
Toxicological
Communication
Biosci. Biotech. Res. Comm. 11(3): 486-495 (2018)
Evaluation of industrial ef uent and domestic sewage
genotoxicity using
Allium cepa
bioassay
Rajesh Paul and Jashodeb Arjun*
Zoology Department Lumbding College, Lumbding, 782447, Assam
ABSTRACT
Living organisms including both plants and animals being exposed to polluted water bodies can be impacted. In
current investigation the genotoxic effect of industrial ef uent and domestic sewage of Barak valley region, Assam,
India was investigated using both morphological and root chromosome assay on Allium cepa. The mean root lengths
of onions exposed to different concentrations of the ef uent and sewage (10%, 25% and 50%) were measured for 3
consecutive days for 24, 48 and 72 hrs and the results were compared. The mean root length was statistically evalu-
ated by the analysis of variance. There was both signi cant increase and decrease in root length among the exposed
onion bulbs. Total aberrations increased signi cantly as concentration increased (p<0.05). Both ef uent and sewage
samples were recorded to cause harmful damages in the exposed onion test samples. These results demonstrated that
the Allium test is a useful screening test for the evaluation of toxicity caused by sewage and ef uent samples not only
at the morphological level but also at the cytogenetic level; and hence pollution in waster bodies in a major cause
of concern. Thus, sincere measures should be undertaken regarding the direct disposable of industrial ef uents and
domestic sewage and protection of water bodies including its ora and fauna.
KEY WORDS: PAPER MILL EFFLUENTS, DOMESTIC SEWAGE,
ALLIUM CEPA
TEST
486
ARTICLE INFORMATION:
*Corresponding Author: jashodeb@gmail.com
Received 19
th
July, 2018
Accepted after revision 17
th
Sep, 2018
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007 CODEN: USA BBRCBA
Thomson Reuters ISI ESC / Clarivate Analytics USA and
Crossref Indexed Journal
NAAS Journal Score 2018: 4.31 SJIF 2017: 4.196
© A Society of Science and Nature Publication, Bhopal India
2018. All rights reserved.
Online Contents Available at: http//www.bbrc.in/
DOI: 10.21786/bbrc/11.3/19

Rajesh Paul and Jashodeb Arjun
INTRODUCTION
Over two third of Earth’s surface is covered by water;
less than a third is taken up by land. As Earth’s popula-
tion continues to grow, people are putting ever-increas-
ing pressure on the planet’s water resources .In a sense,
our oceans, rivers and other inland waters are being
squeezed by human activities, not so that they take up
less room, but so their quality is reduced. Poorer water
quality leads to pollution in water bodies. Water pol-
lution nowadays is a major global problem. It requires
ongoing evaluation and revision of water resource policy
at all levels. It has been recorded that water pollution is
the leading worldwide cause of death and diseases and it
accounts for the death of more than 14,000 people daily.
India and China are the two countries with high levels
of water pollution: An estimated 580 people in India die
of water pollution related illness including water borne
diseases. Water pollution results in contamination of
water bodies like rivers, lakes, aquifer, etc. due to human
activities, (Bennet, 1997, Prasad and Rao, 2010. Bakare
et al, 2017, Anacleto et al, 2017).
Polllution in water bodies occurs when contaminants
gets introduced into the natural environment. For exam-
ple, releasing inadequately treated waste water into
natural water bodies leading to degradation of aquatic
ecosystem. This in turn can cause public health prob-
lems for people living in downstream as people are using
this polluted water for regular domestic activities like
bathing, washing, drinking, irrigation, etc. Researches
have revealed that the sources of water pollution may
be point sources and non-point sources. Point sources
have an identi able cause as storm drain, waste water
treatment plant or streams. Non-point sources are more
diffuse like agricultural runoff, (Zaiad, 2010). With the
increasing development of industrial resources the risk
of water pollution has also increased. Not only the
industrial resources but also the inadequate system of
dumping of municipal sewage has resulted in pollution
of water bodies and its aquatic ecosystem including both
aquatic ora and fauna.
Researches till date have revealed that dissolved con-
taminants in both ef uents and sewages when exposed
to water bodies not only harms plant growth but also
forces plants to absorb dangerous chemicals and pollut-
ants which gets passed to animals and human through
consumption and other modes (Sik et al, 2009). In a
work done on the variable actions of Alium sepa and
its usage as a bio indicator of cadmium toxicity, plants
were exposed to increasing concentrations of cadmium,
where cadmium was observed to cause inhibition of root
and leaves growth and elongation which serve as a tool
for characterizing the bio indication of cadmium expo-
sure in waste and ef uent condition, (Bakare et al, 2017).
The direct application of industrial sludges were
recorded to harm the local biota in an investigation
where the genotoxicity of industrial sludges was assessed
using various plant including A.cepa where A.cepa test
was found to be effective in detection of damages (Ana-
cleto et al, 2017).
