Department of Zoology, Bankura Sammilani College, Bankura, West Bengal, India.
Corresponding author email: muktipbag@gmail.com
Article Publishing History
Received: 10/04/2021
Accepted After Revision: 04/06/2021
Sericulture is a well-established rural agribusiness in India and produces about 40,000 metric tonnes per year of silkworm pupae (SWP) on dry weight basis. This SWP contains enormous amount of crude protein (34.40%) one of the major feed component, besides considerable amount of crude lipid (10.63%), carbohydrate (9.95%) etc. claiming as a promising ingredient of fish feed. Fingerlings of tilapia, Oreochromis niloticus (Linn.) with mean weight 5.0 ± 0.20g were randomly stocked at 15fingerlings per tank in three replicates (tank size, L x B x H; 1m x 1m x1m) for 90 days to evaluate the effect of sericulture byproduct on growth and flesh fatty acid profile of tilapia. Two isonitrogenous pelleted feeds viz. silk worm pupae meal (SWM) and market available feed (MAF) were prepared and fed to the fish twice daily.
The feed (SWM) containing fermented silk worm pupae as principal ingredient was the most efficient in terms of final average weight gain, specific growth rate, protein efficiency ratio and fatty acid profile (Poly unsaturated fatty acid and n3/n6 ratio). Fish PUFA, especially the n3 fatty acids and n3/n6 ratio are affected positively when fed with SWM which is good for the quality of the fish produced with respect to the benefits of human health. The results showed that fermented silk worm pupae can be used in the diet for tilapia which enhances production as well as quality of cultured fish. This research has been conducted to develop scope for future studies on researching sericulture byproduct as a feed. As currently not many researchers are working on this topic, this report will help by acting as a suitable milestone and an updated reference whenever needed.
Fish Pufa, N3/N6 Ratio, Sericulture Byproduct, Tilapia
Bag M. P. Use of Sericulture Byproduct as Feed for Tilapia - Oreochromis niloticus (Linn.). Biosc.Biotech.Res.Comm. 2021;14(2).
Bag M. P. Use of Sericulture Byproduct as Feed for Tilapia – Oreochromis niloticus (Linn.). Biosc.Biotech.Res.Comm. 2021;14(2). Available from: <a href=”https://bit.ly/3uS0ZEK“>https://bit.ly/3uS0ZEK</a>
Copyright © Bag This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Several studies have been observed to make supplementary feeding of fish cost effective have been directed to substitute the high-cost fish meal with less expensive protein sources. This aspect of feed development research is centered on the search for inexpensive, locally available and nutritious protein sources that can supply all the nutritional needs of the fish. Sericulture is a well-established rural agribusiness in India and produces about 40,000 metric tonnes per year of silkworm pupae (SWP) on dry weight basis (Rangacharyulu et al. 2003). A small quantity of this SWP is sun-dried and used in animal feeds, while a considerable amount is discarded in open places though it contains enormous amount of crude protein (34.40%) one of the major feed component, besides considerable amount of crude lipid (10.63%), carbohydrate (9.95%) etc. claiming as a promising ingredient of fish feed (Table 1) (Gabriel et al. 2007; Bag et al. 2013).
An appropriate fermentation ensiling process has been developed to prepare a pathogen free. Annison (1993) reported that microbial fermentation and nutrient synthesis was typically important in organisms with a diet high in fibre. Fermentation is a simple and cheap process where there may be an increase in the nutrient level through xxmicrobial synthesis, apart from microbial degradation. Many studies have been carried out to evaluate the effects of nonconventional ingredients used in diets as FM substitutes on fish fatty acid composition. Surprisingly, such an important farm waste product for making nutritionally balanced fish feed formulation is remained unexplored. Research information on utilization of such alternative fish feed source is scanty (Yashoda et al. 2001; Bag et al. 2013).
