1Saratov State Agrarian University named after N.I. Vavilov, 410012, Teatralnaya pl.1, Saratov, Russia,
2Institute, Microbe, Saratov, Russia.
Corresponding author email: karpuninal@mail.ru
Article Publishing History
Received: 25/04/2021
Accepted After Revision: 24/06/2021
The primary aim of this survey is to investigate the role of Bacillilectin in the regulation of metabolic processes in the body of animals under stress. Evidently, under stress, the processes of lipid peroxidation (LPO) are activated in the body, which affects the structure and function of cell membranes. Of particular interest is the study of changes in the content of primary and secondary products in blood erythrocytes as the main indicators of the intensity of LPO processes and as markers of the degree of endogenous intoxication. To fulfil the aim of the study, the authors have utilized LII lectin isolated from the surface of nitrogen-fixing soil bacteria Paenibacillus polymyxa 1460. The studies were carried out on healthy male white outbred rats with an average body weight of 210 g.
The animals were kept under standard vivarium conditions: 12-hour illumination period, temperature 20 ºС, food, and water ad libitum. As a result, the study demonstrates that the lectin Paenibacillus polymyxa 1460 (LII) plays a significant role in the regulation of metabolic processes in the body of animals against the background of swimming stress, contributing to the normalization of lipid peroxidation processes, and also participates in the normalization of the microflora of the large intestine in conditions of dysbiosis in the body of animals against the background stress, acting as a prebiotic, which in turn is widely used for the correction and restoration of the number and qualitative composition of the intestinal microflora. Furthermore, considering the results, it can be inferred that P. polymyxa 1460 lectin LII exhibits antioxidant properties and can be successfully used as a natural antioxidant, being an effective tonic, as well as bio stimulating agent.
Animals’ Body, Bacilli Lectin, Lectinology, Metabolic Processes, Paenibacillus polymyxa.
L. V. Karpunina, M. V. Proskuryakova, S. V. Ivashenko, T. N. Rodionova, O. M. Popova. The Role of Bacillilectin in the Regulation of Metabolic Processes in the Body of Animals Under Stress. Biosc.Biotech.Res.Comm. 2021;14(2).
L. V. Karpunina, M. V. Proskuryakova, S. V. Ivashenko, T. N. Rodionova, O. M. Popova. The Role of Bacillilectin in the Regulation of Metabolic Processes in the Body of Animals Under Stress. Biosc.Biotech.Res.Comm. 2021;14(2). Available from: <a href=”https://bit.ly/33iKV3Q“>https://bit.ly/33iKV3Q</a>
Copyright © Karpunina et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Lectins are carbohydrate-binding proteins of a non-immunoglobulin nature, capable of specific recognition of carbohydrates and reversible binding to them. In recent years, there has been a transition in lectinology from the study of lectins of plant and animal origin to lectins of microorganisms. Low-toxic lectins of non-pathogenic bacteria and their effect on the metabolism of an animal organism are of particular interest, (Pees et al 2021).
Therefore, studies of the effect of lectins of bacterial origin on the metabolic processes of the body of animals, both in normal conditions and in some disorders, are an interesting and urgent task and find application in biology and medicine. Lectins, being biologically active substances, have long attracted the attention of various researchers. The role of bacterial lectins in animals is not well understood to date.
The ability of bacilli lectin to regulate the activity of certain enzymes, as well as the correction of important indicators of various metabolic processes in pathological conditions of the body against the background of various types of stress, opens up prospects for their possible use (Vorobiev et al., 1997; Gorelnikova, 2006; Sheikhshoaie et al., 2018; Pees et al., 2021).
As compounds of a protein nature, bacterial lectins have antimicrobial, cytokine activity, are able to change the adhesive function of phagocytic cells, which causes scientific and practical interest, which is constantly growing with the understanding of the role of lectins in the regulation of physiological processes and the prospect of their use as biologically active substances and effective pharmacological preparations (Wang et al., 2020; Dasgupta et al., 2020).
The normal intestinal microflora is an important link in the system of the body defense against stress and the preservation of its internal environment (Shenderov, 1987). Normally, all representatives of microflora form a biocenosis, in which each of them has a positive effect on others, ensuring the growth of microorganisms, their metabolism and resistance to damaging factors. Disturbances in the ratio between these biotic components in the intestine reflect microecological changes, which in turn contribute to the development of metabolic, regulatory, metabolic, and immunological disorders in the body and lead to quantitative and qualitative changes in normal microflora, in which not only the total number of intestinal microflora but also its individual representatives (Sajjadi and Moosavi 2018; Liu et al., 2021).