Studies about phytotoxic effects of waste waters and
ef uents started in 1970s where researches were con-
ducted taking sugar cane, eucalyptus, Triticum aestivum,
Brassica campastris, Sorghum, rice, and many more.
Allium cepa is the largest genus of petaloid monocotyle-
dons, containing hundreds of species naturally distributed
in temperate climates of the northern hemisphere (Koçyi
g
˘it & Özhatay, 2010). This test has important advantages
(Zegura, 2009) and has been used from many years in
investigating physical and chemical mutagenesis and
cytogenetic effects in mitotic cell division. Allium cepa
is important since it is an excellent model in-vivo, where
the roots grow in direct contact with the substance of
interest enabling possible damage to the DNA of eukary-
otes to be predicted. It is advantageous to use the Allium
cepa test system since its main component is a vascular
plant, making it an evaluating environmental pollutant ,
detecting mutagens (Gupta et al, 2009).
The present investigation was designed to exam-
ine the level of morphological and genotoxic damages
caused by industrial ef uents as well as domestic sew-
ages on Allium cepa so that proper safety measures can
be taken not only for the protection of water quality but
also preventive measures can be taken against the dam-
ages caused to aquatic ecosystem prior to exposure of
ef uents and sewage.
MATERIALS AND METHODS
For present investigation raw paper mill ef uent sample
was collected from the outlet pipes in the local river Barak
of the valley. The domestic sewage which was selected for
comparative analysis was collected from Silchar, Munici-
pal drainage system at Tarapur area, where all the debris
and discharges of the whole locality have been found to
be discharged. Both ef uent and sewage samples were
collected in plastic gallons, pH was measured and stored
at -20
o
C to prevent further microbial growth.
The common purple onion, Allium cepa (2n=16)
bulbs (1.5–2.0 cm diameter) used for this study was
procured from organic farmers of almost equal weight
age. The dried out scales were carefully removed leaving
the ring of the root primordial intact (Fiskesjo, 2011).
Then they were kept in moist condition to let root grow
for three days, this help select onion with synchronous
growth. For each test, 10 A. cepa bulbs purchased from
organic farmers were set up to produce roots in ltered
and dechlorinized tap water for three days and then
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY 487

Rajesh Paul and Jashodeb Arjun
transferred to the test solutions. Tap water was previ-
ously ltered in a bio-activated coal lter to remove
chlorine and its by-products commonly used for disin-
fecting drinking water. Three litres of water were aerated
over a period of 24 h before lling the test tubes. For
positive control mitomycin C was selected . Mitomycins
are a family of azinidines containing natural products
insulated from Streptomyces lavendulae. Mitomycin C
is a potent DNA crosslinkers. A single cross link per
genome has shown to be effective in killing bacteria.
This is accomplished by reductive activities followed by
2 N –Alkylation. Both alkylation are sequence speci c
for a guanine nucleotide sequence.
Three different concentrations of both the ef u-
ents and sewage were selected as 10%, 25% and 50%
for exposure through prior standardization. During the
Allium cepa assay, all selected onions were exposed
to the selected concentrations of ef uent and sewage
for 24hrs, 48hrs and 72 hrs, respectively. The growth
in roots were recorded till the third day of exposure in
water and after that the variation in root growth were
recorded after every 24 hrs for next three consecutive
days till 72hrs and the data were recorded and com-
pared. For mitotic studies, growth inhibition tests were
carried out for each sample, to nd its toxicity level.
After every 24hrs of exposure, 3 to 4 healthy root tips
from each bulb were prepared for the microscopic slides.
The emerged root tips of the onion bulbs in the dif-
ferent concentration of sewage and ef uent were xed
and macerated in a solution of 45% acetic acid (9 parts)
and 1 N HCl (1 part) at 50 °C for 10 min, followed by
squashing in 2% Acetocarmine stain for 15 minutes. The
modi ed conventional Feulgen-squash method (Sharma
and Dphil, 2012) was used to prepare permanent slides
of root meristems. The root tips were put in 1 normal
hydrochloric acid for ve minutes to soften the tissue.
The macerated and stained root tips were covered with
cover slip and squashed. Minimum 3-4 Slides were pre-
pared per bulb for microscopic observation. Approxi-
mately three thousand cells were examined per onion to
remove the errors and classi ed according to the chro-
mosomal aberrations presented including bridges, frag-
ments and chromosome lagging.
Results were presented as Mean±SE where mean
value was calculated from three individual readings of
a particular set. ANOVA was performed to determine the
level of signi cance from the set of onion bulbs. ANOVA
was done using graph pad PRISM (Graph pad Inc., san
Diego, CA, USA).
