1Department of Clinical Nutrition, College of Applied Health Sciences
in Ar Rass, Qassim University, Al Qassim, Saudi Arabia
2Department of Radiology, College of Medicine and Medical Sciences, Qassim
University, Al Qassim, Unaizah, Saudi Arabia
3Department of Clinical Nutrition, College of Applied Medical Sciences, Jazan
University, Jazan, Saudi Arabia
Corresponding author email: f.masfoor@qu.edu.sa
Article Publishing History
Received: 11/10/2021
Accepted After Revision: 21/12/2021
Hypertension is a major cardiovascular problem resulting in significant mortality. Cissus quadrangularis having several pharmacological effects has not been evaluated for its ability to modulate blood pressure. Thus, the ability of C. quadrangularis aqueous extract (CQE) to modulate blood pressure was evaluated in normotensive and angiotensin II-induced hypertensive rats under urethane anesthesia. The animals were divided into four groups namely, control (saline injection), CQE (extract alone, 10 mg/kg), Ang II (Ang II alone, 0.5 µg/kg) and Ang II + CQE (Ang II + extract). All treatments were delivered by intravenous route and in Ang II + CQE group, Ang II was injected 30 min after injection of the extract. Hemodynamic parameters, systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial blood pressure (MABP), and heart rate (HR) were recorded by the BIOPAC system after the cannulation of the carotid artery and jugular vein.
The results indicated that CQE lowered SBP, DBP, MABP and heart rate to varying degrees in normotensive rats compared to control groups. In case of angiotensin II-induced hypertension, CQE administration resulted in substantial decrease in SBP, DBP, and MABP which were raised by Ang II. CQE reduced SBP, DBP, and MABP by 12, 59, and 11%, respectively. It is worth noting that, while SBP was not brought down to baseline levels by CQE, DBP was, suggesting significant hypotensive/antihypertensive activity of CQE. Further research is required to determine the molecular mechanism of C. quadrangularis extract’s hypotensive/antihypertensive action and to conduct clinical trials to establish its optimal use as an antihypertensive therapeutic.
ANTIHYPERTENSIVE, AT1, AT2, HADJOD, HYPOTENSIVE
Ahmed F, Al-Khalifah B. I. A, Ahmad M. F, Alhodieb F. S. Cissus quadrangularis Aqueous Extract Attenuates Angiotensin II-Induced Hypertension in Urethane-Anesthetized Rats. Biosc.Biotech.Res.Comm. 2021;14(4).
Ahmed F, Al-Khalifah B. I. A, Ahmad M. F, Alhodieb F. S. Cissus quadrangularis Aqueous Extract Attenuates Angiotensin
II-Induced Hypertension in Urethane-Anesthetized Rats. Biosc.Biotech.Res.Comm. 2021;14(4). Available from: <a href=”https://bit.ly/3ECkHKi“>https://bit.ly/3ECkHKi</a>
Copyright © This is an Open Access Article distributed under the Terms of the Creative Commons Attribution License (CC-BY) https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use distribution and reproduction in any medium, sources the original author and sources are credited.
INTRODUCTION
Hypertension is a major cause of death from cardiovascular complications such stroke, chronic kidney disease, and congestive heart disease. Despite many pharmacotherapies and lifestyle changes, the treatment of hypertension is still being investigated and remains difficult, with blood pressure not remaining at a normal level in around half of hypertensive patients. Alternatively, researchers have led concerted efforts to explore novel therapeutics with hypotensive/anti-hypertensive properties from medicinal plants/herbs. As an outcome, some of these medicinal plants with acclaimed hypotensive/antihypertensive properties have been validated, while others have been dismissed (Etuk 2006; Al-Anbaki et al. 2021; Azizah et al. 2021; Verma et al. 2021).Cissus quadrangularis Linn. (Vitaceae), often known as “bone setter” in Ayurvedic medicine owing to its bone fracture healing properties, is a tropical perennial plant found in India, Sri Lanka, Malaysia, Java, and West Africa. It is a perennial climber with four wings internodes and a slender, fleshy fibrous, smooth stem (Sundaran et al. 2020; Kaur et al. 2021).
