Department of Periodontology and Community Dentistry, College of Dentistry, King Saud University, Riyadh, Saudi Arabia
Corresponding author email: raqeely@ksu.edu.sa
Article Publishing History
Received: 05/12/2020
Accepted After Revision: 25/03/2021
Ridge split is one of the techniques utilized to increase bone width before or at the time of implant placement. The paper reviewed the ridge splitting technique since its start, its indications and drawbacks, and the instruments utilized to perform it. The technique can be done in one or two stages depending on initial bone width and cross-sectional form. The aim of this paper is to review the current knowledge about ridge split different techniques with report of 2 cases utilizing those techniques in atrophic maxilla. Two patients with edentulous atrophic maxillae are reported. Ridge split technique was chosen as the treatment modality for dental implant placement. One patient was treated with 2-step ridge splitting approach while the other with simultaneous ridge splitting with implant placement. Patients treated with two-stage and one-stage ridge splitting had their prosthetic delivery after six months of temporization. The survival at that time was 100%. Different techniques of preparing bone for dental implants are well-known. The combination of knowledge and clinical skills are important in deciding the best technique in each clinical scenario. Ridge splitting is one of those techniques that can be used in specific type of clinical presentations.
Atrophic Maxilla, Bone Graft, Dental Implant, Piezo Surgery, Ridge Split.
Al-Hamdan K, Alaqeely R, Gamal A. A. Managing Atrophic Maxilla Using Ridge-Split Technique: a Review Based Analysis with two case Reports. Biosc.Biotech.Res.Comm. 2021;14(1).
Al-Hamdan K, Alaqeely R, Gamal A. A. Managing Atrophic Maxilla Using Ridge-Split Technique: a Review Based Analysis with two case Reports. Biosc.Biotech.Res.Comm. 2021;14(1). Available from: <a href=”https://bit.ly/3pTKc1X”>https://bit.ly/3pTKc1X</a>
Copyright © Al-Hamdan et al., This is an open access article distributed under the terms of the Creative Commons Attribution License (CC-BY) https://creativecommns.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, provide the original author and source are credited.
INTRODUCTION
Jaw atrophy involves a reduction of alveolar height and width together with bone remodeling that affects the external shape and internal bone structure. It occurs chronically and irreversibly following tooth extraction, trauma, infection, pneumatization of the maxillary sinus, or ablative tumor surgery (Ishak and Kadir, 2013). However, the pattern of alveolar ridge atrophy varies between the maxilla and the mandible— the maxilla exhibits centripetal resorption, while the mandible shows centrifugal resorption (Berger et al., 2019).The bearing area available following atrophic maxilla may be inadequate, and this can lead to a lack of prosthesis retention, causing both functional and physiological problems for a patient (Dohiem et al., 2015).Oral rehabilitation in areas where bone width is insufficient is complex.
Insufficient bone width is common in edentulous patients, especially when alveolar fracturing occurs during dental extraction. When the bone loss results from a maxillofacial trauma, vertical dental root fracture, or from extensive periodontal/endodontic diseases, the effects are even more severe. Bone loss might result in insufficient vertical and horizontal support to install dental implants and may impair, or even limit, the options available for prosthetic rehabilitation (Waechter et al., 2017). These problems can be treated for patient satisfaction with an implant-supported fixed or removable complete or partial denture. Atrophic edentulous jaws can represent a significant challenge to the successful use of endosseous implants for prosthetic reconstruction of the edentulous mandible (Eufinger et al., 1997, Tolstunov et al., 2019).
Several methods have been employed to augment the alveolar crest; for example, guided bone regeneration, bone block grafting, ridge splitting for bone expansion, and distraction osteogenesis. Guided bone regeneration (GBR) using resorbable membranes in combination with particulate autologous bone or a mixture of autologous bone chips and xenogenic bone material, autogenous block onlay grafts harvested intraorally or from the hip, or distraction osteogenesis have been suggested for alveolar ridge augmentation. These treatment options increase the treatment time and costs, have a conspicuous risk of dehiscence and infections and negatively affect patients’ morbidity (Bassetti et al., 2016, Starch-Jensen and Becktor, 2019).
Splitting and expanding the edentulous ridge for bone augmentation and implant placement is considered to represent an innovative technique because it avoids the need for a second surgical site, which further reduces the ailment of the patient (Kumar et al., 2016). In 1986, Nentwig reported a bone crest division technique that simultaneously allowed the expansion of the alveolar crest and implant insertion (Nentwig, 1986, Li et al., 2020). Later in the early nineties, Simion et al. aimed to create a ‘‘self-space making defect’’ by splitting the atrophic crests into two parts with a longitudinal greenstick fracture displacing the vestibular cortical bone both in the maxilla or mandible to create a gap into which the implants were subsequently inserted (Simion et al., 1992, Li et al., 2020).