RESULTS AND DISCUSSION
Water pollution can be caused by a number of sources
ranging from industrial resources and sewage treatment
plants and factories to mining activities, paved roads
and agricultural runoff. Such issues have become one of
the biggest problems in many developing and developed
countries. These pollutants when not treated properly,
can cause mutagenic or toxic effects directly on humans,
affecting human health, resulting in diseases like cancer,
congenital malformations, and cardiovascular diseases
(Grover & Kauer, 2009). Siddiqui and his group (Siddiqui
et al., 2011) have worked to validate plant-based tests for
assessing the toxicity of water in India.
The Allium test is advantageous as genotoxicity
screening assay, as Allium root cells posses the mixed
function oxidase system which is capable of activat-
ing promutagens or genotoxic chemicals (Odeigah
et al; 1997a). In the Allium test, inhibition of rooting
and the appearance of stunted roots indicate retardation
of growth and genotoxicty, while root wilting explains
toxicity (Odeigah et al; 1997b). Both growth retarda-
tion and root wilting are accompanied by suppression
of mitotic activity and remarkable chromosomal aber-
ration. The present ndings provides evidence that
ef uent and sewage inhibited root growth and caused
growth retardation. The reason behind growth inhibition
may be due to high rate of chemical oxygen demand
which affected certain physiological processes leading
to the disturbance in the balance between promoter and
inhibitors of endogenous growth regulator (Gill and
Saggoo, 2010).
Growth inhibition was most recorded at 50% con-
centration along with a marked decrease in root length
when compared with the control. This is usually accom-
panied by an increase in chromosome aberrations (Amin
and Muzahid, 2009). The suppression of mitotic activ-
ity was often used in tracing cytotoxicity (Smaka-Kinel
et al., 2013). In our study a decrease in the mitotic index
was found as the concentration of ef uent increased
which indicates the cytotoxic effect of sewage and
industrial ef uent. Chrosomal aberration were observed
to increase as the concentration of ef uent and sew-
age increased. Among the chromosomal aberration
observed, IN, EN and CF were most frequent in all con-
centrations band kept on increasing from concentrations
of industrial ef uent towards higher concentrations of
sewage. Such ndings are responsible for the completely
decayed roots found in 25% and 50% concentration.
The most common abnormalities were c-mitosis and
disturbed metaphase. Sticky chromosomes and binucle-
ated cells were recorded in noticeable amount. In addi-
tion to the above, at anaphase and telophase bridges,
lagging chromosomes and irregular anaphase were also
observed. The mitotic index in the root meristems grown
in the negative control ranged from 17.3 to 19.8.
Table 2 shows the mitotic index values in root mer-
istems growth in different concentrations of ef uent
488 EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS

Rajesh Paul and Jashodeb Arjun
FIGURE 1. Panchgram Paper Mill
FIGURE 2. Silchar Municipal sewage
FIGURE 3. Sample of an onion bulb
showing the normal root growth
FIGURE 4. Organization of the experiment, with
Allium cepa where groups of 5 onion bulbs were
exposed for growth
Table 1. Root length variation of Allium cepa after cultivation in different concentrations of paper mill ef uent and
domestic sewage (10%,25% and 50%)
Treatment gropus Concentration
Root length in different time interval (mean±std.error)
Before treatment After treatment
24 hrs 48 hrs 72hrs 24 hrs 48hrs 72 hrs
Control --- 0.16±0.045 0.8±0.078 3.67±0.136 6.04±1.34 7.57±1.44 9.04±1.65
Positive Control (MMC) 2mg/lit 1.64±0.22 2.97±0.37 4.57±0.93 5.54±0.59 5.7 ±0.55 5.77±0.62
Paper Mill Ef uent
10% 0.26±0.075 1.1±0.129 4.44±0.062 5.03***±0.045 5.13***±0.045 6.1**±0.107
25% 0.2±0.068 1.23±0.091 4.06±0.39 5.05***±0.35 4.86***±0.349 5.36±0.286
50% 0.36±0.045 1.67±0.169 2.96±0.223 3.9***±0.223 4.53***±0.075 4.93±0.062
Domestic Sewage
10% 0.23±0.062 1.93±0.219 4.13±0.164 4.8***±0.165 5.3***±0.165 5.7±0.186
25% 0.13±0.068 1.23±0.091 4.33±0.169 4.9***±0.181 5.3***±0.181 5.66±0.198
50% 0.35±0.062 1.73±0.248 3.73±0.091 3.96***±0.075 4.3(((±0.029 4.5±0.029
Root length unit=cm; n=3.