Since recently considerable attention has been paid to lectins of bacterial origin and the study of their functional role in the human and animal body to form a complete picture of the practical significance of Paenibacillus polymyxa 1460 lectin (LII) and its role in the regulation of metabolic processes in the body of animals, it is of great interest to study its influence on the processes of lipid peroxidation and on the natural intestinal microflora under swimming stress, which was the purpose of this work. Hence, given the significance and necessity of the issue, the authors have chosen to investigate this matter closely.
MATERIAL AND METHODS
We used LII lectin isolated from the surface of soil nitrogen-fixing bacteria Paenibacillus polymyxa 1460 (Karpunin et al., 1993). The studies were carried out on healthy male white outbred rats with an average body weight of 210 g. The animals were kept under standard vivarium conditions: 12-hour illumination period, temperature 20 ºС, food and water ad libitum. The lectin preparation was administered to rats intraperitoneally at a dose of 2 μg per animal in physiological solution in a volume of 0.2 ml for three days daily. Swimming stress was carried out by subjecting the animals to forced swimming with weights (a load of 7% of body weight was tied to the tail) in water at a temperature of 25 ºС, recording the swimming time of the animals (Dasgupta et al., 2020; Liu et al., 2021).
Determination of the activity of peroxidase in the blood of animals was carried out according to the method (Arkhipova, 1988). The decision of microflora in animals was carried out by inoculating the contents of their large intestine on Petri dishes on selective media for lactic acid bacteria, Escherichia coli, staphylococci by the method of serial dilutions (Kostenko et al., 2001). According to the nature of the impact, the experimental animals were divided into 4 groups: group 1 – control animals; Group 2 – animals that received an injection of a solution of lectin LII; Group 3 – animals that were stressed by swimming; Group 4 – animals that previously received an injection of a solution of lectin LII, and they were subjected to swimming stress
Statistical processing of the data obtained was carried out using the Student’s t-test (Wang et al., 2020).
RESULTS AND DISCUSSION
As a result of the studies carried out in the study of the effect of bacillus lectin LII on some biochemical parameters of the blood of male rats under swimming stress, it was found that the swimming time of animals with preliminarily introduced bacillus lectin at a dose of 2 μg / animal was 1.9 times longer than the swimming time in the control group. Bacterial lectin LII increases the physical endurance of male rats. So, if the control animals swam for an average of (99.76 ± 4.26) minutes, then the rats, which were preliminarily injected with lectin of bacteria LII P. polymyxa 1460, swam (190.93 ± 1.73) minutes (Table 1), which in percentage terms was an increase in physical activity by 91.4%.
Table 1. The effect of lectin (LII) on the swimming time of male rats
| The nature of the impact | Swimming time, min | Swimming time,% |
| Swimming | 99,76±4,26 | 100 |
| Lectin LII + swimming | 190,93±1,73* | 191,4 |
Note – the differences are significant at P <0.05 relative to the values: * – swimming.
The data obtained indicate the activation of metabolic processes in the body of experimental animals by lectin (LII).
Thus, in the course of the experiment, it was found that intense physical activity leads to the activation of LPO processes and the accumulation of its products. Possibly, the lectin P. polymyxa 1460 is a natural antioxidant and mobilizes the body, normalizing the pituitary – adrenocortical system, intestinal microflora, brings the lipid peroxidation system and antioxidant defense into equilibrium (Wang et al., 2020; Liu et al., 2021). Thus, P. polymyxa 1460 lectin suppresses oxygen consumption and accumulation of lipid peroxidation products. The activity of blood peroxidase in the administration of P. polymyxa lectin to 1460 male white rats increased 2.3 times, which is a favorable moment for the activation of the antioxidant defense reaction of the body (Fig. 1). It was noted that during exercise, the activity of peroxidase decreases 1.4 times from (0.13 ± 0.015) to (0.09 ± 0.004) mmol / L * s (Fig. 1).
Figure 1: Influence of bacterial lectin LII on the activity of blood peroxidase in male rats during swimming
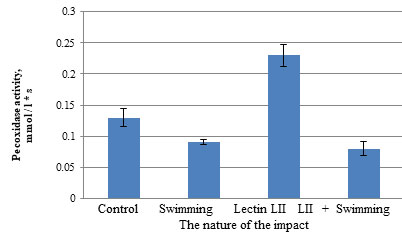
polymyxa 1460 lectin led to an increase in catalase activity by 26% relative to the group of animals that received injections of saline (Fig. 2). This can be considered a positive point, since the body’s defense reaction is activated, the antioxidant system is activated.