The split ridge technique (SRT) is recommended when the ridge width is insufficient, but the alveolar height is acceptable. However, in ridges with low elasticity, trabecular bone volume is compromised, and bone expansion will be less predictable. This can undermine the success of the technique (Mechery et al., 2015, Waechter et al., 2017). A recent systematic review suggested several anatomical requirements are necessary for SRT: 2-3 mm minimally of ridge width, minimum bone height of 10 mm, presence of type III or IV of bone, absence of concavities in bone profile, and 1 mm between adjacent teeth in case of partial edentulism (Bassetti et al., 2016). Ridge width is an essential factor in planning a suitable approach for bone augmentation in isolation or in combination with dental implant placement. An updated decision tree on horizontal bone augmentation suggested the use of the ridge split option when the width of the ridge was a minimum of 3.5mm (Mechery et al., 2015).
This amount of bone is essential to allow the splitting of alveolar bone into three layers of bone: Two cortical plates (buccal and palatal/lingual plates) and one layer of cancellous bone to allow ridge expansion (Tolstunov and Hicke, 2013). However, the bone morphology can have a direct impact on the suitability of the ridge split technique within a given case; the presence of bone concavities, a narrow base of less than 3 mm, and hour-glass shape ridges are factors that should be carefully examined before attempting ridge splitting (Elnayef et al., 2015, Tolstunov et al., 2019). The technique is considered relatively fast as healing occurs in the same way as that observed with bone fractures; by rapid vascularization and remodeling of bone (Kumar et al., 2016, Tolstunov et al., 2019).
To avoid major complications, including plate fracture, several factors must be carefully assessed before planning a ridge split. Cortical plate fracture results from poor case selection in the presence of a thick cortex (Li et al., 2017). In the case of a plate fracture, it is important to avoid dislodgment of the fragmented bone as this might result in bone necrosis and, subsequently, more severe ridge defects due to problems in perfusion and remodeling resorption (Teng et al., 2014, Dohiem et al., 2015, Li et al., 2017). Another issue concerns the risk of buccal exposure of dental implant or osseointegration (Teng et al., 2014, Berger et al., 2019), which can be avoided by maintaining at least 1.5 mm of bone buccally (Spray et al., 2000, Teng et al., 2014, Berger et al., 2019).
If the procedure fails, a massive bone loss will occur and complicate the treatment (Arora and Kumar, 2015) which make it often perceived to be inferior to other augmentation techniques (Kaneko et al., 2013). In some cases, implant stability might be low; however, choosing a tapered implant will increase the primary stability and also decrease the incidence of fracture (Elnayef et al., 2015, Yao et al., 2018). However, implant stability is considered to represent a superior approach as new bone is formed between the two cortical plates (Arora and Kumar, 2015, Berger et al., 2019).The ridge splitting technique is considered an advantageous procedure that eliminates the need for further surgery as it allows simultaneous implant placement. As such, it reduces treatment time and morbidity (Arora and Kumar, 2015, Anitua and Alkhraisat, 2016, Bassetti et al., 2016, Yao et al., 2018, Li et al., 2020). Furthermore, postoperative, swelling and pain are lower with this approach than it is with alternative augmentation techniques (Kumar et al., 2016, Altiparmak et al., 2017).
Ridge Split Technique :The ridge split technique was initially described as a one-stage ridge split in which implants are placed, followed immediately by ridge splitting (Nentwig, 1986). It offers lower morbidity, cost, and time of treatment and is advantageous in terms of bone healing (Bassetti et al., 2016, Li et al., 2020). However, it is important to carefully evaluate the density and width of the bone to achieve an acceptable implant primary stability (30Ncm or more) during one-stage ridge splitting (Demetriades et al., 2011, Zhang and Huang, 2020). In 2013, the Osborn technique was introduced, which involves performing the ridge splitting process over two stages. During the first stage, the inter-cortical area is filled with autogenous bone or bone substitutes, while the implants are placed eight-to-twelve weeks later in a second procedure (Gonza ́lez-Garc ́ıa et al., 2011, Li et al., 2020).