and sewage from the three concentrations of wastewater
treatment. The decrease in the mitotic index of the root
tips reached statistical signi cance only in the highest
tested concentrations. The cytogenetic aberrations most
commonly observed in anaphase-telophase cells were
bridges, fragments and chromosome lagging. Aberrant
mitotic cells were counted and expressed as mean and
SD of the selected onion bulbs. In the root meristems
of the negative control, the percentage of aberrant cells
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY 489
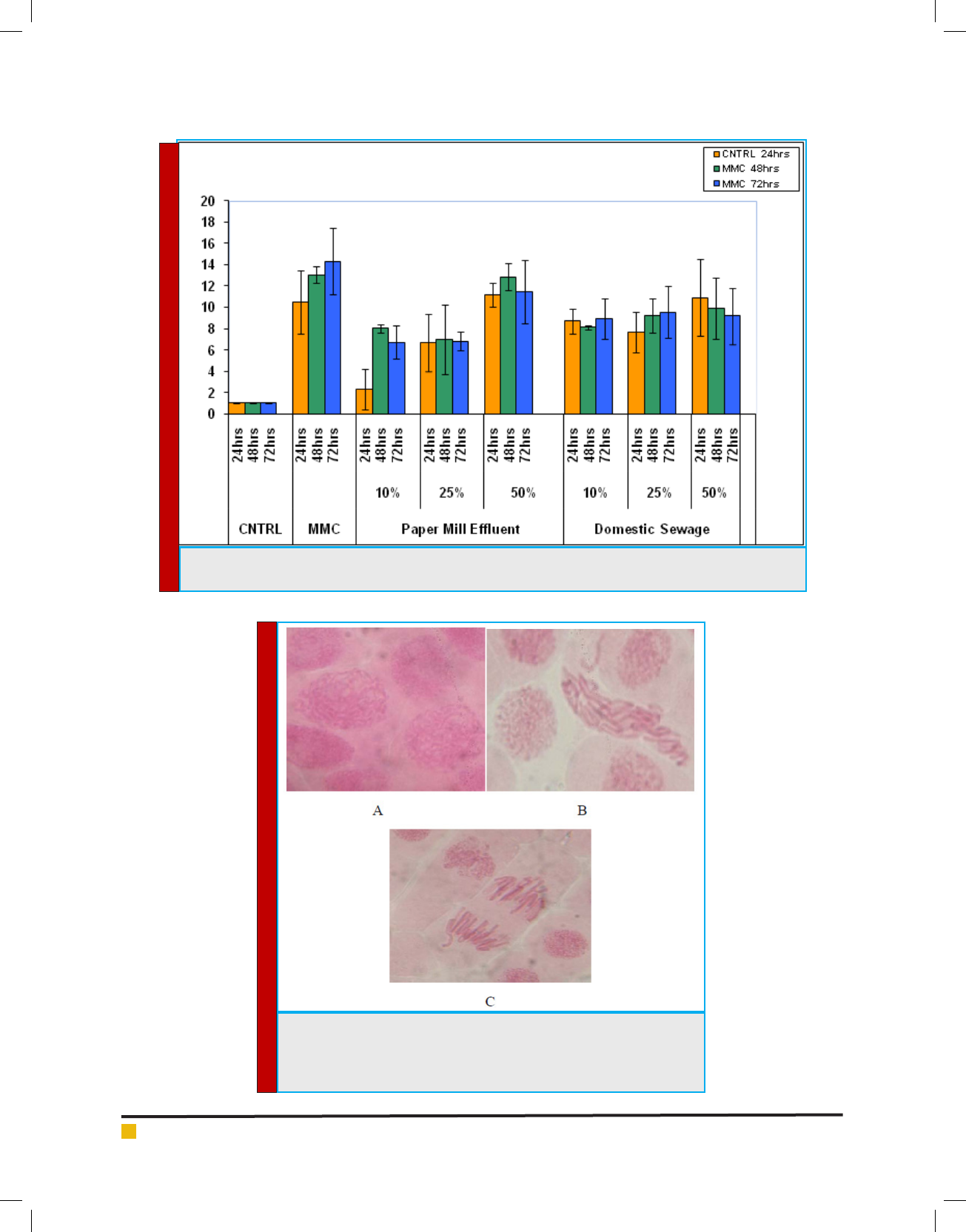
Rajesh Paul and Jashodeb Arjun
FIGURE 5. Graphical representation of various types of meiotic abnormalities after treatment with different
concentrations of Paper Mill Ef uent and Domestic Sewage.
FIGURE 5. Allium cepa cells in regular or normal division. A-Prophase =
the chromosomes are visible andtangled; B-Metaphase= the chromosomes
are arrangedin the equatorial plate; C-Anaphase = the sister-chromatids
separatemoving towards the spindle poles.
490 EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS

Rajesh Paul and Jashodeb Arjun
Table 2. Frequencies of different types of meiotic abnormalities after treatment with different concentrations of Paper Mill Ef uent and
Domestic Sewage.