The beneficial fatty acid in fish body is synthesis from the feed materials they consumed (Horrobin et al. 1990). The beneficial effects of fish lipids on human health have already been well established. It is therefore of utmost importance to determine the influences of feed on growth as well as fat deposition in fish flesh (Cengiz et al. 2003; Mukhopadhyay, 2009). Keeping the above facts in view, the investigation was carried out with the main objective to study the growth performance of O. niloticus by using fermented silkworm pupae as alternative source of protein in fish feed and also to study the qualitative changes in fish flesh as human food (Bag et al. 2013).
MATERIAL AND METHODS
Twenty fingerlings in triplicate groups were used in two different treatments. Altogether one hundred and twenty (120) Nile tilapia (male and female ratio 1:1) fingerlings were utilised in this experiment. The fish fingerlings were treated with potassium permanganate solution (1 mg L–1) to remove any external parasites and were acclimatized in a big tank for five days. Experiments were carried out at the tanks of aquacultural engineering section of IIT-Kharagpur, Paschim Medinipur, West Bengal, India from June to August 2018. Each group of fingerlings also were initially weighed to record the initial biomass content. They were stocked in six rectangular cemented tanks (1000 L).
The water system was static in nature and the bottom of the tank was filled with local agricultural soil (pH 6.4 ± 0.05). The experiment was conducted for 90 days from June to July in the year 2019.Dechlorinated well water (temperature 26 ± 3 °C, pH 7.v0 ± 0.05, free CO2 0.4 ±0.01 mg L–1, available nitrogen 0.5 ±0.05 mg L–1 and dissolved oxygen (DO) 6± 0.5 mg L–1) was used in the experiment. The principal feed ingredient (silkworm pupae) was collected from local sericulture farm after silk reeling at very low cost. These waste substances were economically cheap but contained significant amount (35– 37%) of crude protein.
Biochemical composition of silkworm pupae used in the feed for tilapia is shown in Table 1. Diets used for growth trial were prepared in such a manner, the feed formulations remain almost isonitrogenous (30 g 100 g–1) and isoenergetic (4.0 Kcal g–1) in nature. The choice of these nutrient levels, particularly protein, was intended to reflect the practical diets used in our country like India. Diet formulations are presented in Table 2. Mustard oil cake, wheat flour, rice bran and egg shell dust were the ingredient of Market available feed (MAF). In addition to these ingredients the chief ingredient of SWM feed was fermented silkworm pupae.
These ingredients were used to compensate lipid, protein and ash deficiency in formulated feed. Wheat flour was selected as binder. Prepared feed was fortified with egg shell dust which is available free of cost for calcium supplement. This was added keeping in mind that the developing fish needs huge quantity of calcium for its bone development. The different ingredients were thoroughly mixed using a food mixer (A200 Hobart Ltd). The proportion of different feed ingredients was determined by using Pearson’s square method.
The mixture was given the shape of pellets using a Pellet Mill (Model CL2) with a 12 mm die. The resulting pellets were dried in a hot air oven for 48 h at 50 ºC, packed in polythene bags and kept in dry and cool place. The feed was given ad libitum in a feeding bag hung from an iron rod in four locations in each tank. Unconsumed feed was removed after 1hour from the beginning of feed administration and dried in a hot air oven at 55°C. Growth and nutrient utilization were determined in terms of feed intake (FI), specific growth rate (SGR), feed conversion ratio (FCR), protein efficiency ratio (PER), and hepatosomatic index (HSI) as follows : FI (g fish-1 day-1) = Total feed intake per fish/number of days SGR (% day-1) = 100 × (ln[final body weight]- ln[initial body weight])/no. of days FCR = feed intake/live weight gain PER = live weight gain/crude protein intake HSI (%) = 100 × (liver weight/total body weight) GSI (%) =100 × (weight of gonad /total body weight).
Feed and carcass samples were analyzed following standard procedures (AOAC, 2002), dry matter (DM) after drying in a hot air oven (Gallenkamp, UK) at 105°C for 24 h; crude protein (CP) by Kjeldahl method (N × 6.25) after acid hydrolysis, crude lipid (CL) after extraction with petroleum ether for 7-8 h by Soxhlet method (40–60°C boiling range), total ash by igniting at 550°C for 3 h in muffle furnace (Size 2, Gallenkamp, UK). Organic matter (OM) was calculated by subtracting total ash from DM (Giri et al. 2000). Crude fibre was determined using a moisture free defatted sample which was digested by a weak acid HCl (0.1N) followed by a weak base NaOH (0.1N) using the Fibertec System 2021(FOSS, Denmark).