A two-stage approach may be used when the ridge is narrower than 3 mm, as this is associated with an increased risk of buccal plate fracture, or when the implant stability is questionable (Anitua and Alkhraisat, 2016, Kumar et al., 2016). The presence of a bone graft increases the vascularization during implant bed preparation and protects against compromised implant placement angulations (Cha et al., 2014, Arora and Kumar, 2015). Demetriades and his group analyzed the difference between the two approaches and found that osseointegration did not vary between the one- and two-stage processes; however, there were fewer postoperative complications in patients who underwent the two-stage ridge splitting process (Demetriades et al., 2011, Li et al., 2020).
Traditionally, flap reflection in ridge splitting was advocated to be a full-thickness flap as excessive bleeding can be avoided, making handling and visualization more straightforward (Agrawal et al., 2014, Tolstunov et al., 2019). A partial-thickness flap was suggested to preserve blood supply, which helps to protect the bone from excessive loss (Scipioni et al., 1994, Elnayef et al., 2015, Starch-Jensen and Becktor, 2019).In terms of two-stage SRT, Dohiem et al. (2015), explored the concept of using a full-thickness flap in the first stage to enable better control during the surgical steps, and a partial thickness in the second stage during implant placement to protect from further bone loss.
Osseous ridge splitting can be performed using a variety of instruments, both manually and motor-driven. The use of manual instruments, like blade No. 15, razor-sharp chisel, and beaver blade, is challenging when dealing with cortical bone and attempting to cut small amounts. However, they can provide a good control (Kumar et al., 2016, Li et al., 2017, Li et al., 2020). The use of either round burs or diamond disks can help to remove the bulk of the bone: however, these instruments generate heat, which might affect bone healing or lead to bone necrosis (Kumar et al., 2016, Li et al., 2017, Li et al., 2020). New modalities have been proposed and used in SRT-like laser (erbium: yttrium–aluminum- garnet, erbium, chromium-doped: yttrium-scandium-gallium-garnet), micro saw devices, and piezoelectric devices (Vercellotti, 2000, Zhang and Huang, 2020).
Piezoelectric devices are fast, safer and more accurate than other modalities. These devices work at a 25-30kHz frequency that makes it possible to control the splitting of bone close to vital structures as mental foramen and maxillary sinus (Kumar et al., 2016). Also, the oscillating frequency makes it possible for practitioners to perform a selective and less invasive cut, while the micromovement cuts bone but not soft tissue (Agrawal et al., 2014, Li et al., 2017). Unlike motor-driven and micro saw devices, the piezosurgical saw does not produce heat, which reduces the probability of postoperative bone necrosis (Crespi et al., 2014, Kumar et al., 2016, Li et al., 2020).
Practitioners have traditionally created the split in the bone before expanding it using a hand mallet. However, in more recent times, piezoelectric devices have been used. The piezo-surgical system helps to overcome the risk of displacement of bone fragments and vertigo (benign paroxysmal positional vertigo -BPPV-) associated with hand mallet percussions. As such, the piezo-surgical system can help to reduce the discomfort of patients (Crespi et al., 2014, Kheur et al., 2014, Li et al., 2020).
The ridge split technique is usually carried out in the maxilla, where the bone is more spongier and the cortical plates are relatively thin compared to the mandible (Kumar et al., 2016). The majority of the published cases that describe the use of SRT involved the replacement of a single tooth or multiple teeth in the arch in the maxillary and mandibular arches. The next section presents two case reports that describe full edentulous maxillary arches restoration using the ridge-splitting technique.
CASE PRESENTATION: Two edentulous patients attended the dental college of King Saud University. After an assessment of the patients’ medical and dental conditions, the split ridge technique was chosen to place dental implants in the upper jaw. Consent forms were signed to proceed with the planned treatment.
Case 1: A 47-year-old Moroccan female visited the Dental University Hospital (DUH) at King
Figure 1: Initial clinical and radiographic presentation
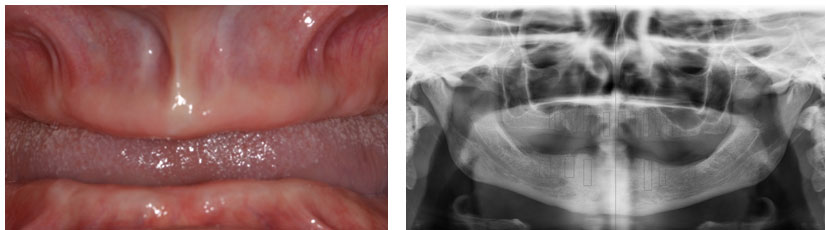
Saud University, Riyadh, SA. The patient was unaware of any medical condition and was seeking a fixed replacement for edentulism. The patient had been referred from the prosthodontic. After a clinical examination of the hard and soft tissue quality, a radiographic stent was constructed, and CBCT scan was performed (Figure 1). The CBCT scans showed a relatively good height (11-14 mm) in the anterior upper, pneumatized sinuses with 5 mm of bone, and a lower arch of acceptable length and width (Figure 2).
