MTCC96 by disc diffusion method (Bavya et al., 2011). The antimicrobial activity of methanol extract of strain SACC4 was evaluated against wide range of bacterial pathogens by disc diffusion method at 100 µg/disc concentration. Zone of inhibition was measured after 24 hours of incubation at 37°C and expressed in millimetre in diameter. Effect of different concentration of YEME medium (2X, 1X, 1/2X, 1/4X and 1/10X) on antimicrobial activity of strain SACC 4 was also tested (Yilmaz et al., 2008).
Antitubercular activity of potential strain of SACC4 was studied against standard laboratory strain Mycobacterium tuberculosis H37Rv, drug sensitive and multi drug resistant (MDR) clinical isolates of M. tuberculosis by adopting LRP assay. The relative light unit (RLU) was measured in a luminometer (Radhakrishnan et al., 2014).
Percentage RLU reduction = Control RLU – Test RLU / Control RLU X 100
Extract showing RLU reduction by 50% or more when compared to control were considered as having anti-tubercular activity.
RESULTS AND DISCUSSION
Totally 28 endophytic actinobacterial cultures were isolated from the Rhizophora apiculata leave. Based on their morphology, about 75 % of the cultures were tentatively identified as Streptomyces and the remaining 25% are rare actinobacterial strains. Actinobacteria from understudied ecosystems are the promising source for novel bioactive natural products. Limited reports on endophytic actinobacetria from plants revealed their enormous bioprospecting potential for clinical, environmental and agricultural applications (Golinska et al., 2015; Singh et al., 2018). In India, reports on endophytic actinobacteria are very few. In a previous study, Du et al. (2013) analyzed the reported 600 actinobacteria belonging to 34 genera and 7 unknown taxa from 37 medicinal plants in which maximum from root followed by stem and least from leaves. However in the present study 25% of the actinobacterial strains recovered from leaves were found to be rare actinobacterial strains.
In preliminary screening, 18 actinobacterial cultures (64%) showed inhibition against any one of the bacterial pathogens tested. Notably, strain SACC 4 showed promising inhibition against S. aureus with 15-18 mm zone of inhibition against S. aureus, E. coli, K. pneumoniae, P. aeruginosa and the fungi C. neoformans.
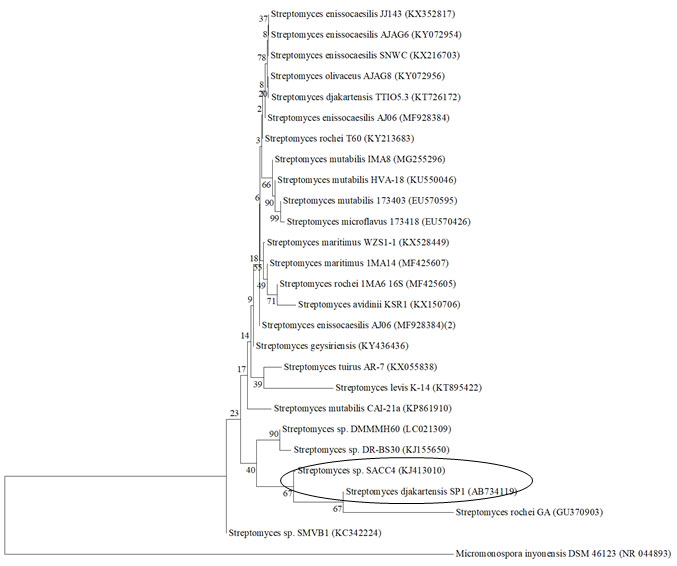
Microscopic, cultural and physiological characteristics of strain SACC4 was given in table 1. PCR amplification and BLAST analysis revealed that the SACC 4 sequence showed 99.9% similarity with the genus Streptomyces. Phylogenetic tree constructed based on neighbor-joining method also indicated its closest similarity with Streptomyces djakartensis (99%) and other species (Figure 1). The 16s rRNA gene sequence of strain SACC 4 was published in GenBank with the accession number KJ413010. Strain SACC4 was showed 99.8% close similarity with the 16SrRNA sequence of Streptomyces djakartensis.
However, certain differences were noted when comparing the physiological characteristics of strain SACC 4 with the characteristics of S. djakartensis (Kounouz et al., 2017). Hence it was assumed that the strain SACC 4 is found to be novel at strain level. In another previous study, it was reported that 85 % of the total 123 endophytic actinobacterial isolates studied were determined to be unique at the strain level (Janso et al., 2010). Further DNA-DNA hybridization and G+C content analysis is needed to confirm its novelty.
