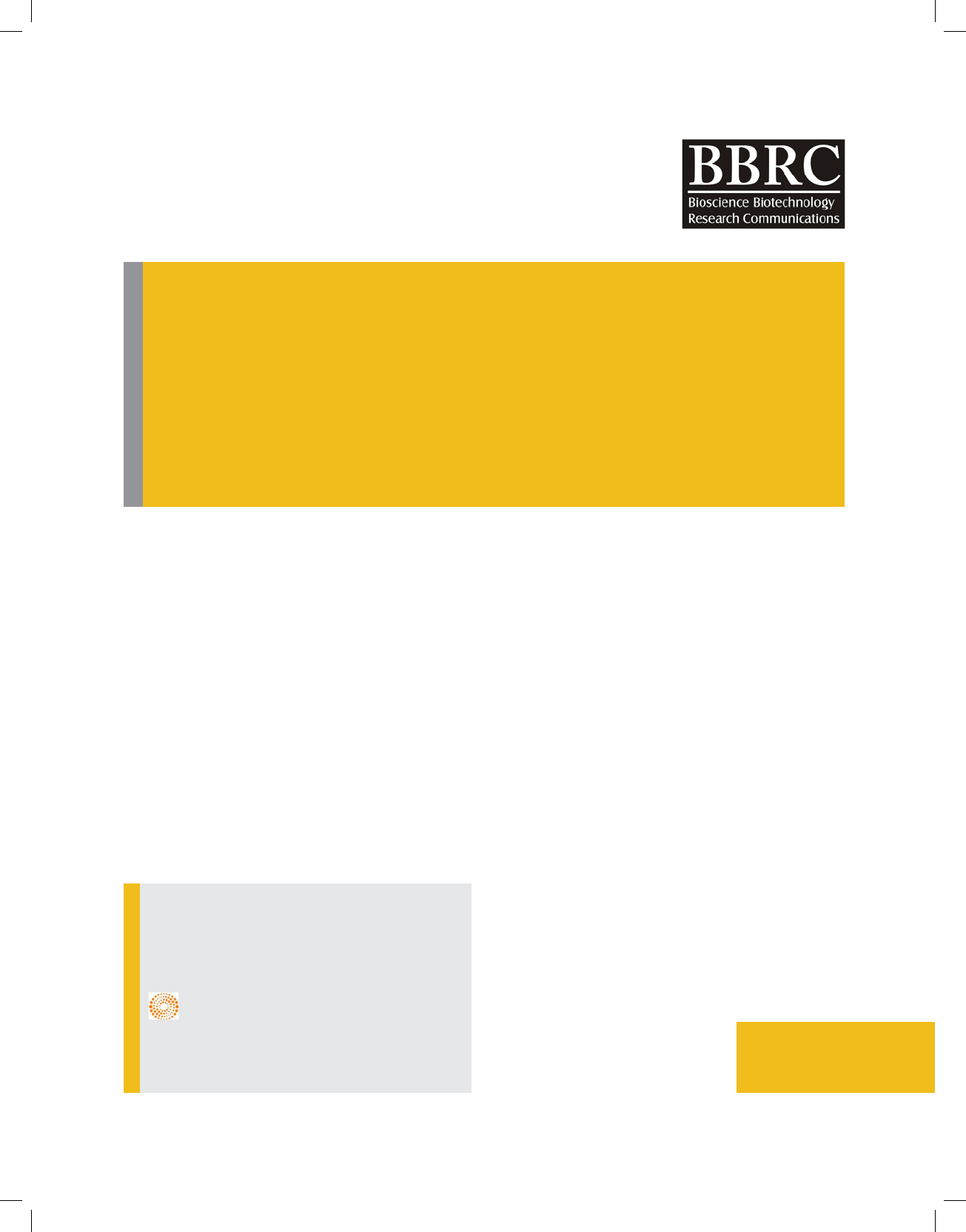
Medical
Communication
Biosci. Biotech. Res. Comm. 10(4): 797-801 (2017)
The production and evaluation of biologically synthesized
anticancer Chlorambucil - DTPA – Methionine
Elham Poonaki
1
, Leila Pishkar
2
, Mahdyeh Zarandooz
3
, Hadi Hejazinia
3
, Marzyeh Hosseini
4
,
Artin Assadi
3
, Mohammad Zahedi
3
and Shahin Hadadian*
5
1
Department of Biotechnology, I.A.U of Damghan, Damghan, Iran
2
Young Researchers and Elite Club, Islamshahr Branch, Islamic Azad University, Islamshahr, Iran
3
Department of Radiopharmacy, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
4
Department of Cellular and Molecular Biology, Islamic Azad University, Tehran North Branch,Tehran, Iran
5
Nanobiotechnology Department, Pasteur Institute of Iran, Tehran, Iran
ABSTRACT
Chromabucil is a direct acting direct acicular anticancer drug which is still widely used in the treatment of some
cancers as a primary treatment, but its use is often limited due to the unwanted side effects of this drug due to the
lack of speci city in targeting cancer cells. In this project, our effort to increase the speci city of Chlorambucil
using methionine amino acid has led to the production and evaluation of biological antimicrobial Nano conjugate
Chlorambucil-DTPA-Methionine. Research has shown that the consumption and harvesting of cancer cells increases
signi cantly over nitrogen and polyamide compounds, while some amino acids, including methionine amino acids,
are more expressed on the surface of the cancer cells, resulting in tissue Cancer cells increase to methionine amino
acid, so in our study, we have been using methionine amino acid as a carrier and enhancer of the uptake of Chloram-
bucil antimicrobial to produce this conjugate from the connector We used DTPA. After studying this Nano conjugate
and examining its structure, we investigated the therapeutic and biological effects of this nanoagonergic drug as
compared to Chlorambucil on the MCF-7 and HT-29 cell line (breast cancer and colon cancer), including The MTT
assay tests the determination of cell death and cell necrosis and a test for the determination of conjugate toxicity on
mice, which ultimately led to the understanding that the new nano conjugate Chlorambucil-DTPA-Methionine not
only retained its anti-cancer properties against the Chlorambucil drug But has shown less abnormal toxicity.
KEY WORDS: BREAST CANCER / CHLORAMBUCIL / METHIONINE / DTPA / CONJUGATE
797
ARTICLE INFORMATION:
*Corresponding Author: hadadian@yahoo.com
Received 10
th
Oct, 2017