Nitrogen-free extract was determined by subtracting the sum of Crude protein, crude lipid, crude fibre and ash from DM (Giri et al. 2000). Gross energy was determined using a Bomb Calorimeter Model-DFU 24 following the process as described below. The sample was combusted in a chamber pressurized with pure oxygen and resulting heat measured by increase in the temperature of the water around the bomb (Giri et al. 2000).
The total lipids were extracted from all the samples, (fish flesh-2, feed-2) following the method of using methanol-chloroform (2:1, v/v), methanol-chloroform-water (2:1:0.8, v/v/v), and then again with the first solvent system viz., methanol-chloroform (2:1, v/v). Sample was dust with the solvent methanol-chloroform (2:1, v/v), filtered through Whatman no. 1 filter paper and residue was extracted with the next solvent system, consisting of methanol-chloroform water (2:1:0.8, v/v/v). The process was repeated once again with methanol chloroform (2:1, v/v) (Bligh et al., 1959; Giri et al. 2000).
Finally, the three extracts were pooled, diluted with three volumes of water (100-200 ml, depending on the volume of pooled extracts) and layer was allowed to separate in a separatory funnel made by Pyrex glass Co. The chloroform layer at the bottom of the separatory funnel was withdrawn and dried over anhydrous sodium sulphate in glass stoppered conical flasks, by Pyrex. Total lipid of various (fish flesh-2, feed-2) samples was dissolved in anhydrous methanol containing concentrated Sulfuric acid (1.0%, v/v) and the mixture was refluxed for 2 hours. Methanol was evaporated to a small volume (1-3 ml) and cooled to 4°C, in a freezer.
Distilled water 10–15 ml was added to the cooled mixture (1-3 ml) in hard glass test tubes by Pyrex and the methyl esters of fatty acids were extracted 3 times with aliquots (5-10 ml) of diethyl ether, vortexed in a Vortex mixer. The ethereal extracts were taken out by Pasteur pipettes, pooled and dried over anhydrous sodium sulphate, (1-2 gm) in conical flasks (25-50 ml capacity) with glass stopper, filtered through Whatman no. 1 filter paper, vacuum dried, redissolved in n-hexane (1-2 ml volume) and kept in a freezer at 4 °C for future use. Fatty acid methyl esters were purified by TLC using a solvent system of n-hexanediethyl ether (90:10, v/v) (Mangold 1969; Christie1982).
A standard methyl ester was also run on the same plate in a separate lane, for identification of the methyl ester bands in the samples. The location of methyl ester bands was indicated by placing the TLC plate in an iodine vapour chamber by Pyrex glass co. The methyl ester bands corresponding to the standard were marked and then scrapped off the plate with a sharp rajor blade. GLC of fatty acid methyl esters were done on a Chemito 1000 instrument, equipped with Flame Ionization Detector (FID). Quantifications were done by computer using specific Clarity Lite software. GLC of FAME was done on a BPX- 70 mega bore capillary column of 30 mt length and 0.53 mm internal diameter obtained from SGE, Australia (Christie1982).
Oven temperature was programmed from 150 °C – 240 °C with a rate of 8 °C/min.Initial and final temperatures were kept isothermal for 1 minute and 20 minutes respectively. Injection port and detector temperatures were 250 °C and 300 °C respectively. Nitrogen gas was used as carrier gas and its flowing rate was 6.18ml/min. Data from each treatment were subjected to one-way Analysis of Variance (ANOVA). The data are presented as mean±SE of three replicate groups; statistical analysis was performed using the SPSS 11.0 for windows. Duncan multiple range test was used to compare the mean values between individual treatments (Duncan, 1955).
