Biochemical alterations due to carbaryl exposure in
glucose content of liver and alimentary canal of
Clarias batrachus
Parveen Qureshi
Department of Zoology, Sai a Science College, Bhopal, MP 462001 India
ABSTRACT
Pesticides provide useful tools to agriculturists and hygienist for crop protection and disease control. The use of pes-
ticides has undoubtedly increased the agricultural output, but on the other hand, they have also poisoned the aquatic
environment. Present study indicates that carbaryl is highly toxic to sh. It produces severe damage to the organs: ali-
mentary canal and liver, concerned with the digestion and absorption of glucose. Due to this, glucose metabolism was
signi cantly affected throughout the exposure period. Carbaryl intoxication reduced glucose content both in liver and
alimentary canal after an initial increase, generally more energy is needed to mitigate any stress conditions. This may be
obtained from glucose which is one of the most available sources of energy. All these changes in glucose content may
be due to cumulative effect of enzymes, hormones, and metabolic disturbance caused by the pesticide.
KEY WORDS: HYGIENIST, TOXIC, MITIGATE, BIOCHEMICAL, HEALTH
773
Short
Communication
Biosci. Biotech. Res. Comm. 11(4): 773-775 (2018)
INTRODUCTION
Pesticides provide useful tools to agriculturists and
hygienists for crop protection and disease control (occu-
pational Environmental Health 1997). The use of pesti-
cides has undoubtedly increased the agricultural output,
but on the hand they have also poisoned the aquatic
environment. Carabmates are comparatively of recent
development in the eld of pesticides. Carbaryl, which
was introduced in 1956 under the trade name “Sevin” is
the most widely, used carbamate today (Oluah and Aga-
tha 2014). The in uence of pesticides on the inhabitants
of the water systems may manifest itself. Both direct tox-
i cation (acute chronic toxicity) and indirectly (dimin-
ishing of the content of oxygen dissolved in water, a