Figure 2: Effect of bacterial lectin LII Paenibacillus polymyxa 1460 on the activity of catalase in blood plasma of male rats during swimming
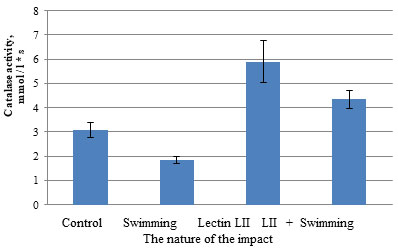
It was noted that during physical exertion, there is a decrease in catalase activity by 1.7 times from (3.07 ± 0.305) to (1.83 ± 0.141) μat/L. Preliminary administration of LII lectin to male white rats before swimming leads to an increase in catalase activity from (1.83 ± 0.141) to (4.34 ± 0.370) μat / L. P. polymyxa 1460 lectin LII had a positive effect on the activity of antioxidant enzymes (catalase and peroxidase). Catalase activity increased from (3.07 ± 0.305) to (5.90 ± 0.850) μat/L. Thus, the activity of catalase in the blood serum of rats during swimming against the background of prolonged administration of lectin from bacteria LII P. polymyxa 1460 corresponded to those of animals in the control group (4.34 ± 0.370) μat/L. It was shown that the administration of P. polymyxa lectin LII to 1460 male white rats did not change the MDA content in blood erythrocytes. However, it was noted that during exercise, the content of malondialdehyde increases more than three times, or rather 3.4 times from (6.03 ± 0.490) to (20.46 ± 1.250) μmol / L.
Figure 3: Influence of lectin of bacteria Paenibacillus polymyxa 1460 on the content of malondialdehyde in the blood of rats during swimming
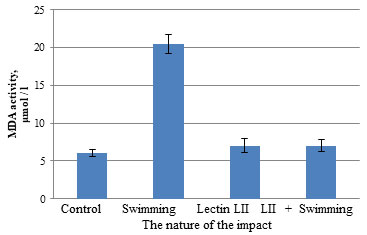
Preliminary administration of P. polymyxa LII bacterial lectin to 1460 male white rats before swimming leads to a decrease in the MDA content from (20.46 ± 1.250) to (7.03 ± 0.840) μmol/L. Under combined exposure (introduction of bacterial lectin P. polymyxa 1460 followed by swimming) a significant change in the content of lipid peroxidation products and antioxidant activity was observed: the MDA content corresponded to the control values (7.03 ± 0.840) μmol/L. When P. polymyxa lectin LII was administered to 1460 male white rats, no changes were observed in the DC content in rat erythrocytes, but during swimming, the content of diene conjugates in the blood erythrocytes of male white outbred rats decreased by 1.2 times relative to the control group with (8.23 ± 0.690) to (6.59 ± 0.110) μmol/L (Fig. 4). Since diene conjugates are the primary product of lipid peroxidation, it can be assumed that their decrease is due to the fact that a sufficient amount of time has passed since the start of exposure (Pees et al., 2021; Liu et al., 2021).
Figure 4: Influence of lectin LII Paenibacillus polymyxa 1460 for the content of diene conjugates in the blood of rats during swimming
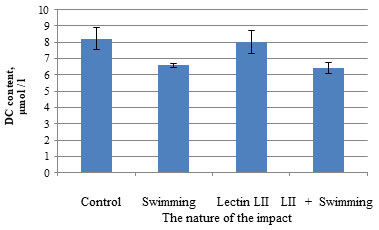
Preliminary administration of P. polymyxa lectin LII to 1460 male white rats before swimming almost does not change the content of diene conjugates. Administration of P. polymyxa 1460 lectin LII does not affect the content of lipid peroxidation products in rat erythrocytes. The content of diene conjugates in the erythrocytes of the blood of male rats was (8.23 ± 0.69) µmol / L in the control group and (8.03 ± 0.70) µmol / L in the group of animals receiving lectin.
Under combined exposure (introduction of LII lectin Paenibacillus polymyxa 1460 followed by swimming), a significant change in the content of lipid peroxidation products and the activity of antioxidants was observed: the content of diene conjugates did not reach the control values (8.23 ± 0.690) μmol / L and remained at the level of animal values, exposed to swimming (6.43 ± 0.340) μmol/L. Taking into account the fact that LPO processes are the limiting factor in the development of fatigue, it can be assumed that the use of bacterial lectin is justified in order to increase efficiency at maximum physical exertion (Wang et al., 2020).
In the course of the studies, it was found that the introduction of bacterial lectin LII to rats led, as can be seen from Table 2, to an increase in the number of lactic acid bacteria in the large intestine (bifidobacteria by 46% and lactobacilli by 57%) and a decrease in the number of E. coli by 75% and staphylococci by 85%, relative to the values of the control group. In the group of animals that were subjected to swimming, a decrease in the number of bifidobacteria and lactobacilli by 39% and 37%, respectively, and an increase in the amount of facultative microflora were observed.