RESULTS AND DISCUSSION
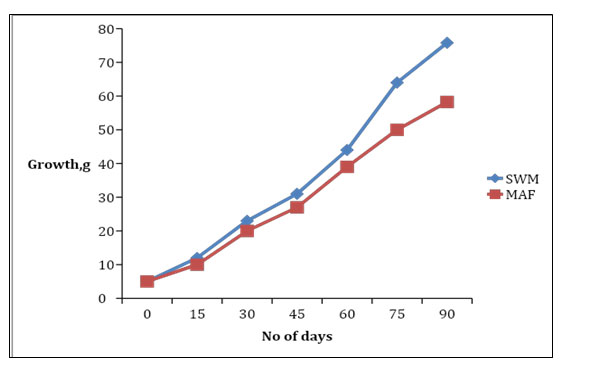
The highest weight gain (75.80g) was obtained in the SWM applied feed. The growth rate was always faster in SWM fed fish than fish fed with MAF (Figure 1).
Table 1. Biochemical composition of silkworm pupae used as feed for tilapia
| Ingredient (%) | Silkworm pupae |
| Dry matter | 90.72 |
| Crude protein | 34.54 |
| Crude lipid | 10.52 |
| Carbohydrate | 9.59 |
| Ash | 13.14 |
| Nitrogen free extrac | 13.93 |
| Crude fibre | 9.76 |
| Gross energy (Kcal g-1) | 3.71 |
This indicates that fish can assimilate the SWM feed well. This was possibly due to their higher palatability and preference of the fish to take it as their potential food. Amount of feed intake was highest (2.21g) in SWM fed fish.
Table 2. Detailed calculations of SWM and MAF diet
| S l. No. | Name of feed | Ingredients | % of CP in ingredient | % of ingredient in formulated feed | % of crude protein in feed | % of lipid in feed | % of carbohydrate in feed | Calorific value of feed
(kcal/g) |
|
1
|
SWM
|
Silk Warm Pupae | 22.25 | 40.0 | 25.46 | 8.1 | 10.4 | 4.0 |
| MOC | 34.65 | 30.0 | ||||||
| Wheat flour | 9.08 | 28.0 | ||||||
| Egg shell dust | 1.8 | 2.0 | ||||||
|
2 |
MAF
|
Fish meal | 40.54 | 36.0 | 30.4 | 8.2 | 10.5 | 4.0 |
| MOC | 33.52 | 34.0 | ||||||
| Wheat flour | 8.80 | 28.5 | ||||||
| Egg shell dust | 1.7 | 1.5 |
The feed conversion ratio (FCR) was differed significantly and lowest value (2.29) was recorded from SWM fed fish indicating an encouraging effect on economic involvement in fish farming.
Figure 1: Growth rate of O. niloticus fed with SWM and MAF
The specific growth rate (0.91) and protein efficiency ratio (1.66) were highest in SWM fed treatment. This indicates the better quality of protein in the feed produced from fermented silkworm pupae (SWM). The hepatosomatic index (HSI) and gonadosomatic index (GSI) were not differed significantly between two treatments (Table 3).
Table 3. Growth performance and nutrient utilization of O. niloticus fed with SWM and
| Particulars | SWM | MAF |
| Initial weight (g) | 5.10
±0.01a |
5.10
±0.02a |
| Final weight (g) | 75.80
±0.13a |
58.25
±0.24b |
| Initial length (cm) | 4.50
±0.03a |
4.50
±0.01a |
| Final length (cm) | 14.50
±0.12a |
11.50
±0.10b |
| Feed intake (g fish-1 day-1) | 2.15
±0.05a |
1.79
±0.02b |
| Specific growth rate (% day-1) | 0.90
±0.19a |
0.72
±0.05b |
| Feed conversion ratio | 2.28
±0.23a |
2.65
±0.05b |
| Protein efficiency ratio | 1.66
±0.12a |
1.26
±0.04b |
| Hepatosomatic index | 1.55
±0.06a |
1.17
±0.07b |
| Gonado somatic index | 1.62
±0.07a |
1.12
±0.06b |
In two feeds, SFA was less and MUFA was high in amount. But in other fed fish the situation was just opposite indicating that fish may be converted MUFA of feed into the SFA of its flesh. The amount of 22:1ω11 fatty acid was reduced absolutely in fed fish which is the main reason for such reduction.