Figure 2: Cone-beam CT scans (CBCT) showing the narrow width of planned implants in the upper arch at upper first premolars and incisors.
A treatment plan was discussed with the patient who signed the consent for upper-arch two-stage ridge splitting and a conventional implant placement in the lower jaw.One hour prior to surgery, the patient was given 1 g amoxicillin. Post-surgery, she was prescribed 1 g two times per day for a period of seven days. Surgery was performed under local anesthesia (Lidocaine 20 mg/mL with adrenaline 1:80,000).On the day of the surgery, a crestal incision was made, and a full-thickness flap was raised (Figure 3). A longitudinal mid-crestal osteotomy was performed using the piezosurgical saw in a side-to-side cutting motion. The depth of the first cut was 8-10 mm in relation to the anterior and premolar areas (Figure 3).
Figure 3: Full thickness flap reflection (left), Arrows indicating ridge after splitting (right).
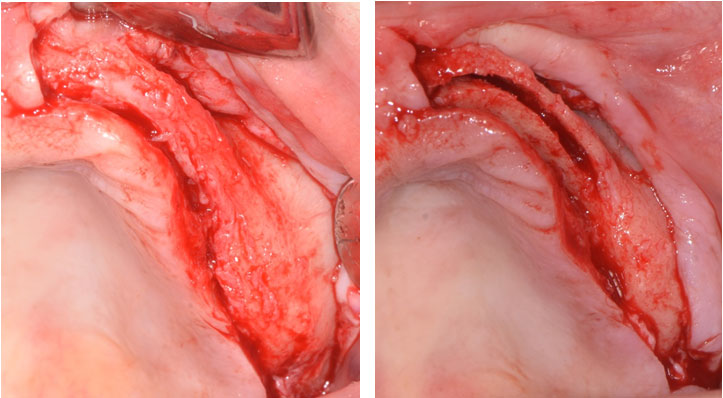
Two vertical bone incisions were made: one at the mesial and other at the distal aspect. The osteotomy site was expanded using expansion osteotomes until it was 6-7 mm wide (Figure 4).
Figure 4: Expansion of the osteotomy (left), and filling the site with allograft (right)
The site was grafted with allogenic cortical particulates allograft and covered with a resorbable membrane (Biomend Extend, Zimmer).After achieving primary closure, the site was sutured with horizontal mattress and interrupted sutures using 3-0 silk suture material. Healing was uneventful and within normal limits. The same surgical technique was performed on the other side.Unfortunately, the patient did not attend follow-up appointments for a couple of months due to family issues, but later returned to the clinic seeking completion of the treatment. A new CBCT scan was taken of the upper arch (Figure 5). The gain of the bone after the first split-ridge procedure was (1-2 mm).
Figure 5: cross sections of the new CBCT for the planned areas to place dental implants.
The new plan consisted of a one-stage ridge split with simultaneous implants placement. The same surgical protocol was followed as that performed during the first procedure. However, the implant was also placed (Figure 6).
Figure 6: Implant placement simultaneously with ridge splitting.
The patient was given a temporary denture until complete healing and maturation were observed (Figure 7). A screw-retained final prosthesis was delivered to the patient six months later (Figure 8).
Figure 7: Upper and lower temporary dentures.