change in the chemical composition of the water) (Singh
ARTICLE INFORMATION:
Received 23
rd
Aug, 2018
Accepted after revision 11
th
Dec, 2018
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007 CODEN: USA BBRCBA
Thomson Reuters ISI ESC / Clarivate Analytics USA
Mono of Clarivate Analytics and Crossref Indexed
Journal Mono of CR
NAAS Journal Score 2018: 4.31 SJIF 2017: 4.196
© A Society of Science and Nature Publication, Bhopal India
2018. All rights reserved.
Online Contents Available at: http//www.bbrc.in/
DOI: 10.21786/bbrc/11.4/30
Parveen Qureshi
774 BIOCHEMICAL ALTERATIONS DUE TO CARBARYL EXPOSURE BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
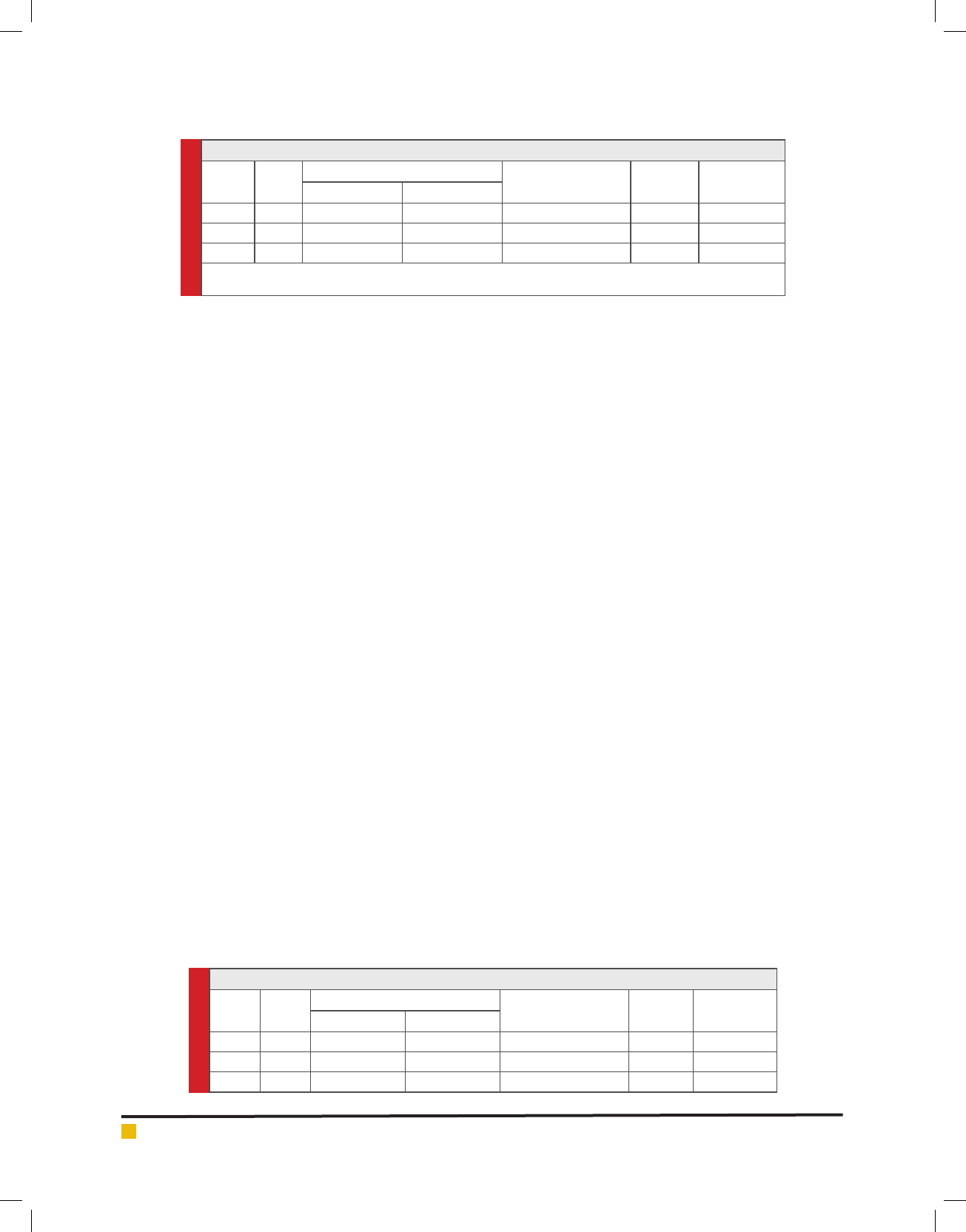
Table 1. Effect of Carbaryl (0.04 PPM) on glucose content in liver of Clarias batrachus
S. No. Time
Amount of glucose
% Age change
(increase/decrease)
‘t’ value Probability
Control (mg) Treated (mg)
1 7 5.068 ± 0.2051 7.282 ± 0.4090 43.685879 4.3276 ≤ 0.01
2 15 5.192 ± 0.2514 4.854 ± 0.3529 18.4024 2.3212 ≤ 0.02
3 30 5.212 ± 0.3457 3.254 ± 0.1959 37.5671 4.4164 ≤ 0.01
Value expressed as mg/100mg wet-weight of tissue
Each value is the mean ± standard error of 5 individual observations.
Table 2. Effect of Carbaryl (0.04PPM) on glucose content in alimentary canal of Clarias batrachus
S. No. Time
Amount of glucose
% Age change
(increase/decrease)
‘t’ value Probability
Control (mg) Treated (mg)
1 7 0.392± 0.032 0.541 ± 0.0448 37.500 -2.41921 ≤ 0.05
2 15 0.408 ± 0.0511 0.322 ± 0.026 -21.0784 1.2837 ≤0.10
3 30 0.411 ± 0.051 0.286 ± 0.0308 -28.713861 1.7247 ≤ 0.10
and Singh 2010). When pesticides pass over from water
into other links of biological chain, their content grows
hundreds and thousands of times. Besides observed by
ltering organisms, persistent poisonous chemicals may
be deposited in the tissues and then get into the organ-
ism of a sh in the interconnected links of food chains,
the action of the pesticides, being cumulative is ampli-
ed several times, (Omoniyi 2018).