Aside from being used as a vegetable in India, it is also used as a folkloric medicine to treat menstruation irregularities, dyspepsia, flatulence, colic, convulsions, asthma, inflammation, infections, and obesity. A number of bioactive compounds, including flavonoids (quercetin, kaempferol, daidzein, and genistein), triterpenoids (friedelin, β-amyrin and 7-oxo-onocer-8-ene-3 β 21-α diol), stilbene derivatives (quadrangularin A, quadrangularin B, quadrangularin C, resveratrol piceatannol, pallidol, and parthenocissus), iridoids (6-O-[2,3-dimethoxy]-trans-cinnamoyl catalpol and 6-O-meta-methoxy-benzoyl catalpol, picroside and pallidol), phytosterols (β-sitosterol, β-sitosterol-O-b-D-glucoside and ketosetosterol), phenolic compounds and tannins have been isolated from the aerial parts of C. quadrangularis (Shah 2011; Sundaran et al. 2020; Zaki et al. 2020; Bafna et al. 2021; Kaur et al. 2021).
Various extracts and formulations of C. quadrangularis have been attributed with potential pharmacological effects including anti-osteoporotic, anti-obesity, antioxidant, antidiabetic, anti-nociceptive, anti-ulcer, analgesic and anti-inflammatory effects (Murthy et al. 2003; Shirwaikar et al. 2003; Jainu and Devi 2004; Jainu and Devi 2006a; Jainu and Devi 2006b; Oben et al. 2006; Rao et al. 2007; Mate et al. 2008; Lekshmi et al. 2015; Jain et al. 2020; Bafna et al. 2021; Kaur et al. 2021).
Despite the fact that researchers have investigated into several pharmacological effects of C. quadrangularis, some of which have even entered clinical trials, its potential use against hypertension has yet to be revealed. Thus, the potential of C. quadrangularis aqueous extract to modulate blood pressure and heart rate in urethane-anesthetized rats was investigated against angiotensin II-induced hypertension.
MATERIAL AND METHODS
C. quadrangularis aerial parts were collected from Mysore region in India, subsequently identified by Dr. Sharanappa and a reference specimen was retained in the laboratory. Normal saline, heparin and adrenaline were purchased from local pharmacy. Angiotensin II and urethane was procured from Sigma Aldrich, USA. All of the other chemicals and reagents used in the analysis were of the finest analytical purity.The sample was washed with running water to eliminate dirt before being spread out on large trays. The trays were placed in a shady area for 12 hours before being chopped into small pieces. The chopped pieces were then dried for 24 hours in a hot air oven set at 60°C. In a cyclonic laboratory blender, the dry material was pulverized until it passed through a 60-mesh screen.
The powder was kept in an airtight container in the refrigerator for subsequent use. C. quadrangularis extract (CQE) was prepared by mixing the powder with hot distilled water at 70°C (1:8 w/v) and extracting it using a mechanical shaker for 24 hours. The extract was then filtered, and the residue was re-extracted with hot water for 12 hours before being filtered again. Both filtrates were pooled and evaporated in a flash evaporator set at 60°C to yield CQE, which was stored in an airtight container in the refrigerator for future use.
The research ethics committee approved the study (RU/REC-001/1.2/1441-42), and standard animal experimentation protocols were followed. Male albino rats weighing 350±30g were kept separately in polyacrylic cages in an air-conditioned animal house maintained at 25±2°C and 55±5% relative humidity. The animals were fed a standard laboratory pellet diet and were given free access to potable water. The rats were randomly categorized into four groups, each with six animals and received following treatment intravenously at the time of experiment : Group 1 – Control: received 0.2 mL normal saline.Group 2 – CQE: received CQE (10 mg/kg) dissolved in 0.2 mL normal saline.Group 3 – Ang II: received angiotensin II (0.5 µg/kg) with 0.2 mL normal saline.Group 4 – Ang II+CQE: received angiotensin II (0.5 µg/kg) + CQE (10 mg/kg) dissolved in 0.2 mL normal saline.
The hypotensive/antihypertensive activity of C. quadrangularis extract was evaluated using the procedure reported by Mohebbati et al. (2020) with some modifications. An intraperitoneal injection of urethane (1.25 g/kg) was used to anesthetize the animals. The rat’s necks and inguinal regions were shaved using an electric shaver after the anaesthesia was confirmed. The jugular vein was surgically cannulated for medication delivery, while the left carotid artery got PE-50 tubing cannula to record cardiovascular data. The arterial cannula was connected to a blood pressure transducer (SS13L) and the venous cannula to a syringe using a three-way plastic stop cock and a stainless-steel needle at the end of the PE tubing. Before cannulation, both cannulae were prefilled with heparinized saline.
Systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial blood pressure (MABP), and heart rate (HR) were all constantly monitored by the system (BIOPAC Systems Inc, CA, USA). A decrease in SBP of 10-20 mm Hg was deemed significant hypotensive/antihypertensive activity. Data on different cardiac parameters (SBP, DBP, MABP and HR) were expressed as mean ± SD in all groups. The data was subjected to one-way ANOVA and Tukey’s post hoc tests using SPSS ver. 20.0 (IBM, USA) software. Values were considered significant at p≤0.05.
RESULTS AND DISCUSSION
Hypertension is a major risk cardiovascular risk factor in reducing people’s life expectancy and quality of life. Because the activation of the renin-angiotensin system (RAS) is thought to be a key precipitating factor in hypertension, medications that suppress the RAS may be therapeutically valuable (Chen et al. 2018). Because many medicinal plants have been shown to regulate the RAS system to varying levels (Etuk 2006), the current study explored into the hypotensive/antihypertensive potential of C. quadrangularis aqueous extract against angiotensin II-induced hypertension in urethane-anesthetized rats.
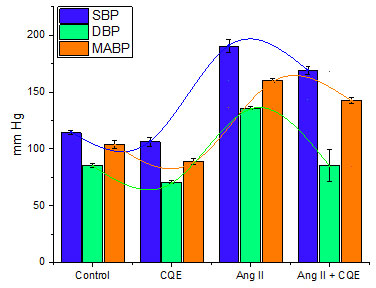
Table: 1 summarizes the data on systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial blood pressure (MABP), and heart rate (HR) of different groups after injecting designated treatments intravenously after cardiac parameters were stabilized and responses were monitored for 15 minutes.
Table 1. Effect of C. quadrangularis extract on cardiac parameters (SBP, DBP, MABP and HR)
| Groups | SBP | DBP | MABP | HR |
| Control | 114.4b ± 1.8 | 85.4b ± 1.7 | 104.0b ± 3.3 | 271b ± 0.7 |
| CQE | 106.1a ± 3.8 | 70.6a ± 1.4 | 88.9a ± 2.9 | 258a ± 0.6 |
| Ang II | 190.4d ± 5.6 | 136.0d ± 1.7 | 159.9d ± 1.7 | 269b ± 1.2 |
| Ang II + CQE | 168.9c ± 3.2 | 85.4c ± 13.9 | 142.8c ± 2.6 | 270b ± 2.2 |
*Values were represented as mean ± SD (n=6). Values with different superscript letters in columns differ significantly from each other at p ≤ 0.05.
In control group, wherein saline was injected did not elicit any significant changes in any of the cardiac parameters, whereas the injection of C. quadrangularis extract at a dosage of 10 mg/kg significantly (p<0.05) lowered SBP, DBP, MABP and heart rate to varying degrees in the CQE group. In group 3, conversely, angiotensin II (0.5 g/kg) injection resulted in a substantial rise in SBP, DBP, and MABP to the extent of 67, 59, and 54 percent, respectively, while heart rate remained unchanged compared to control group. CQE efficiently counteracted the blood pressure rising effect of angiotensin II in group 4 rats when CQE (10 mg/kg) was injected with angiotensin II (0.5 g/kg). CQE reduced SBP, DBP, and MABP by 12, 59, and 11%, respectively. It is worth noting that, while SBP was not brought down to baseline levels by CQE, DBP was, suggesting significant hypotensive/antihypertensive activity of CQE (Fig: 1).
Figure 1: Effect of C. quadrazsngularis extract on cardiac parameters (SBP, DBP and MABP)
Antihypertensive medicinal herbs usually modulate RAS by inhibiting angiotensin converting enzyme (ACE) activity or by modulating angiotensin II levels by inhibiting binding to AT1 and AT2 receptors (Mohebbati et al. 2020). Some investigations have found that antioxidant flavonoids like quercetin, triterpenes, anthocyanins and proanthocyanidins suppress RAS via reducing ACE and Ang II binding. As a result, natural products containing antioxidant compounds are thought to be beneficial against hypertension (Parichatikanond et al. 2012; Mohebbati et al. 2020). Various extracts of C. quadrangularis have been found to be efficient free radical quenchers, including nitric oxide (NO), superoxide anion, singlet oxygen, and hydroxyl radicals produced by the human body’s cells as a result of oxygen utilization (Murthy et al. 2003; Dhanasekaran 2020; Jain et al. 2020; Bafna et al. 2021; Kaur et al. 2021).