Accepted after revision 16
th
Dec, 2017
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007 CODEN: USA BBRCBA
Thomson Reuters ISI ESC and Crossref Indexed Journal
NAAS Journal Score 2017: 4.31 Cosmos IF: 4.006
© A Society of Science and Nature Publication, 2017. All rights
reserved.
Online Contents Available at:
http//www.bbrc.in/
DOI: 10.21786/bbrc/10.4/27

798 THE PRODUCTION AND EVALUATION OF BIOLOGICALLY SYNTHESIZED ANTICANCER BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Elham Poonaki et al.
INTRODUCTION
Since cancer is a fatal illness worldwide, it is the sec-
ond leading cause of death in the world after car-
diovascular disease and, according to annual reports,
causes many deaths from cancer. Today’s treatments
are very costly and have unpleasant side effects in the
patient’s body. Today, efforts are being made to nd
newer and more effective treatments, including these
treatments, for chemical treatments. Nowadays, there
is a lot of research to discover new drugs, appropriate
drug delivery routes, and optimal drug targeting with
less side effects. Recently, therapeutic studies have been
conducted on the transmission of antibody-dependent
drugs, but since the therapeutic method of drug delivery
using antibodies also has inef ciencies and transitional
problems, costs and side effects, in particular, itself, a
newer research for the transfer of anticancer drugs is
taking place. Since then, research has shown that the
consumption and removal of cancer cells increases sig-
ni cantly over nitrogen and polyamide compounds, and
the expression of some amino acids such as methionine
amino acids on the surface of the cancer cells is more
pronounced and therefore cancer tissue cells to increase
the amino acid methionine, (Levine et al., 2000, Palmer
et al. 2009, Roché et al. 2011).
In this study we have used methionine amino acid
as carrier and antimicrobial agent for the treatment of
Chlorambucil. Cancer treatment as an anticancer drug,
like other anticancer drugs, has side effects on the can-
cerous patient’s body, the reason is that by designing
an anticancer drug conjugate using methionine amino
acid (as an enhancement of cellular uptake and DTPA
interface and conducting biologic studies of the effect
of this conjugate on cancer cells through drug delivery
to reduce side effects and Increase the ef cacy of Chlo-
rambucil to as an appropriate drug delivery method, we
tried to do that. So far, hybrids have been made of non-
interfacing Chlorambucil, such as Chlorambucil glucose
and Chlorambucil –tyrosine (Gupta A et al. 2010).