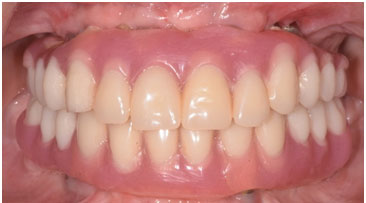
Figure 8: Final prosthetic treatment in intra and extra oral views
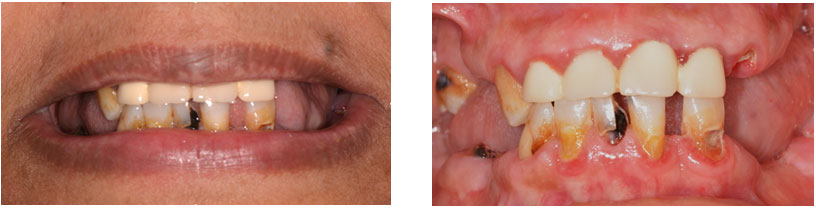
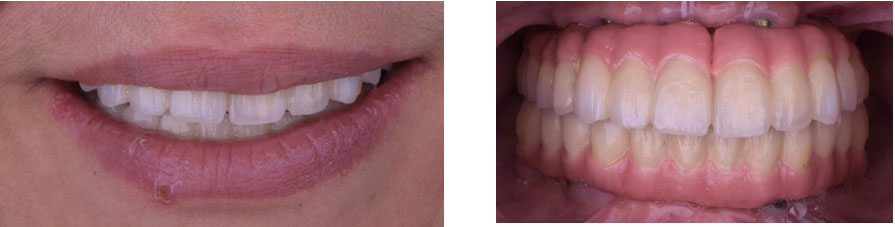
Case 2 : A 52-year-old Saudi female visited the dental clinic at the Dental University Hospital seeking treatment for her missing teeth and was referred for implant placement (Fig9). Consent forms were signed to proceed with the planned treatment.
Figure 9: Initial extra and intra oral status
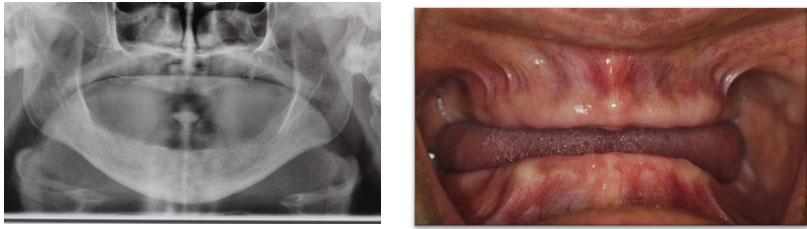
A full mouth extraction was done in addition to soft tissue grafting (free gingival graft) for lower right and left sides (Fig10).
Figure 10: Clinical and radiographic appearance after extraction and tissue grafting
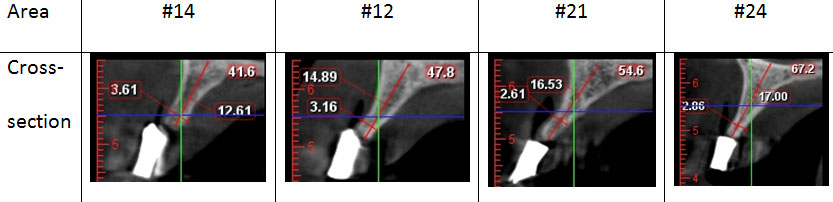
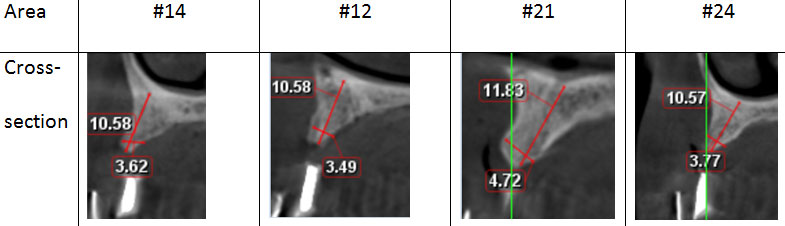
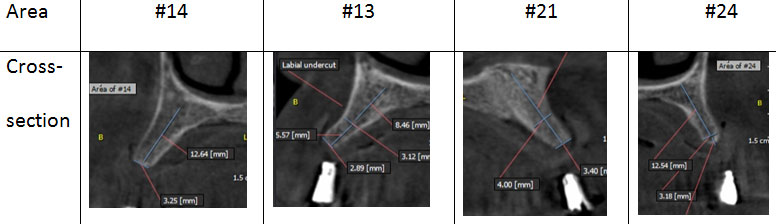
The CBCT was taken after the construction of complete dentures, and some measures are illustrated in Figure 11. The treatment plan consisted of the placement of eight implants in the upper arch with simultaneous ridge splitting from Area of #14 to Area of #26.
Figure 11: Cross sections for some implants planned areas as appeared in CBCT
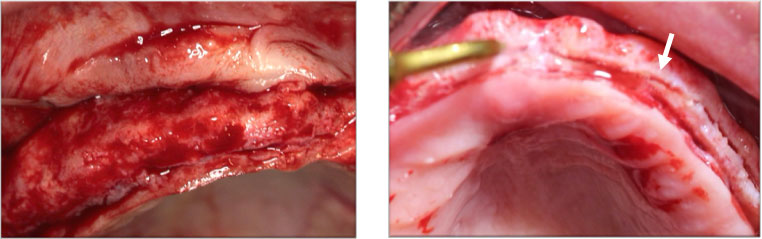
The same pre-operative medications were given to the patient as those administered to the patient in the first case. Local anesthesia was administered (Lidocaine 20 mg/mL with adrenaline 1:80,000), and a full-thickness flap was reflected before ridge splitting was performed using piezosurgery (Figure 12).