Amount of DUFA was reduced nearly 3 times in fish than in feed (table 4). The value of linoleic acid (18:2ω6) of formulated feed was nearly 15% which has been found to be much lower (nearly half) in both the fed fishes. The mechanisms behind this conversion depend on the efficiency of the experimental fish by adopting desaturation and chain elongation process. This result indicates that fish may convert DUFA to SFA. The amount of PUFA and n3 fatty acids (EPA and DHA) was less in feed and more in fed fish. But n6 PUFA was reduced significantly in all fed fish in comparison to the feed offered. The n3/n6 ratio showed an increasing tendency in all fed fish and SWM fed fish exhibited significant enhancement of n3/n6 ratio (1.45).
Because amount of n6 fatty acid was lowest in SWM fed fish indicating transformation of n6 fatty acid to n3. The most interesting aspect of the present experiment is that the fishes provided with high level of n6 fatty acids in feeds offered however, the fishes were able to covert n6 fatty acids to n3 fatty acids efficiently to a very low n3/n6 ratio to comparative higher value of n3/n6. The mechanism of this conversion could not be explained at present. However, it proves that these fishes have the ability for such a conversion for the maintenance of physiological homeostasis (Ackman et al. 2002).
Ackman (2000) stated that only 14 fatty acids are really needed to describe the fatty acids of fish. However, Ackman (2000) Enlisted 64 fatty acids from 5 fresh water fishes of West Bengal, India. The fish under discussion recorded 28 fatty acids of the total lipid (TL) and the result is more or less similar to those reported from other tropical and certain temperate zone fresh water fishes. According to Ackman et al (2002) dominant fatty acids in lipids of all the fishes were myristic (14:0), palmitic (16:0), stearic (18:0), palmitoleic (16:1ω7), oleic (18:1ω9), linoleic (18:2ω6), linolenic(18:3ω3), arachidonic (20:4ω6), eicosapentaenoic (20:5ω3) and docosahexaenoic (22:6ω3) acids. The present results ascertain with the above findings. The total SFA of the experimental fish was nearly double than the amount reported by (Ackman et al. 2002).
Fatty acid deficiency in fish species is indicated by the presence of eicosatrienoic acid (20:3ω9) (Watanabe, 1982). Thus, the absence of eicosatrienoic acid in these fish indicates that these fish are not suffering from any fatty acid deficiency and the formulated feeds fulfill the requirement of fatty acids for tilapia. This observation corroborates that for hybrid striped bass in the USA (Nematipour, 1993). The n-3 PUFA is the chief group of components through which the beneficial effects of fish are mediated. The principal effects of n-3 PUFA are antithrombogenic and antiarrhythmic, whereas that of n-6 PUFA is antiatherogenic (Watanabe, 1982; Nematipour, 1993; Ackman et al. 2002).
Ackman et al. (2002) stated that n3/n6 ratio should range 1–2 for fresh water fish. The n3/n6 ratio of SWM fed fish was within the same range. The fish fed with SWM stores more n3 fatty acids than n-6 fatty acids which increase n3/n6 ratio (1.45) in return. Fish oil, lipids and their constituent fatty acids have high digestibility and energy value. Fish lipids contain high levels of n3 PUFA which may be essential for worm blooded animals like human. Fish is more beneficial than fish oil but for CAD patients prescribed amount of n3 PUFA (EPA and DHA) is required as the best insurance against sudden death24. Low fat and easy digestibility of the two batches of experimental fed fishes under investigation together with its EFA (n3 fatty acid) resource and other attributes discussed above can be recommended as a better diet on par with the recognized fish diet (Ackman et al. 2002).