Dose/Treatment Fixn.
time
Total no.
of cells
(n=3)
No. of
dividing
cells
MI
(mean±std.
deviation) MN
Cytotoxic effects Total
Aberrant
Cells
% Aberrant
Cells (mean±
std.error)
STK IN EN VC BNC CB CF
Contrlo
48h
72h
24h
3060 542
17.34±2.10
0 0 0 0 0 0 0 0 0 1.02±0.02
3060 610
19.66±1.96
0 0 0 0 0 0 0 0 0 1.03±0.05
3060 623
19.8±1.56
0 0 0 0 0 0 0 1 1 1.03±0.05
Positive control
(MMC)
48h
72h
24h 3080 248 8.04±0.44 15 22 85 15 3 2 5 22 323 10.48±2.98
3099 224
6.8±0.30
26 24 98 18 6 6 16 21 404 13.03±0.78
3119 246
7.87±0.28
33 28 101 22 8 8 23 18 447 14.33±5.08
Paper
Mill
Ef uent
10%
24h
3061 491
16.36±0.14
0 30 93*** 15 12 13 17 51 231 2.31**±1.86
48h 3083 450 14.57±3.48 0 19 133*** 6 4 3 11* 71*** 247 8.01***±2.42
72h 3075 472 15.33±2.19 0 16 156*** 9 10 4 9 69*** 273 6.73**±2.59
25%
24h
3061 429
14.03±1.08
0 16 80*** 20 15 10 17 46 204 6.72**±3.68
48h 3050 461 15.11±0.11 0 20 122*** 18 18 20 13 22 233 6.98**±3.24
72h
3020 456
15.09±0.22
0 17 85*** 20 20 21 22 21* 206 6.82**±0.86
50%
24h 3072 337 10.97±0.9 0 56*** 127*** 27 22 17 14 80 343 11.16***±4.13
48h
3067 408
13.29±0.84
3 59*** 156*** 39 25* 24 20 82*** 408 12.87***±2.27
72h 3033 437 14.42±1.4 3** 80*** 145*** 40 37*** 19 30 98*** 452 11.5***±6.98
Domestic
Sewage
10%
24h 3031 359 11.85±1.1 0 0 94*** 137*** 1 24 5 3 264 8.7**±1.2
48h
3051 436
14.29±1.49
0 0 92*** 110*** 1 36 6 3 248 8.12**±0.17
72h 3063 512 16.34±1.22 0 1 96*** 116*** 3 36 13 10 275 8.94**±1.89
25%
24h
3044 551
18.07±3.66
0 15 66* 66** 8 24 26* 34 239 7.65**±1.91
48h 3089
617
27.56±10.49
0 25 60* 66** 26 34 42*** 33 286 9.22***±1.64
72h 3051 652 21.38±1.28 0 30 67** 72** 32*** 30 24 34 289 9.55***±3.44
50%
24h
3023 431
14.25±0.58
2 13 72*** 115*** 22 44 33*** 30 331 10.91***±5.61
48h 3046 457 14.66±1.46 1 35* 55* 6**8 26* 31 42*** 43 301 9.88***±4.88
72h 3045 473 15.58±1.57 4 57*** 34 16 36*** 20 57*** 55* 279 9.19***±3.63
MN=Micronucleus, STK=Stickiness, IN=Irregular Nuclues, VC=Vagrant Chromosome, BNC=Bi Nucleated Cells, CB=Chromosome Bridge, CF=Chromosome Fragment.
Control : Dechlorinated tap water, MMC: Mitomycin C was used as positive control, When compared PME and DS with Control P<0.05=* P<0.01=**; P<0.001=***
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY 491
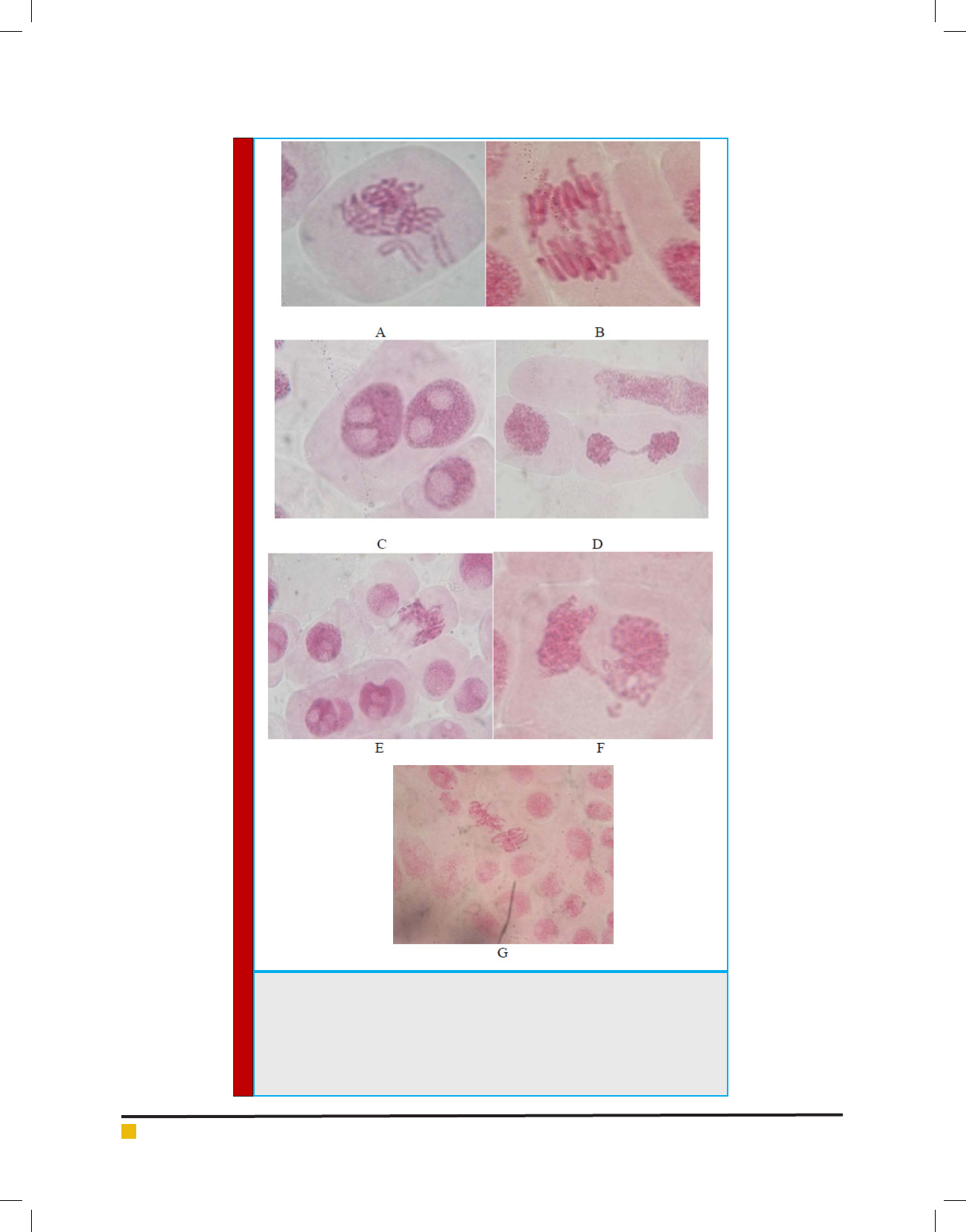
Rajesh Paul and Jashodeb Arjun
FIGURE 6. Allium cepa meristematic cells showing the alterations due to the action
of industrial ef uent and domestic sewage; A-irregular metaphase, with unor-
ganized chromosome, alsoknown as C-metaphase, showing chromosomes with no
orientation on the equatorial plate; B-irregular anaphase,with anaphasicmicro-