Figure 12: Full-thickness flap reflection (left) and splitting of the ridge using piezosurgery tip (right) .Arrow pointing to the splitting of the ridge
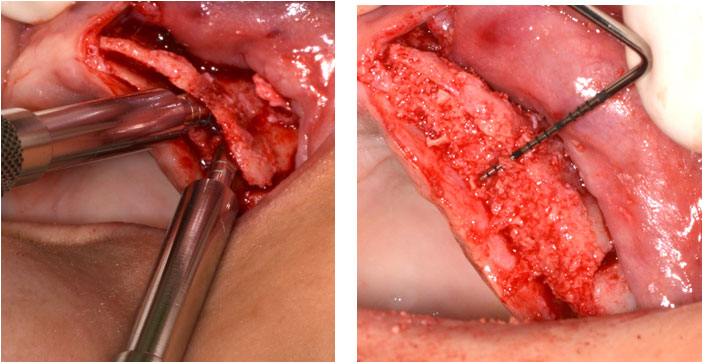
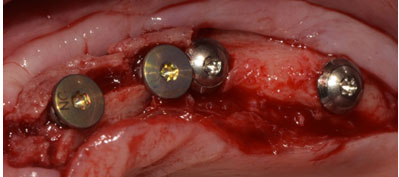
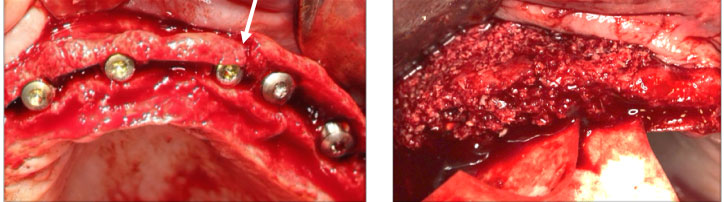
After ridge expansion, 3.3*10 mm Straumann implants were placed in the areas planned for the surgical stent. However, a vertical fracture occurred between Implant 23 and Implant 24 implants (Fig13). Cancellous particulate bone graft was packed and covered with a collagen resorbable membrane (Figure 13).
Figure 13: (left) implants placed after ridge expansion. Note the vertical fracture between #23 and24 implant fixtures(arrow). (Right) particulate bone graft and membrane were applied over the implants and secured.
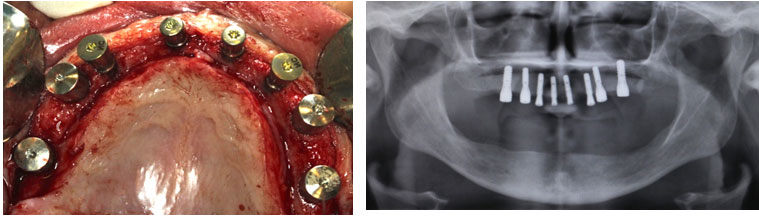
After 6 months, implant exposure was carried on placing healing abutments (Fig14) and final prosthesis was delivered after appropriate healing time.
Figure 14: Clinical and radiographic presentation after placing healing abutments
DISCUSSION
Different techniques have been developed to provide patients with high-quality dentition replacement. Dental implants are considered the ideal treatment modality in most clinical scenarios. Ridge splitting was developed to utilize existing bone to expand the ridge width dimensions and, thereby, aid implant placement. Since this procedure was first introduced in the 1970s by Tatum, many instruments have been developed for use with the ridge-splitting technique; for example, peizosaws and ridge-splitting osteotomes (Mechery et al., 2015, Li et al., 2020). In the 1990s, Summers and Scipiono et al. published data in which the five-year survival rate reached 98% (Scipioni et al., 1994, Summers, 1994). At that time, the criteria for case selection were more definite for the bone type to be more trabecular (D3 or D4) with proper vertical bone quantity.
In this paper, two patients’ clinical and radiographic findings mandate increase in bone thickness prior to dental implant placement. The decision of bone grafting technique was determined according to bone height and width present. The two patients’ dental condition as shown from CBCT sections were high enough to place a ten-millimeter ling implant but the width was compromised. Yet, the bone density and the bone form ,narrow crest and wide base, were key factors in selection of ridge splitting technique(Tolstunov et al., 2019, Zhang and Huang, 2020). Moreover, a recent systematic review concluded the efficacy of bone splitting with high implant survival(Starch-Jensen and Becktor, 2019).