The main stream of the earlier toxicological studies
on the pesticides on shes was con ned to histopatho-
logical eld. The effect of pollutants on tissue systems
of sh have shown to produce gross structural changes
such as atrophy, hyper trophy, necrosis, haemorrhage,
liqui cation, cytoplasmic vacuolation and degeneration
of blood vessels, (Mekkawy et al. 2016). Histological
changes and histopathological studies pressurized scien-
tists to go in for biochemical changes. Several scientists
have paid signi cant contribution in the histochemical
and biochemical eld (Ahmad et al. 2015). The present
attempt has been made to investigate the biological
change in the Clarias batrachus induced by sublethal
dose of carbaryl. The alimentary canals and liver were
selected for the present study because digestion, absorp-
tion and metabolism are cumulatively responsible for
energy production.
The amount of glucose in alimentary canal and liver
of Clarias batrachus was measured by Anthrone method
(Nicholas et al. 1956). About 100mg of tissue sample
was homogenized in 5 mL of chilled de-ionized water.
Then centrifuged at 5000rpm for 10min the nal volume
of supernatant was noted. Soon after the supernatant
collection, 1mL of 0.01% Sodium uoride solution was
added to supernatant to stop the conversion of glucose
to lactic acid. From the supernatant, 0.01mL was taken
as test sample. The volume in the test tube was made up
to 1mL with DDW. Then de-proteinization was done by
mixing 0.5mL of 1% H
2
SO
4
to 1mL of test sample. Then
4mL of anthrone reagent was added slowly with constant
stirring. The tubes were dipped in chilled water during
the mixing of anthrone reagent. The mixture was kept in
boiling water bath for 4min and then cooled. Develop-
ment of green color indicated the presence of glucose in
the supernatant. The intensity of the color was read at
on 540nm on spectrophotometer. Each experiment was
repeated 5times and the mean value with standard error
were calculated.
Carbohydrate, protein, lipid, enzymes and vitamins
are important component of the body and play a vital
role in the body construction, metabolism and detoxi-
cation. Therefore, in present investigation biochemi-
cal changes in glucose content have been studied in
liver and alimentary canal of control and intoxicated
Clarias batrachus on interval of 7-15-30 days. In the
present investigation an initial increase in glucose was
observed, which may be due to the greater absorption of
glucose by the intestine, under created stress conditions
and acceleration of glycogenolysis and gluconeogenesis,
which is similar to the ndings of Sharma et al. (2012).
These process synthesize glucose which is the major fuel
for energy production and energy demand of animal
increase to face the toxicity stress. Similar observations
were also made by (Singh and Singh 2017).
Besides glucose, they also observed increase of lac-
tate dehydrogenase (LDH) activity which elevates the
amount of lactic acid. These ndings are supported by
Michael (2018), as they have also reported an increase
LDH activity. Ahmad et al 2015 also reported the sig-
ni cant increased value in blood glucose and signi cant
decrease value in serum total protein level. Mekkawy
et al. (2016) also observed a decrease in LDH activity
which indicates pyruvate not dehydrogenated to yield
acetyl CoA and converted into lactic acid due to the ele-

Parveen Qureshi
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS BIOCHEMICAL ALTERATIONS DUE TO CARBARYL EXPOSURE 775
vation of LDH activity. The other cause of initial eleva-
tion in glucose level in alimentary canal may be due to
immediate increase of some digestive enzyme activity
which is responsible for the carbohydrate digestion. The
decrease in glucose level after 15
th
and 13
th
day of expo-
sure is probably due to the lesser secretions of hormones,
enzymes and inhibition of enzyme activity, which are
responsible for the carbohydrate digestion that’s why
the absorption of glucose is also decreased because de -
ciency of glucose content occur in the intestine. Present
ndings get con rmed with the ndings of Mahmoud
et al. (2013) as they have also observed depletion in glu-
cose content in the intestine of sh Clarias gariepinus,
after the exposure to mercury chloride. Effects of pesti-
cidal intoxication in intestine of different shes were also
reported by workers like Siakpere et al. (2011); Kumar
et al. (2011) and Ajani et al. (2018).