These extracts have been shown to contain a wide range of chemical compounds, including flavonoids (quercetin, kaempferol, daidzein, and genistein), triterpenoids (friedelin, β-amyrin and 7-oxo-onocer-8-ene-3 β 21-α diol), stilbene derivatives (quadrangularin A, quadrangularin B, quadrangularin C, resveratrol piceatannol, pallidol, and parthenocissus), iridoids (6-O-[2,3-dimethoxy]-trans-cinnamoyl catalpol and 6-O-meta-methoxy-benzoyl catalpol, picroside and pallidol), phytosterols (β-sitosterol, β-sitosterol-O-b-D-glucoside and ketosetosterol), phenolic compounds and tannins, the majority of which have been associated with significant antioxidant and antihypertensive activities (Amarowicz 2007; Wang et al. 2007; Frombaum et al. 2012; Rodrigo et al. 2012; Kucharska et al. 2017). As a reason, the hypotensive/antihypertensive effect of CQE can be attributed to the antioxidant compounds’ blocking of angiotensin II binding to angiotensin receptors.
CONCLUSION
The findings of the present study revealed that C. quadrangularis aqueous extract had strong hypotensive potential in normotensive rats and antihypertensive effect in angiotensin II-induced hypertension in urethane-anesthetized rats. Further research is required to determine the molecular mechanism of C. quadrangularis’ hypotensive/hypertensive action and to conduct clinical trials to establish its optimal use as an antihypertensive therapeutic.
Conflict of Interests: Authors declare no conflicts of interests to disclose.
Data Availability Statement: The database generated and /or analysed during the current study are not publicly available due to privacy, but are available from the corresponding author on reasonable request.
REFERENCES
Al-Anbaki, M., Cavin, A. L., Nogueira, R. C., et al. (2021). Hibiscus sabdariffa, a treatment for uncontrolled hypertension. Pilot comparative intervention. Plants, 10, 1018.
Amarowicz, R. (2007). Tannins: the new natural antioxidants? European Journal of Lipid Science and Technology, 109(6), 549-551.
Azizah, N., Halimah, E., Puspitasari, I. M., et al. (2012). Simultaneous use of herbal medicines and antihypertensive drugs among hypertensive patients in the community: A review. Journal of Multidisciplinary Healthcare, 14, 259-270.
Bafna, P. S., Patil, P. H., Maru, S. K., et al. (2021). Cissus quadrangularis L: A comprehensive multidisciplinary review. Journal of Ethnopharmacology, 279, 114355.
Chen, Y. J., Li, L. J., Tang, W. L., et al. (2018). First-line drugs inhibiting the renin angiotensin system versus other first-line antihypertensive drug classes for hypertension. Cochrane Database of Systematic Reviews, 11(11), CD008170.
Dhanasekaran, S. (2020). Phytochemical characteristics of aerial part of Cissus quadrangularis (L) and its in-vitro inhibitory activity against leukemic cells and antioxidant properties. Saudi Journal of Biological Sciences, 27(5), 1302–1309.
Etuk, E. U. (2006). A Review of medicinal plants with hypotensive or antihypertensive effects. Journal of Medical Sciences, 6(6), 894-900.
Frombaum, M., Clanche, S. L., Bonnefont-Rousselot, D., et al. (2012) Antioxidant effects of resveratrol and other stilbene derivatives on oxidative stress and *NO bioavailability: Potential benefits to cardiovascular diseases. Biochimie, 94(2), 269-276.
Jain, M., Nivedhitha, M. S., Deepak, S., et al. (2020). A novel natural product for bone regeneration in dentistry – a review. Journal of Evolution of Medical and Dental Sciences, 9(38), 2833-2838.