Table 4. FA profiles of O. niloticusus fed with SWM and MAF feeds (% w/w of eachcomponent in total fatty acids)
| Components | MAF feed | SWM feed | MAF fed fish | SWM fed fish |
| Saturated | ||||
| 14:0 | 0.7 | 0.6 | 5.0 | 4.4 |
| 15:0 | 0.2 | 0.5 | 0.9 | 1.3 |
| 16:0 | 5.3 | 5.4 | 30.5 | 27.9 |
| 17:0 | 0.4 | 0.2 | 2.8 | 2.5 |
| 18:0 | 2.0 | 2.31.8 | 7.8 | 8.0 |
| 20:0 | 0.7 | 0.7 | 0.5 | 0.3 |
| 22:0 | 0.7 | 0.1 | 3.9 | 4.0 |
| 24:0 | 1.1 | 2.0 | 0.9 | 1.8 |
| ∑SFA | 11.1 | 11.3 | 52.3 | 50.2 |
| Monoene | ||||
| 14:1 | 0.0 | 0.0 | 1.0 | 0.3 |
| 15:1 | 0.0 | 0.0 | 0.3 | 0.2 |
| 16:1 | 1.2 | 1.4 | 7.5 | 7.6 |
| 17:1 | 0.0 | 0.0 | 0.4 | 0.7 |
| 18:1ω9 | 22.6 | 21.9 | 14.6 | 12.0 |
| 20:1ω9 | 7.0 | 8.1 | 1.6 | 2.1 |
| 22:1ω11 | 27.1 | 27.7 | 1.6 | 0.9 |
| 24:1 | 1.3 | 1.7 | 1.7 | 1.6 |
| ∑MUFA | 59.2 | 60.8 | 28.7 | 25.4 |
| Diene | ||||
| 16:2 | 0.0 | 0.0 | 0.2 | 0.5 |
| 18:2ω6 | 16.2 | 14.6 | 8.7 | 7.7 |
| 20:2 | 0.0 | 0.0 | 0.0 | 0.0 |
| ∑DUFA | 16.2 | 14.6 | 8.9 | 8.2 |
| Polyene | ||||
| 18:3ω6 | 0.5 | 0.4 | 0.3 | 0.4 |
| 18:3ω3 | 2.6 | 2.9 | 2.3 | 3.0 |
| 20:3ω6 | 1.1 | 1.1 | 1.4 | 1.0 |
| 20:3ω3 | 0.00 | 0.02 | 0.07 | 0.02 |
| 20:4ω6 | 0.9 | 1.0 | 1.1 | 1.0 |
| 20:5ω3 | 1.2 | 1.7 | 1.1 | 1.6 |
| 21:5ω3 | 0.5 | 0.5 | 0.5 | 0.4 |
| 22:5ω6 | 0.2 | 0.5 | 0.4 | 0.6 |
| 22:5ω3 | 2.0 | 2.6 | 2.1 | 2.8 |
| 22:6ω3 | 4.5 | 5.6 | 4.0 | 5.6 |
| ∑PUFA | 13.5 | 13.32 | 10.17 | 16.22 |
| Total – ω3 | 10.8 | 9.82 | 12.57 | 13.42 |
| Total – ω6 | 17.8 | 17.6 | 11.9 | 9.2 |
| n3/n6 | 0.60 | 0.75 | 1.05 | 1.45 |
CONCLUSION
The feed prepared from fermented silkworm pupae enhances growth and thereby yield of O. niloticus. It improves quality of fish by accumulating more n3 PUFA in the flesh of the fish as well as increasing the n3/n6 ratio which is beneficial for human health in relation to cardiovascular diseases. Moreover, the feed can be formulated by sericulture byproduct at local level leading to control pollution, lowering feed cost and employment generation in rural areas of India.
ACKNOWLEDGEMENTS
Due acknowledgement to the Principal, Bankura Sammilani College, Bankura for the help and support for conducting the research work.
Conflict of Interests:The author declares that no conflict of interests while writing this manuscript.