Perhaps, in this case, stress leads to a slowdown in the general metabolic processes in the body and to a slowdown in the growth of lactic acid bacteria. The results obtained are in good agreement with the literature data, according to which many stress factors lead to a decrease in the amount of lactic acid microflora in the intestines of animals and an increase in the pathogenic one (Pees et al., 2021).
The preliminary introduction of lectin before swimming stress contributed to the normalization of the microflora of the large intestine (Table 2).
Table 2. Effect of Paenibacillus polymyxa 1460 lectin (LII) and the effect of stress on the natural microflora in the large intestine of rats
| Microorganisms | Amount КОЕ х 106 /г | ||
| Control | Swimming | LII + Swimming | |
| Bifidobacteria | 3,80±0,20 | 2,30±0,10* | 3,95±0,22* |
| Lactobacillus | 3,25±0,22 | 2,05±0,12* | 3,80±0,10* |
| Colibacillus | 1,20±0,20 | 2,75±0,15 | 1,15±0,20 |
| Staphylococcus | 1,35±0,16 | 2,60±0,16 | 1,60±0,12 |
Note – * p <0.05 relative to the control group.
Thus, as evidenced by the conducted studies, Paenibacillus polymyxa 1460 lectin LII is able to normalize the microflora of the large intestine under stress by swimming (Dasgupta et al., 2020; Liu et al., 2021).
CONCLUSION
Lectin LII P. polymyxa 1460 suppresses oxygen consumption and the accumulation of lipid peroxidation products, which contributes to an increase in performance, endurance, the resistance of the body, increasing the swimming time. Thus, it can be assumed that P. polymyxa 1460 lectin LII is a natural antioxidant and thereby suppresses the accumulation of DCs in the blood erythrocytes of white male rats, which leads to a decrease in LPO products. Based on the results obtained, we can say that P. polymyxa 1460 lectin LII exhibits antioxidant properties and can be successfully used as a natural antioxidant, being an effective tonic, bio stimulating agent, providing a complex healing effect on the body, playing an important role in the regulation of metabolic processes of the animal body against a background of stress. The data obtained indicate that Paenibacillus polymyxa 1460 lectin LII increases the resistance of animals in extreme conditions caused by stress.
ACKNOWLEDGEMENTS
The authors received no direct funding for this research.
Conflict of Interests: the authors state that there is no conflict of interest.
REFERENCES
Arkhipova OG (1988) Research methods in occupational pathology. M Medicine, pp 153-154.
Dasgupta M, Shashikanth M, Gupta A, et al., (2020). NHR-49 transcription factor regulates immunometabolic response and survival of Caenorhabditis elegans during Enterococcus faecalis infection. Infection and immunity, Vol 88 No 8 pp 1-16.
Gorelnikova EA (2006). The effect of bacillus lectin on the cytokine activity of phagocytes: discand. biol. sciences. Saratov, pp 123.
Karpunin LV, Melnikova UYu, Vishnevetskaya O.A and Nikitina VE (1993). Microbiology. Vol 62 No 2 pp 307-313.
Kostenko, T.S., Radionova, V.B., Skorodumov, D.I. (2001). Workshop on Veterinary Microbiology and Immunology. M, Kolos, pp 344.
Liu J, Wu P, Guo Q, et al., (2021). Kaolinite weakens the co-stress of ampicillin and tetracycline on Escherichia coli through multiple pathways. Environmental Science and Pollution Research, pp 1-13.
Pees B, Yang W, Kloock A et al., (2021). Effector and regulator: Diverse functions of C. elegans C-type lectin-like domain proteins. PLoS pathogens, Vol 17 No 4 e1009454.
Sajjadi A, Moosavi SM (2019). Synthesis of polymer-coated RDX/AP nano-composites using supercritical CO2. Journal of Medicinal and Chemical Sciences Vol 1 No 1 pp 9-10.
Sheikhshoaie I, Sheikhshoaei M and Ramezanpour S (2018). Synthesis and Characterization of Nano Sized ZnO and CdO by Direct Thermal Decomposition of Their Nano Sized Metal Schiff base Complexes. Chemical Methodologies. Vol 2 No 2 pp 103-113.
Shenderov BA (1987). Normal microflora and some issues of microecological toxicology. Antibiotic. and honey. Biotechnol, Vol 32 No2 pp 18-24.
Vorobiev AA, Abramov NA, Bondarenko VM and Shenderov BA (1997). Dysbacteriosis – an urgent problem of medicine. Bulletin of the Russian Academy of Medical Sciences, Vol No 3 pp 4-7.
Wang W, Wang G, Zhuo X et al., (2020). C-type lectin-mediated microbial homeostasis is critical for Helicoverpa armigera larval growth and development. PLoS Pathogens, Vol 16 No 9 pp e1008901.