But the proper interface for ef cient drug delivery is
very important. We have been working to produce this
conjugate for the amino acid linkage of methionine We
used the DTPA connector for Chlorambucil amine, which
looks very good interface due to amine groups, because
amine receptors from cancer cells increase and drug
delivery will be more successful. We then performed the
relevant biological tests on the MCF-7 and HT-29 cell
line breast cancer cells.
MATERIAL AND METHODS
Following chemicals were used in the study.Chloram-
bucil (Sigma-Aldrich, USA) USA • Cell line MCF-7 and
HT-29 (Pasteue Institute of Tehran) • Sulfo-NHS (Sigma-
Aldrich, USA) USA) • EDC (Sigma-Aldrich, USA) • PBS
(Merck, Germany) • DMSO (Merck, Germany) • Sepha-
dex G-10 Fine (Sigma-Aldrich) • Chloroform (Merck,
Germany) • FCS (Seromed Biochrom, Germany) ) • FBS
(Merck, Germany) • RPMI medium (Sigma-Aldrich, USA)
• Penicillin powder (Sigma-Aldrich, USA) • Streptomy-
cin powder (Sigma-Aldrich, USA) • The TNF-alpha kit
(The RayBio® Human TNF -alpha ELISA) Kitin Annex-
inV-PI (BD Pharmingen, UK).
Combination or hybridization of an antimicrobial
agent of Chlorambucil and methionine amino acid,
produced by the interface of DTPA herein. 2.320 g of
methionine (if used 3 mg of Chlorambucil) is added in
5-5 milliliters of water Soluble. 2. Add 1 mg DTPA to
the solution and dissolve it by sterilizer. 3. After 1 to 2
minutes, add twice as much amino acid as EDC to the
previous solution. To the product of the rst stage after
1 to 5 minutes of production, 758 mg / kg of Chloram-
bucil is added. 2. To complete the Chlorambucil disso-
lution, dissolve it with sterilization. 3. The product is
ready for the next reaction. Use of DSMO is due to fat-
loving Chlorambucil (CBL) and DMSO helps to dissolve
it and accelerate the reaction. At this stage, puri cation
of the conjugated product is studied. The purity of the
compound produced was investigated by TLC chroma-
tography.
A 10 milliliter TLC solvent was prepared containing
3 milliliters of chloroform (nonpolar solvent to dissolve
lipophilic compounds) and 7 milliliters of methanol
(polar solvent to dissolve hydrophilic compounds) (30%
chloroform and 70% methanol) TLC papers were cut in
rectangles with dimensions, length 8-7 cm and width of
5-4 cm. Then 1 millimeter above the solvent line (the
highest part of the paper that is placed inside the sol-
vent), with the capillary tube, there were delicate spots
spaced apart, which included, respectively, methionine,
DTPA, Sulfur-NHS, conjugate synthesis They were Chlo-
rambucil and EDC. Each paper was placed in a solvent
containing a 45° angle inside the container and due to
the evaporation of chloroform and methanol, as well as
the toxicity of chloroform, it should be closed in a con-
tainer. The required amount of 25-26 C° and the required
time in this method is about 20 minutes. Be Due to the
fact that all materials and stains are colorless, UV light
was used to observe the movement rate of the material,
and stains were observed in the range of 280-220. The
patches created with DTPA, sulfur-NHS, EDC, and CBL
stain were a blurry and sharp spot. By this method, the
result was that the synthesis product, conjugate Chlo-
rambucil-methionine-DTPA, was present in the fourth to
seventh test tubes.
Mass Spectroscopy analysis was carried out to con-
rm methionine - DTPA- Chlorambucil
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS THE PRODUCTION AND EVALUATION OF BIOLOGICALLY SYNTHESIZED ANTICANCER 799
Elham Poonaki et al.
FIGURE 1. Mass Spectrum Display
Table 1. The values of Rf are calculated from the TLC paper from the distance that the
material goes through to the distance that the solvent travels.
E DCCBLCBL-Met DTPASulfo-NHSDTPAMethionineMaterial
88.032.040.049.082.072.0Rf
Conjugation. MTT is a colorimetric assay which is
used to assess cell viability based on metabolic activity.