They have reported necrosis, vacuolation in mucosa,
complete degeneration in the serosa, muscularias sub-
mucosa and broken vili in the intestines. These changes
possibly deteriorate the secretion of enzyme in the gut
and absorption is also decreased due to damaged vili of
intestine. This is also supported by the nding of Ade-
woyeso (2010); Kumar and Banerjee (2016) as they have
observed depletion in glucose content in the sh Clarias
garipinus.
ACKNOWLEDGEMENTS
The authors are thankful to the Secretary and Principal,
Sai a Science College Bhopal for inspiring guidance and
valuable suggestions.
REFERENCES
Adewoye SO (2010) Haematological and biochemical changes
in Clarias gariepinus exposed to Trephosia vogelil extract.
Advances in Applied Science Research 1(1):74-79.
Ahmad A, Khan A, Sharma GD and Thoker Muddaassir (2015)
Biochemical changes induced by carbaryl carosulphone and
parathion on fresh water cat sh, Clarias batrachus. Interna-
tional Research Journal of Biological Science Volume 4 (8):4-9.
Ajani EK and Karim OK (2018) Haematological and immuno-
logical changes in the blood of African cat sh Clarias garie-
pinus reared under different sex combination. Hematologia
5:1-5.
Kumar R and Banerjee TK (2016) Arsenic induced haemato-
logical and biochemical responses in nutritionally important
cat sh, Clarias batrachus. Toxicology Reports 3:148-152.
Mahmoud M, Mekkawy A (2012) Biochemical response of the
African cat sh, Clarias gariepinus to sub lethal concentra-
tion of mercury chloride with supplementation of selenium
and vitamin E. Toxicology and Environmental Health Science,
4:218-234.
Mekkawy A, Mahmoud UM and Sara Salah (2017) Atrazine
induced changes in some biochemical parameter of the early
developmental stages of the African cat sh, Clarias gariepi-
nus. American Journal of Biochemistry and Molecular Biology
7 P. 21-40.
Olua N and Agatha C (2104) Alterations in the biochemical
parameters of the African cat sh Clarias Gariepinus to sub
lethal concentration of Lambda Cyhalothrian. Annals of Envi-
ronmental Science, 8:1-7.
Omoniyi Michael (2018) Toxicity effect of atrazine on histol-
ogy, haemotology and biochemical indices of Clarias garie-
pinus. International Journal of Fisheries and Aquatic Studies
Volume 6(3) P. 87-92.
Satheesh Kumar P. Ananthan G, Senthil Kumar D (2011) Hae-
matology and biochemical parameters of different feeding
behavior of teleost shes from Velar estuary, India. Comp Clin.
Pahtol, DOI 10-1007/00580-011-1259-7.
Sharma K, Prabhakaran K, Krishnam P and Grinsom G (2012)
Response of a fresh water air breathing sh Clarias batrachus
to salinity stress: an experimental case for their farming in
brackish water area in Andaman, India. Aquacult Int. DOI
10.1007/10499 - 012-9544 – 2.
Siakpere O, Ikomi RB and Ogbe MG Biochemical response of
the African cat sh Clarias garipepinus to sub lethal concentra-
tion of potassium permanganate. Annals of Biological Rsearch
2(2) P. 1-10.
Singh A and Singh A (2017) Studies on toxicity, stress, and
behavioral alteration and biochemical changes induced by
glyphosate herbicides on the fresh water sh Channa punc-
tatus. International Journal of Food and Agriculture Volume
7 (3) P. 39-48.
Singh A, Singh S, Bhartiya P and Yadav K (2010) Toxic effect of
phorate on the serum biochemical parameters of snake headed
sh Channa punctatus. Advances in Bioresearch 1,177-181.