Table 1. Characteristics of endophytic actinobacterial strain SACC 4
| Characteristics | SACC4 | ||
| Micromorphology | Aerial mycelium | + | |
| Substrate mycelium | + | ||
| Fragmentation | – | ||
| Cultural characteristics | Colony consistency | Powdery | |
| Aerial mass colour | Gray | ||
| Reverse side pigment | – | ||
| Soluble pigment | – | ||
| Physiological Characteristics | ISP1 (Tryptone Yeast Extract Agar) | – | |
| ISP2 (Yeast extract malt extract agar) | + | ||
| ISP3 (Oat meal agar) | – | ||
| ISP4 (Inorganic salt starch agar) | + | ||
| ISP5 (Glycerol asparagine agar) | – | ||
| ISP6 (Peptone yeast Iron agar) | + | ||
| ISP7 (Tyrosine agar) | + | ||
|
Arabinose | + | |
| Xylose | + | ||
| Inositol | + | ||
| Mannitol | + | ||
| Fructose | + | ||
| Rhamnose | + | ||
| Enzyme production | Asparagine | + | |
| Glutamine | + | ||
| PH tolerance | 5 | – | |
| 6 | – | ||
| 7 | + | ||
| 8 | + | ||
| 9 | + | ||
| 10 | + | ||
| 11 | + | ||
| Temperature tolerance (° C ) | 20 | + | |
| 30 | + | ||
| 40 | + | ||
| NaCl tolerance ( % ) | 0 | + | |
| 1 | + | ||
| 2 | + | ||
| 3 | + | ||
| 5 | – | ||
“+” grown, “ – “ no growth
Figure 1: The phylogenetic relationship of the potential actinobacterial strain SACC4 based on 16S rRNA gene homology. The tree was constructed using the neighbor-joining method with pairwise-deletion model analyses, which were implemented in the Molecular Evolutionary Genetics Analysis (MEGA), version 6.0 program. The resultant tree topologies were evaluated by bootstrap analysis based on 1000 replicates. Micromonosporainyonensis was used as out group. Scale bar indicates the number of substitutions per site.
In fermentation study, even until 20 days of fermentation, strain SACC 4 was failed to produce bioactive compounds in submerged fermentation whereas it produced bioactive compounds in agar surface fermentation. The agar plug from ISP2 agar showed activity against S. aureus even from 2nd day (11.2±0.72 mm in dm) of fermentation. The zone of inhibition eventually increased from 2ndday to 8th day (17.1±1.09 mm in dm) of fermentation. This is due to the antibiotics production through solid state fermentation are higher quantities and more stable than liquid state fermentation (Subramanian et al., 2012). This finding is in agreement with the antibiotic production by desert soil, Streptomyces strain D25 which showed antibacterial activity and also produced antitubercular pigment only in solid culture (Radhakrishnan et al., 2015).
In solid state fermentation, strain SACC4 showed good growth and the methanol extract obtained from the fermented medium showed 18mm inhibition against S. aureus but failed to show activity in liquid state fermentation. Isolation of crude extract by solvent extraction is an important phenomenon to find a good solvent that has the potential to extract high yield and most potent bioactive compounds. Our research findings demonstrated that the extract of methanol have significant antimicrobial and antitubercular activity. These results were clearly comparable to that, who evaluated Streptomyces mutabilis which produced an antitubercular activity and antimicrobial activity against gram positive and gram negative pathogens (Mahmoud et al., 2015). Among the different concentrations of YEME agar, 1X concentration of YEME agar was found to support good growth and antimicrobial compound production which was expressed by its activity against S. aureus (10.2±0.12), B. subtilis (15.0±0.64) and E. coli (9.8±0.44). The methanol extract of strain SACC 4 was found to be active against S. aureus and wide range of Gram negative bacterial pathogens (Table 2).
Table 2. Antimicrobial activity of methanol extract of endophytic actinobacterial strain SACC 4.
| Bacterial pathogens | Zone of inhibition (mm in diameter)* |
| Staphylococcus aureus | 18.2 ±0.15 |
| Klebsiella pneumonia | 17.0 |
| Pseudomonas aeruginosa | 18.0 |
| Vibrio cholerae 01 ogawa | 8.1±0.17 |
| Vibrio cholerae 0139 | 10.2±0.15 |
| Vibrio parahaemolyticus 03:K6 | 12.8±0.47 |
| Aeromonassorbia | 8.3±0.17 |
| Aeromonashydrophilla | 10.2±0.15 |
| Enteropathogenic E.coli 0115 | 10.2±0.37 |
| Enteroaggregative E.coli 015 | 10.9±0.35 |
| Enteropathogenic E.coli 0114 | 10.9±0.21 |
| Salmonella typhimurium | 12.0±0.32 |
| S. worthington | 12.9±0.38 |
| S. infantis | 11.4±0.31 |
| Shigella sonnei | 0 |
| S. dysenteriae sero 5 | 0 |
| S.. boydii sero 1 | 0 |
| S. flexneri type 2a | 0 |
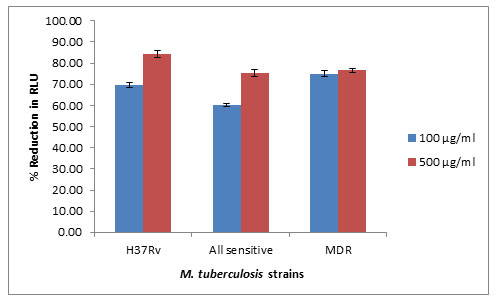
In LRP assay, the methanol extract of strain SACC 4 was showed more than 60% inhibition against standard laboratory strain Mycobacterium tuberculosis H37Rv, drug sensitive and MDR M. tuberculosis strains (Figure 2).