REFERENCES
Annison, G. (1993). The chemistry of dietary fibre. In: S. Samman and G. Annison (Eds.), Dietary Fibre and Beyond – Australian Perspectives. Nutritional Society of Australia, Occasional Publications, 1–18.
AOAC. (2000). Official methods of analysis, 17th edition. Association of Analytical Chemists, Gaithersburg, Maryland, USA.
Ackman, R.G.(2000). Fatty acids in fish and shell fish. In: Chow, C.K. (Ed.). Fatty Acids in Foods and their Healh Implication. M. Dekker, Inc, N.Y. and Basel, 153–172.
Ackman, R.G., Mcleod, C., Rakshit, S and Misra, K.K. (2002). Lipids and fattyacids of five freshwater food fishes of India. J. Food Lipids, 9:127–145.
Bag, M. P., Mahapatra S .C and Rao P.S. (2013). Sericulture byproduct as feed for tilapia, Oreochromis mossambicus (Peters). Current Biotica, 6(4): 489-500.
Bligh, E.G and Dyer, W.J. (1959). A rapid method of total lipid extraction and purification. Can. J. Biochem.Physiol; 37: 911–917.
Christie, W.W. (1982). In Lipid Analysis 2nd Edn. Pergamon Press, Oxford (England).
Christensen, J.H., Skou, H.A., Fog, L and Hansen, V. (2001). Marine n-3 fatty acids, wine intake, and heart rate variability in patients referred for coronary angiography. Lipids Circulation, 103: 65.
Cengiz, E.I. and Bashan, E. M. (2003). The effect of dietary fatty acids on the fatty acid composition in the phospholipids fraction of Gambusia affinis. Turkish J. Biol, 27:145–148.
Dewailly, E., Blanchet, C.S., Gingras, S and Sauve,L. (2001). Relations between n-3 fatty acid status and cardiovascular disease risk factors among Quebecers. Am. J. Clin. Nutr, 74: 603–611.
Duncan. D.B. (1955). Multiple range and multiple F-tests. Biometrics, 11: 1– 42.
Giri, S.S., Sahoo, S.K., Sahu, A.K and Mukhopadhyay, P.K. (2000). Nutrient digestibility and intestinal enzyme activity of Clarias batrachus (Linn.) juveniles fed on dried fish and chicken viscera incorporated diets. Bioresource Technology, 71: 97–101.
Gabriel, U.U., Akinrotimi O.A., Bekibele D.O., Onunkwo, D.N., and Anyanwu. P.E. (2007). Locally produced fish feed: potentials for aquaculture development in sub-Saharan Africa. African Journal of Agricultural Research, 2(7): 287– 295.
Horrobin. D., F, Manku. M.S. (1990). Clinical biochemistry of essential fatty acids. In D. F. Horrobin (Ed.), Omega-6 essential fatty acids. Pathophysiology and roles in clinical medicine, Wiley-Liss, 21–53.
Karthick,P., Aanad, R., Sampathkumar, S. J and Padmavathy, P. (2019). Silkworm pupae meal as alternative source of protein in fish feed. Journal of Entomology and Zoology studies, 7(4):78-85.
Mukhopadhyay,P.K. (2009). Fish as a nutritional source of long chain fatty acids. Science and Culture, 75: 53–60.
Mangold, H.K. (1969). In Thin Layer Chromatography (E. Stahl Ed.). Springer, New York, 155pp.
Misra, S., Ghosh, A and Dutta, J. (1984). Production and composition of microbial fat from Rhodotorulaglutinis. J. Sci. Food. Agric, 35: 59–65.
Nematipour, G.R. and Gatlin, D.M. (1993). Requirement of hybrid striped bass for dietary (n-3) highly unsaturated fatty acids. J. Nutr, 123:744–753.
Watanabe, T. (1982). Lipid Nutrition in Fish. Comparative Biochemistry and Physiology, 73B:3–15.
Yashoda, K., Sachindra, P., Rao, M.N and Mahendrakar. N.S. (2001). Changes in microbial population during fermentation of silk worm pupae. Sericologia, 41(2): 253–261.