bridges; C-irregular cell, binucleate, with an elliptical aspect; D-telophase bridge;
E-cell with adherent or damaged nucleus, F-irregular cell; G-metaphase with
numerical alteration, due to duplication of the number of Chromosomes.
492 EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS

Rajesh Paul and Jashodeb Arjun
represents an avg. of 1.03 which was recorded as 8.34
and 10.09 in ef uent and sewages.
Results obtained in our study showed that sewage
and ef uent induced chromosomal aberration through
interactions with DNA and proteins leading to chromo-
some stickiness, mitotic disturbances and cell damage.
Present nding is similar to the results of Odeigah et al.
(1997b) in which roots of Allium cepa exposed to ef u-
ent from industrial wastes resulted to cells with chro-
mosomal aberration. It would be bene cial to apply A.
cepa chromosomal assay as a tool for monitoring the
genotoxic effects of industrial ef uent and wastewaters
thereby providing information on the need for environ-
mental managers to further subject treated industrial
ef uent to Toxicity Identi cation Evaluation and Tox-
icity Reduction Evaluation before they are nally dis-
charged. This will enable proper chemical analysis of
industrial ef uent in order to identify the constituent
that is really genotoxic and its prompt removal from the
ef uent before discharge.
CONCLUSION
According to the environmental campaign organization
WWF,” Pollution from toxic chemicals threatens life on
this planet. Every ocean and every continent, from the
tropics to the once-pristine polar regions, is contami-
nated. Clearly the problems associated with water pol-
lution can disrupt life on our planet to a great extent.
Increasing population and industrialization and direct
exposure of sewage in water bodies has resulted in pro-
duction of huge amount of liquid waste which ultimately
nds its way to water bodies like rivers, lakes, streams
and oceans. These ef uents and sewages if provided to
plants, animals and human for consumption, can lead
to spreading of different types of hazardous diseases.
Hence, it becomes mandatory to analyze the level of
pollution caused to water and its ora and fauna.
We can conclude from our investigation that Allium
cepa test is an excellent biomarker of chromosomal
aberrations and has occupied an important place for the
prevention and prediction of environmental impact that
will be caused by the exposure of ef uents and sew-
ages. The above ndings revealed the fact that industrial
ef uent and domestic sewage have almost equivalent
capabilities to cause harmful damages to the water bod-
ies. Life is ultimately about choices and so is pollution.