This assay is based on reduction of yellow tetrazolium
salt (MTT) to form dark colored formazan dye by dehy-
drogenase enzymes in metabolically active cells the cells
(MCF7) and (HT-29) were brooded with different concen-
trations of methionine - DTPA- Chlorambucil (0.1, 0.2,
0.5 mg/ml and Chlorambucil 0.5 mg/ml and untreated
cell (as a negative control) for 48h.. After incubation
(48 h), 50 μl XTT detection solution was added to each
well of 96-well plate and the plate is kept in the incu-
bator for 2 h. The formazan dye is soluble in aqueous
solution and can be measured by evaluating the absorb-
ance at 450 nm using a spectrophotometer. The results
were compared to the untreated control cells. Statistical
data analysis was done using Prism and excels software
(Microsoft Of ce 2013). For quantitative data analysis
paired One Way ANOVA in case of cluster comparison,
were applied. P < 0.05 were considered statistically sig-
ni cant.
RESULTS AND DISCUSSION
In gel ltration chromatography, the solution was
removed from the chromatography column with 1 ml
(20 drops) in 20 separate test tubes. Then, each tube was
subjected to thin layer chromatography (TLC) to verify
the accuracy of the nal product. On the TLC paper, one
millimeter above the solvent line with a capillary tube,
delicate spots were separated from each other, which
consisted of methionine, DTPA, Sulfo-NHS, synthetic
conjugate, Chlorambucil-methionine- DTPA, Chloram-
bucil (CBL), and EDC. The stain was then investigated
by the UV lamp and the presence of the product in the
fourth to seventh tubes was determined, and then the
Rf of each compound was calculated according to the
formula given in Chapter 2, as follows (Table 1)
LC / MC mass spectrometry (LC / MC) spectroscopy
was used to study the molecular structure and iden-