In the first clinical case, ridge splitting was done in two-stage technique. The decision was based on the bone width presented initially as 2-3 mm, which in turns, mandate another stage with wider bone volume (Li et al., 2020) . In the second presented case, there was an evidence of bone fracture while placing the implants. This fracture as shown by Yao et al. (2018), aid in decreasing the tension on the buccal bone in anterior maxilla Improving the surgical operations with digitalized techniques will decrease the possible complications of exposing the bone and jeopardizing the blood supply, yet the accuracy of these approaches is sometimes questioned.
CONCLUSION
The fast and non-invasive nature of ridge splitting, and the superior bone healing observed after the application of this technique entails that it represents a preferred solution when the bone height and quality are adequate to allow the safe separation of the plates. Careful planning and utilization of instruments when splitting and expanding the ridge can provide a high standard treatment for function and esthetics with low morbidity and a short treatment time.
ACKNOWLEDGEMENTS
Not applicable
Competing Interests : Authors declare that they have no competing interests.
REFERENCES
Agrawal, D., Gupta, A. S., Newaskar, V., Gupta, A., Garg, S. & Deshraj, J. (2014). Narrow Ridge Management with Ridge Splitting with Piezotome for Implant Placement Report of 2 Cases. J Indian Prosthodont Soc, 14, 305–309.
Altiparmak, N., Akdeniz, S. S., Bayram, B., Gulsever, S. & Uckan, S. (2017). Alveolar Ridge Splitting Versus Autogenous Onlay Bone Grafting: Complications and Implant Survival Rates. Impl Dent, 26, 284-287.
Anitua, E. & Alkhraisat, M. H. (2016). Is Alveolar Ridge Split a Risk Factor for Implant Survival? J Oral Maxillofac Surg, 74, 2182-2191.
Arora, V. & Kumar, D. (2015). Alveolar ridge split technique for implant placement. Med J Armed Forces India, 71, S496-8.
Bassetti, M. A., Bassetti, R. G. & Bosshardt, D. D. (2016). The alveolar ridge splitting/expansion technique: a systematic review. Clin Oral Implants Res, 27, 310-24.
Berger, S., Hakl, P., Sutter, W., Meier, M., Roland, H., Bandura, P. & Turhani, D. (2019). Interantral alveolar ridge splitting for maxillary horizontal expansion and simultaneous dental implant insertion: A case report. Ann Med Surg (Lond), 48, 83-87.
Cha, M. S., Lee, J. H., Lee, S. W., Cho, L. R., Huh, Y. H. & Lee, Y. S. (2014). Horizontal Ridge Augmentation with Piezoelectric Hinge-Assisted Ridge Split Technique in the Atrophic Posterior Mandible. Maxillofac Plast Reconstr Surg, 36, 124-30.
Crespi, R., Capparé, P. & Gherlone, E. F. (2014). Electrical mallet provides essential advantages in split-crest and immediate implant placement. Oral Maxillofac Surg, 18, 59–64.
Demetriades, N., Park, J. I. & Laskarides, C. (2011). Alternative Bone Expansion Technique for Implant Placement in Atrophic Edentulous Maxilla and Mandible. J Oral Impl, 37, 463-471.
Dohiem, M., Nassar, H. & Charkawi., E. (2015). Bone changes in ridge split with immediate implant placement. Future Dental Journal, 1, 6-12.
Elnayef, B., Monje, A., Lin, G. H., Gargallo-Albiol, J., Chan, H. L., Wang, H. L. & Hernandez-Alfaro, F. (2015). Alveolar ridge split on horizontal bone augmentation: a systematic review. Int J Oral Maxillofac Implants, 30, 596-606.
Eufinger, H., Gellrich, N. C., Sandmann, D. & Dieckmann, J. (1997). Descriptive and metric classification of jaw atrophy. An evaluation of 104 mandibles and 96 maxillae of dried skulls. Int J Oral Maxillofac Surg, 26, 23-8.
Gonza ́Lez-Garc ́Ia, R., Monje, F. & Moreno, C. (2011). Alveolar split osteotomy for the treatment of the severe narrow ridge maxillary atrophy a modified technique. Int. J. Oral Maxillofac. Surg., 40 57–64.