We can live with sewage strewn beaches, dead rivers,
and consume sh that are too poisonous for us to eat.
Or we can work together to keep the environment clean
so that plants, animals and people who depend on it
remains healthy. It is ultimately up to us to be informed,
responsible and involved when it comes to the problem
we face with our water. We must become familiar with
our water resources and learn about ways for disposing
harmful household wastes so that they do not end up in
sewage treatment plants that can’t handle them or land-
lls not designed to receive hazardous materials. Hence
proper measures should to be taken for the management
of water quality and the aquatic life.
ACKNOWLEDGEMENTS
We are grateful to UGC for providing fellowship to RP
and BR.
REFERENCES
A.A. Bakari, O.A. Alabi, A.M. Gbadebo, O.I. Ogunsuyi and C.G.
Alimba, (2017) In vivo cytogenotoxicity and oxidative stress
FIGURE 7. Photographs of onion bilbs captured after exposure to 50% concentration
of ef uent and sewage
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY 493

Rajesh Paul and Jashodeb Arjun
induced by electronic waste leachate and contaminated well
water Challenges, vol. 4, pp.169-187
A. Gill and M.I.S. Saggoo (2010) Mutagenic Potential and Nutri-
tive Quality of Turnip Plants Raised over Chromium Amended
Soils International Journal of Botany., vol . 6, pp. 127-131
A.H. Siddiqui, S. Tabrez, and M. Ahmed (2011) Validation of
Plant based bioassays for the toxicity testing of Indian waters,
Environ Monitor Assess., vol. 179,1-4, pp. 241-253, ISSN
1573-2959
A.K. Sharma, and A.S Dphil (2012) Chromosome techniques,
Fakenham press ltd, Norfolk. Pp. 95-100
A. Prasad, and K.V.B. Rao, (2010) Physico Chemical Analysis
of Textile Ef uent and Decolorization of Textile Azo Dye by
Bacillus endophyticus Strain VITABR 13, “The IIOAB Journal.,
vol. 2, pp. 55-62
A.S Margiassi, M.S. Goldbergs, A.J. Wheeler, C. Plante, M.F.
Valois, G. Mallach, (2014) Associations between perreneal expo-
sure to air pollutants and lung function tests and cardio vascu-
lar indices among children with asthma living near industrial
complex and petroleum re neries Environ. Res.,132, pp.38-45
A.W. Amin (2008) Cytotoxicity testing of sewage water treat-
ment using Allium cepa chromosome aberration assay Paki-
stan Journal of Biological Sciences. 5(2): pp.184 –188
Amin AW and M.M. Muzahid (2009) Cytogenetic effect of sea
water irrigation on irradiated wheat grains Egyptian Journal of
Genetics and Cytology. 29: pp. 199 –213
Zegura B F. Heath, A. C
ˇ
ernoša, M. Filipic (2009) Combination
of in vitro bioassays for the determination of cytotoxic and
genotoxic potential of wastewater, surface water and drinking
water samples Chemosphere, 75, PP. 1453–60
Michael C, and G.C. Odeigah (2009) The genotoxicity effect of
sewage ef uent on Allium cepa Report and Opinion., vol. 1(6),
pp. 36-41
Marin-Morales DM (2009) Allium cepa test in environmental
monitoring: a review on its application Mutat Res; 682: pp.
71–81
Schieltz ES A.A. Litomjua, E. Melen (2017) (Effects Of Long
term exposure to Traf c related Air Pollution on Lung function
in children Curr. Allergy Ashthma Rep. vol. 41: pp.1-13
Friskesjo G (2011) The Allium test, an alternative in environ-
mental studies; the relative toxicity of metal ions Mutation
Research. 197, pp. 243-280
Zaiad, GM (2010) Physico-chemical Analysis of Soils in Al-
Khums city, Libya Journal of Applied Sciences Research, vol.
6, pp. 924-929
Pink HD (2006) Heavy metals, Investing in Tomorrow’s Liquid
Gold, 3748, 2006.
Grover IS, and S. Kaur (2009) Genotoxicity of wastewater sam-
ples from sewage and industrial ef uent detected by the Allium
root anaphase aberration and micronucleus assays Mutat. Res.,
426, pp. 183–188, ISSN 0027-5107
Krishnamurthi K, S.S. Devi, J.G. Hengstler, M. Hermes, K. Kumar,
D. Dutta, S.M. Vannan, T.S. Subin, R.R. Yadav, T. Chakrabarti,
(2008) Archieves of Toxicilogy. , vol. 82, pp. 965-971
Souza LFB H.D. Laughinghouse, T. Pastori, M. Tedesco, A.W.
Kuhn, T.S. Canto-Dorow, S.B. Tedesco (2010) Genotoxicity
potential of aqueous extract of Artemisisa verlotorum on the
cell cycle of Allium cepa, Int. J. Environ Stud., vol. 67,6,pp.