tify the conjugated Chlorambucil -methionine DTPA.
The results are presented Figure 1.The 1983 molecular
weight spectrum shows well that the molecular weight
of 1983 is exactly equal to the conjugate molecular
weight, which is proof of conjugated synthesis. The syn-
tactic conjugate molecule is well visible
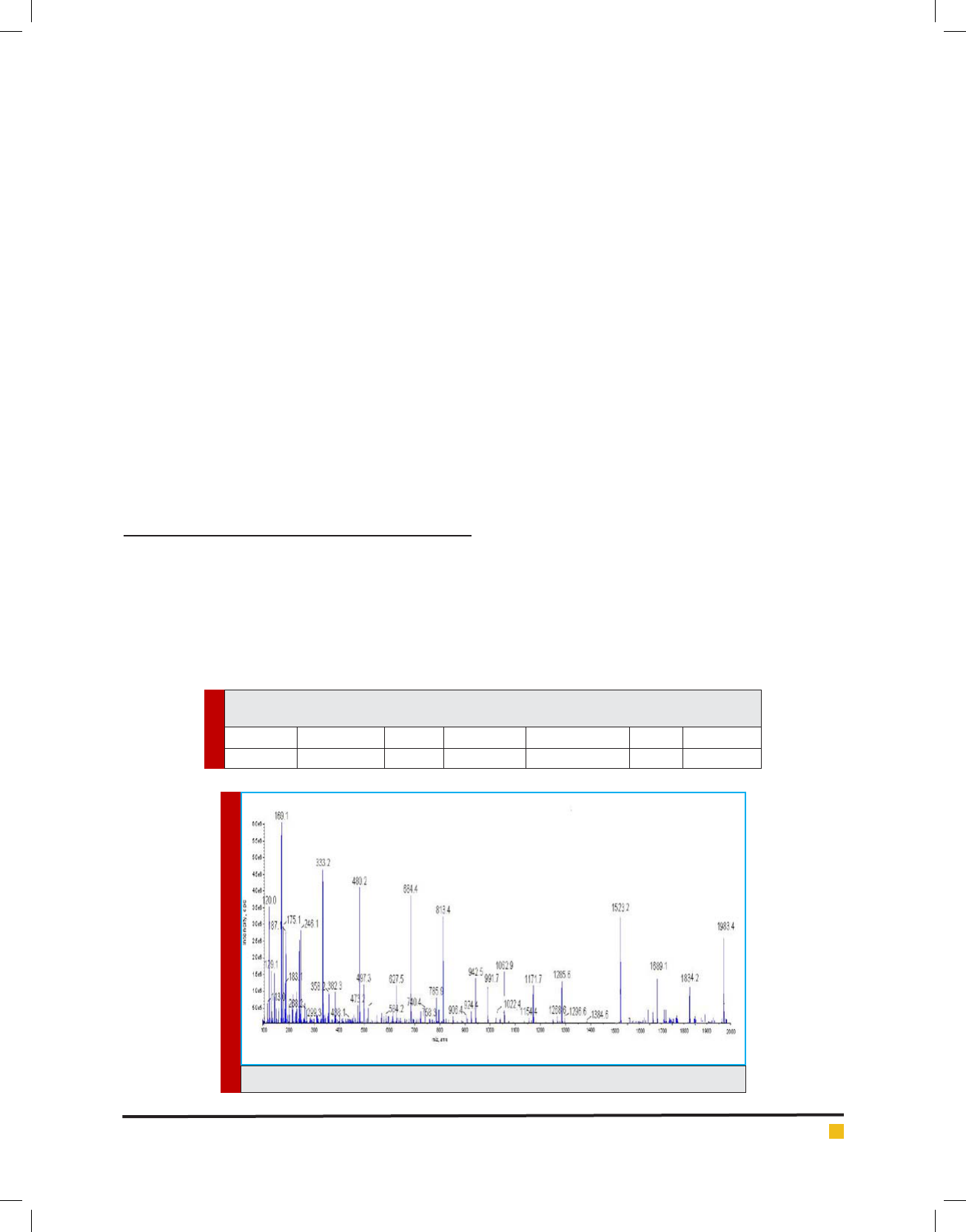
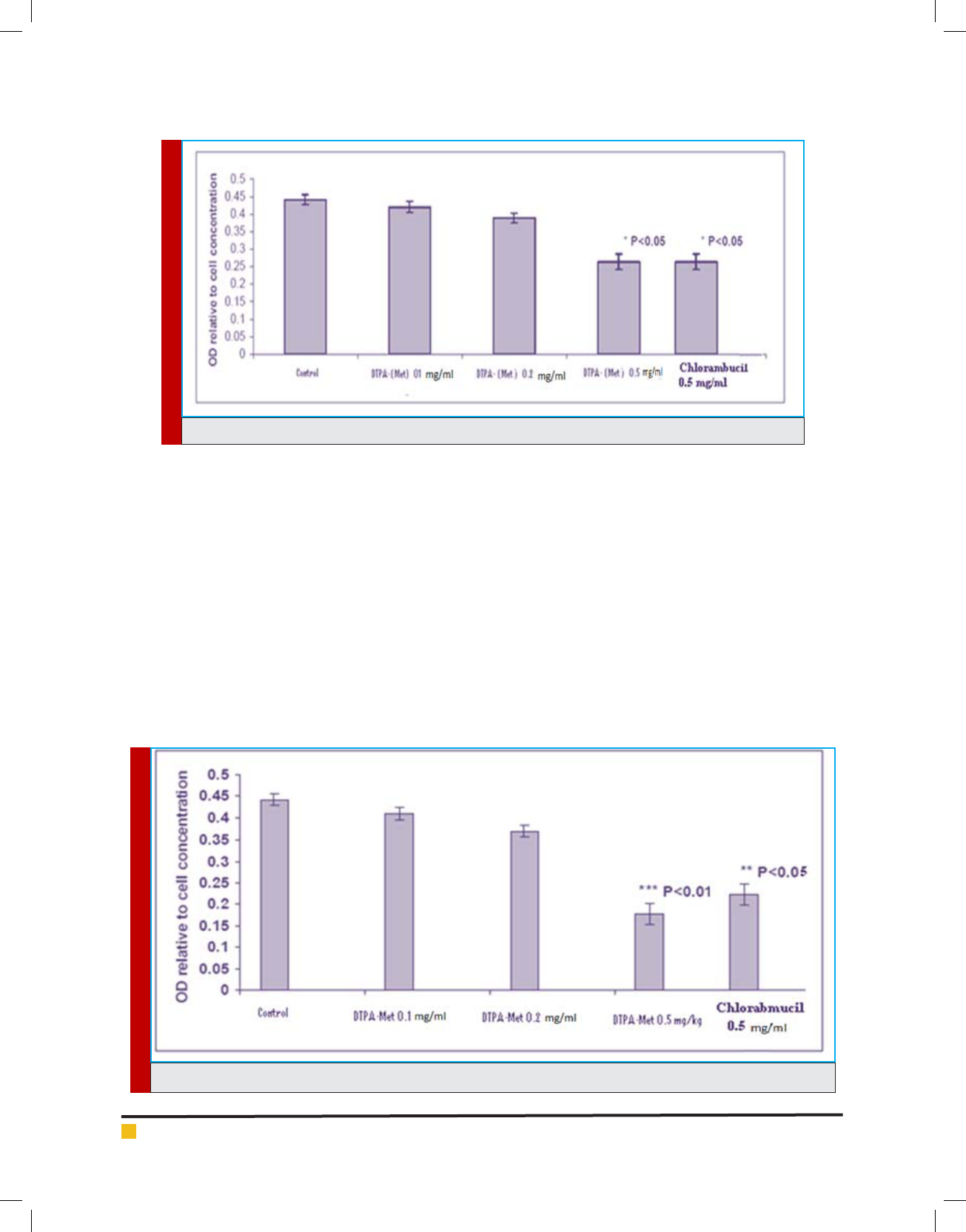
In Figure 2 and 3 the results of the compound toxicity
on the MCF-7 and HT-29 cell line respectively indicate
that our combination at 5 mg / ml dose has a signi -
cant toxicity (p < 0.05), and this toxicity is completely
equal to Chlorambucil. Making targeted drugs is a very
important part of cancer treatment. The use of non-can-
cerous anticancer drugs causes various side effects in the
patient. One of the oldest anti-cancer drugs is Chloram-
bucil, which belongs to the group of alkalizing drugs,
its effects on the cells are non-speci c and mechani-
cally unknown and have many side effects for patients.
The production of a molecular conjugate of Chloram-
bucil -methionine was performed with the goal of hav-
ing anticancer properties and also acting purposefully.
The ability to kill Conjugate Chlorambucil-Methionine-
Elham Poonaki et al.
800 THE PRODUCTION AND EVALUATION OF BIOLOGICALLY SYNTHESIZED ANTICANCER BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
FIGURE 2. Showing the results of the MTT assay test on the MCF-7 cell line
FIGURE 3. Showing the results of the MTT assay test on the HT-29 cell line
DTPA in similar doses compared to Chlorambucil on the
HT-29 and MCF-7 cell line was higher.
According to the results of experiments on conju-
gate and its comparison with Chlorambucil, the results
were that: the binding of methionine amino acid to
Chlorambucil not only does not eliminate the antican-