Ishak, M. & Kadir, M. (2013). Treatment Options for Severely Atrophic Maxillae. Biomechanics in Dentistry: Evaluation of Different Surgical Approaches to Treat Atrophic Maxilla Patients. Springer Science & Business Media: Springer.
Kaneko, T., Masuda, I., Hino, S., Horie, N. & Shimoyama, T. (2013). Dental implants placed in thin maxilla expanded using a modified bone-splitting procedure Case series. J Oral Maxillofac Surgery, Medicine, and Pathology 25, 250–254.
Kheur, M., Gokhale, S., Sumanth, S. & Jambekar, S. (2014). Staged ridge splitting technique for horizontal expansion in mandible: a case report. J Oral Implantol, 40, 479-83.
Kumar, A. T., Triveni, M., Priyadharshini, V. & Mehta, D. (2016). Staged Ridge Split Procedure in the Management of Horizontal Ridge Deficiency Utilizing Piezosurgery. J Maxillofac Oral Surg, 15, 542-546.
Li, X., Xu, P., Xu, X. & S., L. (2017). The application of a delayed expansion technique for horizontal alveolar ridge augmentation in dental implantation. J. Oral Maxillofac. Surg., 46, 1451–1457.
Li, X. M., Bao, J. B. & Xie, Z. G. (2020). Application of two-stage ridge splitting technique in atrophic mandibular alveolar ridge. Hua Xi Kou Qiang Yi Xue Za Zhi, 38, 338-342.
Mechery, R., Thiruvalluvan, N. & Sreehari, A. K. (2015). Ridge split and implant placement in deficient alveolar ridge: Case report and an update. Contemp Clin Dent, 6, 94-7.
Nentwig, G. H. (1986). Technic of bone splitting for alveolar recession in anterior maxillary region. Quintessenz, 37, 1825-34.
Scipioni, A., Bruschi, G. & Calesini, G. (1994). The edentulous ridge expansion technique: A five-year study. Int J Periodontics Restorative Dent 14, 451– 459.
Simion, M., Baldoni, M. & Zaffe, D. (1992). Jawbone enlargement using immediate implant placement associated with a split-crest technique and guided tissue regeneration. International Journal of Periodontics & Restorative Dentistry, 12, 463-73.
Spray, J., Black, C. & Morris, H. (2000). The influence of bone thickness on facial marginal bone response: stage 1 placement through stage 2 uncovering. Ann Periodontol 5, 119-128.
Starch-Jensen, T. & Becktor, J. P. (2019). Maxillary Alveolar Ridge Expansion with Split-Crest Technique Compared with Lateral Ridge Augmentation with Autogenous Bone Block Graft: a Systematic Review. J Oral Maxillofac Res, 10, e2.
Summers, R. (1994). The osteotome technique: Part 2—The ridge expansion osteotomy (REO) procedure. Compendium, 15, 422.
Teng, F., Zhang, Q., Wu, M., Rachana, S. & Ou, G. (2014). Clinical use of ridge-splitting combined with ridge expansion osteotomy, sandwich bone augmentation, and simultaneous implantation. Br J Oral Maxillofac Surg, 52, 703-8.
Tolstunov, L., Hamrick, J. F. E., Broumand, V., Shilo, D. & Rachmiel, A. (2019). Bone Augmentation Techniques for Horizontal and Vertical Alveolar Ridge Deficiency in Oral Implantology. Oral Maxillofac Surg Clin North Am, 31, 163-191.
Tolstunov, L. & Hicke, B. (2013). Horizontal augmentation through the ridge-split procedure: a predictable surgical modality in implant reconstruction. J Oral Implantol, 39, 59-68.
Vercellotti, T. (2000). Piezoelectric surgery in implantology: A case report—A new piezoelectric ridge expansion technique. Int J Periodontics Restorative Dent 20, 358.
Waechter, J., Leite, F. R., Nascimento, G. G., Carmo Filho, L. C. & Faot, F. (2017). The split crest technique and dental implants: a systematic review and meta-analysis. Int J Oral Maxillofac Surg, 46, 116-128.
Yao, Y., He, K., Gong, P. & Tang, H. (2018). U-Shaped Bone Splitting and Osteotome Techniques for Narrow Alveolar Ridge in Implant Surgery. Implant Dent, 27, 507-511.
Zhang, L. & Huang, Y. (2020). Radiographic Evaluation of the Alveolar Ridge Splitting Technique Combined with Guided Bone Regeneration vs Guided Bone Regeneration Alone in the Anterior Maxilla: A Retrospective Controlled Study. Int J Periodontics Restorative Dent.