871-877, ISSN 00207233
Anacleto, LR M.M.Roberto,M.A.Marin-Morales (2017) Toxi-
cological effects of the waste of the sugarcane industry, used
as agricultural fertilizer, on the test systemAllium cepa Chem-
osphere. vol.173, pp. 31-42
Sik, L O. Acar, and C. Aki, (2009) Genotoxic Effect of Industrial
Waste water on Allium cepa African Journal of Biotechnol-
ogy., vol. 8, pp. 1919-1923
Saavedera, MA and P. Arambari (1989) Use of sewage water
for irrigation of olive trees Cytologia. 54: pp. 489–492
Bennet MD (1997) Heterochromatin, aberrant endosperm
nuclei and grain shriveling in wheat-rye genotypes. Heredity.
39: pp. 411-418
Kataeva M , V. Kotseruba, N. Terekhina, N. Kutlunina, A. Bel-
jaeva (2010) Allium Root Micronuclues Test on the Genotoxic-
ity of Soil rosamples Contaminated with Heavy Metals World
Applied Science Journal., vol . 17, pp. 992-1000
Koçyigit M and N. Özhatay, A contribution to the genus Allium
L. (Sect. Codonoprasum) in Turkey. Turkish Journal of Botany,
34, pp. 391-395, 2010.
Kurt OK, J. Zhang, K.E. Pinkertan, (2016) Pulmonary health
effects of water pollution Pulm. Med., 22, pp. 138-43
Kolawole OM, and T.O. Sunmonu (2010) Effect of Wasterwa-
ter Treated with Metanolic Bark Extract of Bridelia ferruginea
Benth on Rat Kdiney and Liver, Journal of Applied Sciences in
Environmental Sciences., vol. 5, pp. 55-64
Gupta P, N. Mathur, P. Bhatnagar, P. Nagar, and S. Srivastava
(2009) Genotoxicity Evaluation of Hospitality Waste water
Ecotoxicology and Environment Safety, vol. 72, pp. 1925-32
Odeigah PGC, O. Nurudeen, O Amund (1997) Genotoxicity of
oil eld waste water in Nigeria Hereditas. 126, pp.161-167
Odeigah PGC, J. Makinwa, B. Lawal and R. Oyeniyi (1997b)
Genotoxicity screening of leachates from solid industrial waste
evaluated Allium test ATLA. 25, pp. 311-321
Mannucci, PM S. Harasi, I. Martinelli, M. Franchini (2015)
Effects on health of air pollution: A narrative review Intern.
Emerg. Med. Springer Milan, 10: pp. 657-662
Nirel R, N. Maimon, E. Fireman, S. Agami, A. Eyal, A. Peretz.
(2015) Respiratory Hospitalization of children living near a
hazardous industrial site adjusted for prevalent dust: A case
central study Int. Journal of Hyg. Environ. Health. 218: pp.
273-279
Alrumman, SA A.F. El-kott, M.A. Kehsk, “Water pollution:
Source and treatment”, American journal of Environmental
Engineering. 6(3), PP. 88-98, 2016.
Bibi S, R. L. Khan, R. Nazir (2016) Heavy metals in drinking
water of Lakki Marwat District, KPK, Pakistan World applied
sciences journal, 34(1), PP.15-19
Wong, SL A.L. Coates, T.To, (2016) Exposure to industrial air
pollutant emission and lung functionin children: Canadian
494 EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS

Rajesh Paul and Jashodeb Arjun
Health Measures Survey, 2007 to 2011. Health Reports. 27: pp.
3-9
Rajkumar S R, P. Velmurugan, K. Shanti, P.M. Ayyasamy, and
P. Lakshmanaperumalsamy (2004) Water Quality of Kodaika-
nal Lake, Tamilnadu in relation to physico-chemical and bac-
teriological characteristics Capital Publishing Company, Lake.,
pp. 339-346
Suriyanarayanan S., G. Jessen, L. Divya, and S. Balasubrama-
nian, (2012) Effec t of waste paper based paper industry ef u-
ents on the growth of tree seedlings, J. Environ. Res. Develop.,
vol. 7(2A), pp. 1117-1126
Oyeniyi T, C. Latunji (2012) Industrial Ef uent Induced Abnor-
mal Sperm Cells in mice (Mus musculus) New York Science
Journal vol. 5, pp. 60-64
Smaka-Kinel V, P. Stegner, M Lovka, and M. J, Toman (2013)
The evaluation of waste, surface and ground water qual-
ity using the Allium test procedure, Mutation Research. 368:
PP.177-179
Ahmed ZH , and S.N. Suhanija (2013) Physiological and
biochemical responses of Malaysian red alga, Gracilaria
manilaensis treated with copper, lead, mercury, J. Environ. Res.
Develop., vol.7(3), pp. 1246-1253
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EVALUATION OF INDUSTRIAL EFFLUENT AND DOMESTIC SEWAGE GENOTOXICITY 495