cer effect of this drug, but also increases its antican-
cer effect and increases the conjugated effect Because
cancer cells tend to receive high amounts of polyam-
ine compounds, the binding of the methionine amino
acid to Chlorambucil will result in a targeted formu-
lation that can absorb cancer cells of this conjugate.
In 2004, according to Bothenichen’s research on polyam-
ine and cancer, it was discovered that the accumulation
of polyamide compounds in cancerous tissues increased,
as well as the concentration of these compounds in
the body uids of patients with cancer, (Buchrach
et al. 2004) and in 2005 researchers it was found that
the transfer of some essential amino acids in cancer
cells increases, because in cells with high metabolism,
the absorption of amino acids also increases. There-
fore, using drugs reduces the synthesis of polyamine or
inhibits the carriers of these amino acids, it can inhibit
cancer, and these drugs can be a good option for can-
cer treatment (Yoon et al. 2005). In 2009, Palmer and
colleagues in their research stated that increasing the
concentration of polyamines plays an important role
in the development of cancer, from the onset to the
maintenance of the phenotype of the transformation,
(Palmer et al. 2009).

Elham Poonaki et al.
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS THE PRODUCTION AND EVALUATION OF BIOLOGICALLY SYNTHESIZED ANTICANCER 801
One of the ways of cancer cells to increase the con-
centration of polyamine compounds is the absorp-
tion of polyamines by the polyamine (PTS) trans-
fer system in these cells. PTS is a rugged system that
transmits a large amount of polyamines. Therefore, it
may be possible to transfer cytotoxic drugs by bind-
ing to polyamine vesicles and using this system to
target cancer cells selectively (Yoon et al. 2005).