Figure 2: Antitubercular activity of potential actinobacterial strainSACC4
Findings of the present study evidenced that mangroves are the promising source for the isolation of bioactive endophytic actinobacteria.
ACKNOWLEDGEMENTS
We thank the management of Sathyabama Institute of Science and Technology (Deemed to be University), Chennai for the research facilities provided. Authors (Dr MRK and AS) thank the DST-SERB for their support in the form of research grant (Ref:YSS/2015/001887) under Young Scientist Scheme.
CONFLICT of INTEREST: Authors have no conflict of interest
REFERENCES
Bavya M, Mohanapriya P, Pazhanimurugan R, Balagurunathan R.(2011) Potential bioactive compound from marine actinomycetes against biofouling bacteria. Indian J Geo-Marine Sci 40(4): 578-582.
Berdy J.(2012) Thoughts and facts about antibiotics: where we are now and where we are heading. J Antibiot 65(8): 385-395.
Du H, Su J, Yu L, Zhang Y.(2013) Isolation and physiological characteristics of endophytic actinobacteria from medicinal plants. Wei Sheng Wu Xue Bao 53(1):15-23.
Golinska P, Wypij M, Agarkar G, Rathod D, Dahm H, Rai M (2015) Endophytic actinobacteria of medicinal plants: diversity and bioactivity. Antonie van Leeuwenhoek 108: 267-289.
Janso JE, Carter GT.(2010) Biosynthetic potential of phylogenetically unique endophytic actinomycetes from tropical plants. Appl Environ Microbiol 76: 4377-4386.
Kounouz Rachedi, Ferial Zermane, Radja Tir, Fatima Zohra Ayache, Robert Duran, Béatrice Lauga, Solange Karama , Maryse Simon and Boulahrouf Abderrahmane (2017). Characterization of tribenuron-methyl resistant Actinobacteria strains. Asian Journal of Microbiology, Biotechnology and Environmental Sciences. 19. 81-87.
Mahmoud AY, Abdallah HM, El-Halawani MA, Jiman-Fatani AAM (2015) Anti-tuberculous activity of Treponemycin produced by a Streptomyces strain MS-6-6 isolated from Saudi Arabia. Molecules 20: 2576-2590.
Radhakrishnan M, Gopikrishnan V, Balaji S, Balagurunathan R, Vanaja K.(2014) Bioactive Potential of Actinomycetes from Less Explored Ecosystems against Mycobacterium tuberculosis and Other Non mycobacterial Pathogens. Int Scholarly Res Notice Article ID 812974.
Radhakrishnan M, Gopikrishnan V, Suresh A,Selvakumar N, Balagurunathan R, Vanaja Kumar(2013) Characterization and phylogenetic analysis of antituberculous compound producing actinomycete strain D25 isolated from Thar Desert soil, Rajasthan. Bioinformation 9: 018-022.
Radhakrishnan M, Venugopal G, Ramasamy B, Kumar V.(2015) Effect of critical medium components and culture conditions on anti-tubercular pigment production from novel Streptomyces sp D25 isolated from Thar dessert, Rajasthan. J App Pharm Sci 5(06): 015-019.
Shirling BE, Gottlieb D.( 1966) Methods for characterizing Streptomyces species. Int J Syst Bacteriol 16: 313-340.
Simpkin VL Matthew JR, Ruth K, Elias M.(2017) Incentivising innovation in antibiotic drug discovery and development: progress, challenges and next steps. J Antibiot 70: 1087-1096.
Singh R, Dubey AK.(2018) Diversity and applications of endophytic actinobacteria of plants in special and other ecological niches. Front Microbiol 9; 1767.
Subramaniyam R, Vimala R.(2012) Solid state and submerged fermentation for the production of bioactive substances: a comparative study. Int J Sci Nat 3: 480-486.
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30: 2725-2729.
Wright GD (2017) Opportunities for natural products in 21st century antibiotic discovery. Nat Prod Rep 34: 694-701.
Yilmaz EB, Yavuz M, Kizil M. (2008) Molecular characterization of rhizosphere soil Streptomycetes isolated from indigenous Turkish plants and their antimicrobial activity. World J Microbiol Biotechnol 24: 1461-1470