Jainu, M., and Devi, C. S. (2004). Effect of Cissus quadrangularis on gastric mucosal defensive factors in experimentally induced gastric ulcer- a comparative study with sucralfate. Journal of Medicinal Food, 7(3), 372-376.
Jainu, M., Mohan, V. K., and Devi, C. S. (2006a). Gastroprotective effect of Cissus quadrangularis extract in rats with experimentally induced ulcer. Indian Journal of Medical Research, 123(6), 799-806.
Jainu, M., Mohan, V. K., and Devi, C. S. (2006b). Protective effect of Cissus quadrangularis on neutrophil mediated tissue injury induced by aspirin in rats. Journal of Ethnopharmacology, 104(3), 302-305.
Kaur, J., Sharma, G., Mahajan, A., et al. (2021). Role of Cissus quadrangularis in the Management of Osteoporosis: An Overview. Critical Reviews in Therapeutic Drug Carrier Systems, 38(5), 27-51.
Kucharska, A. Z., Sokół-Łętowska, A., Oszmiański, J., et al. (2017). Iridoids, phenolic compounds and antioxidant activity of edible honeysuckle berries (Lonicera caerulea var. kamtschatica Sevast.). Molecules, 22(3), 405.
Lekshmi, R. K., Rajesh, R., and Mini, S. (2015). Ethyl acetate fraction of Cissus quadrangularis stem ameliorates hyperglycaemia-mediated oxidative stress and suppresses inflammatory response in nicotinamide/streptozotocin induced type 2 diabetic rats. Phytomedicine, 22(10), 952-960
Mate, G. S., Naikwade, N. S., Magdum, C. S., et al. (2008). Evaluation of anti-nociceptive activity of Cissus quadrangularis on albino mice. International Journal of Green Pharmacy, 2(2), 118-121.
Mohebbati, R., Kamkar-Del, Y., Kazemi, F., et al. (2020). Hypotensive effect of Centella asiatica L. extract in acute angiotensin II-induced hypertension. Iranian Journal of Pharmaceutical Sciences, 16(4), 71-80.
Murthy, K. N. C., Vanitha, A., Swami, M. M., et al. (2003). Antioxidant and antimicrobial activity of Cissus quadrangularis L. Journal of Medicinal Food, 6(2), 99-105.
Oben, J., Kuate, D., Agbor, G., et al. (2006). The use of a Cissus quadrangularis formulation in the management of weight loss and metabolic syndrome. Lipids in Health and Disease, 5, 24.
Parichatikanond, W., Pinthong, D., and Mangmool, S. (2012). Blockade of the renin-angiotensin system with delphinidin, cyanin, and quercetin. Planta Medica, 78(15), 1626-1632.
Rao, M. S., Kumar, P. B., Swamy, V. B. N., et al. (2007). Cissus quadrangularis plant extract enhances the development of cortical bone and trabeculae in the fetal femur: The metabolic syndrome. Pharmacologyonline, 13(3), 190-202.
Rodrigo, R., Gil, D., Miranda-Merchak, A., et al. (2012). Antihypertensive role of polyphenols. Advances in Clinical Chemistry, 58, 225-254.
Shah, U. (2011). Cissus quadrangularis L.: phytochemicals, traditional uses and pharmacological activities – a review. International Journal of Pharmacy and Pharmaceutical Sciences, 3(4), 41-44.
Shirwaikar, A., Khan, S., and Malini, S. (2003). Antiosteoporotic effect of ethanol extract of Cissus quadrangularis Linn. on ovariectomized rat. Journal of Ethnopharmacology 89(2-3), 245-250.
Sundaran, J., Begum, R., Vasanthi, M., et al. (2020). A short review on pharmacological activity of Cissus quadrangularis. Bioinformation, 16(8), 579-585 (2020)
Verma, T., Sinha, M., Bansal, N., et al. (2021). Plants used as antihypertensive. Natural Products and Bioprospecting, 11(2), 155-184.
Wang, T., Hicks, K. B., and Moreau, R. (2002). Antioxidant activity of phytosterols, oryzanol, and other phytosterol conjugates. Journal of the American Oil Chemists’ Society, 79(12), 1201-1206.
Zaki, S., Malathi, R., Latha, V., et al. (2020). A review on efficacy of Cissus quadrangularis in pharmacological mechanisms. International Journal of Clinical Microbiology and Biochemical Technology, 3, 49-53.