In 1997, the dependence of the growth of a num-
ber of solid tumors on high levels of methionine and
the increased need for tumor cells to methionine were
observed in relation to normal cells, which suggests
that methionine can be considered as an appropri-
ate choice for tumor treatment. (Buchrach et al. 2004).
Conjugate Chlorambucil -methionine DTPA-D, due to
its high chemical content, is considered as a polyamide
compound and also has a methionine-like acid compo-
nent in its structure, which is due to two reasons for
its absorption in cancer cells. So far, much research has
been done to transfer targeted drugs through speci c
molecules to tumors. The use of transfusion carriers and
the manufacture of drug conjugates can have a more
effective and speci c effect on drugs. Due to the high
side effects of Chlorambucil anticancer drugs and its
nonspeci c effects on the cells of the body, the purpose
of this drug and its side effects have been many studies
on Chlorambucil conjugation.
In 2010, they used Chlorambucil-estradiol for chemo-
therapy for breast cancer and found that they had bet-
ter and better ef cacy compared to Chlorambucil (Gupta
et al. 2010). In 2011, researchers, given the frequent
expression of receptors Folate (FRS) at the level of the
malignant tumor cells synthesized two new conjugated
folate carrying Chlorambucil, and they were biologically
evaluated on the leukemia cell line. The results of the
antitumor activity showed more conjugates compared
to non-conjugated Chlorambucil (Guaragna et al. 2011).
By reviewing the articles, it was concluded that the
synthesized conjugates of Chlorambucil had a posi-
tive effect and their effectiveness was much better than
that of Chlorambucil, and these considerations made
the conjugated Chlorambucil -DTPA-methionine II as a
novel drug in The treatment of breast cancer has also
been strengthened. Also, according to the aforemen-
tioned articles, the synthesis of conjugate Chlorambucil
and the study of binding methods led to the conclusion
that the binding of Chlorambucil to amino acids was
carried out from its carboxyl side, not from the chlo-
rine region. Because chlorine is a very important factor
for killing cells by alkylating them. The carboxyl side
of the Chlorambucil has a pharmacokinetic aspect (no
dynamic aspect). Through this position, the drug enters
the body, it traverses and excretes a pathway. Therefore,
the manipulation and binding of the amino acid in this
region, with the exception of the effect on absorption
and its shelf-life in the body will not have an effect on
inhibiting the effectiveness of the medicine.
ACKNOWLEDGMENTS
The authors would to thank Tehran University of Medi-
cal Sciences as well as all laboratories and technicians
who provided supported during within the course of the
study.
CONFLICT OF INTEREST
The authors declared no con ict of interest.
REFERENCES
Buchrach U. Polyamines and cancer: Mini review article.
(2004) Amino Acids. 26(4), 307-9
Gupta A, Saha P, Descôteaux C, Leblanc, Asselin É, Bérubé
G. (2010) Design, synthesis and biological evaluation of estra-
diol-chlorambucil hybrids as anticancer agents. Bioorganic &
Medicinal chemistry letters. 20(5), 1614-1618.
Gupta A, Saha P, Descôteaux C, Leblanc V, Asselin E, Bérubé G,
2010 Design, synthesis and biological evaluation of estradiol-
chlorambucil hybrids as anticancer agents. Bioorg Med Chem
Lett. 2010 Mar 1;20(5):1614-8.
Levine RL, Moskovitz J, Stadtman ER. (2000) Oxidation of
methionine in proteins: roles in antioxidant defense and cel-
lular regulation. IUBMB Life. 50, 301-7.
Palmer AJ, Ghani RA, Kaur N, Phanstiel O, Wallace HM. (2009)
A putrescine-anthracene conjugate: a paradigm for selective
drug delivery. Biochem J. 424(3), 431-8.
Roché H, Vahdat LT, (2011) Treatment of metastatic breast can-
cer: second line and beyond. Ann Oncol. 22(5), 1000-10.
Yoon JH, Kim IJ, Kim H, Kim HJ, Jeong MJ, Ahn SG, Kim SA,
Lee CH, Choi BK, Kim JK, Jung KY, Lee S, Kanai Y, Endou H,
Kim DK. (2005) Amino acid transport system L is differently
expressed in human normal oral keratinocytes and human oral
cancer cells. Cancer Lett. 222(2), 237-45.