
Biotechnological
Communication
Biosci. Biotech. Res. Comm. 11(2): 238-245 (2018)
Effects of probiotic in expression of RUNX-2, ALP,
OCN and CASP-3 genes in Wistar albino rat receiving
2Gy gamma radiation
Omid Azadbakht
1
, Vahid Changizi
1
*, Elahe Motevaseli
2
, Sedighe Kolivand
3
and
Pegah Khanzadeh
1
1
Technology of Radiology and Radiotherapy Department, Allied Medical Sciences School, Tehran University of
Medical Sciences, Tehran, Iran
2
Molecular Medicine Department, Advanced Technologies in Medicine School, Tehran University of Medical
Sciences, Tehran, Iran
3
Medical Biotechnology Department, Advanced Technologies in Medicine School, Tehran University of
Medical Sciences School, Tehran, Iran
ABSTRACT
Today, radiotherapy is responsible for most of the therapies in a variety of cancers. However it could causes harm effects
like bone de ciency. Bone reinforces human body. On the other side probiotics are living microorganisms that have been
used in many studies to reduce the incidence of certain cancers or treat them. Therefore we decided to study if it is possible
to use probiotics against bone lost. Male albino Wistar rats for four weeks were put under a diet of two types of probi-
otic Lactobacillus casei and Lactobacillus acidophilus. After the diet, the groups were subjected to radiation with a cobalt
60 apparatus. Then the bone marrow immediately was extracted and examined the expression of the osteogenic genes
(RUNX-2, OCN, ALP) and CASPASE-3 (which is effective in apoptosis) by the Real Time PCR machine for the rst time .In
this study, we found that osteogenesis was much higher in groups with the probiotic diet than those without the probiotic
diet. Lactobacillus acidophilus was also found to be more effective than Lactobacillus casei. Also, the use of these probiot-
ics increases the expression of the CASPASE-3 gene in the common pathway of apoptosis, which means that probiotics
increase apoptosis. This study showed probiotics could repair the harmful effects of ionizing radiation on bones.
KEY WORDS: BONE MARROW, RADIATION, PROBIOTICS,
LACTOBACILLUS ACIDOPHILUS, LACTOBACILLUS CASEI
238
ARTICLE INFORMATION:
*Corresponding Author: changizi@sina.tums.ac.ir
Received 17
th
March, 2018
Accepted after revision 27
th
June, 2018
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007 CODEN: USA BBRCBA
Thomson Reuters ISI ESC / Clarivate Analytics USA and
Crossref Indexed Journal
NAAS Journal Score 2018: 4.31 SJIF 2017: 4.196
© A Society of Science and Nature Publication, Bhopal India
2018. All rights reserved.
Online Contents Available at: http//www.bbrc.in/
DOI: 10.21786/bbrc/11.1/7

Omid Azadbakht et al.
INTRODUCTION
All living things, including humans, are constantly
exposed to natural and human sources of ionizing radia-
tion. Effective dose of the background is about 2.4 mSv
per year. The major man-made origin of human expo-
sure is radio diagnosis and Radiation therapy for cancer
patients, (Wang, et al., 2016). Radiotherapy uses high-
energy rays to kill cancer cells. This may be done alone
or in mixture with other treatments such as surgery or
chemotherapy, (Spyropoulos, et al., 2011). Radiation
therapy is an important part of the treatment regimen in
various human malignancies, and for many non-treat-
able pain management. It is estimated that 50-70% of
all oncology patients are treated with radiation therapy,
or a combination of chemotherapy and radiation ther-
apy programs, (Michelin et al., 2004). To eliminate most
of the malignant tumors, ionizing radiation requires
approximate dose regimens near tolerance to adjacent
tissues. On the other hand tissues that contain renewal
cells, such as bone marrow and gastric mucosal mucosa,
require fast cell proliferation and there are more sus-
ceptible to toxic effects of ionizing, (Michelin, et al.,
2004). Bone damage in due to radiotherapy has been
con rmed in epidemiological and animal studies. Bone
is one of the most common natural tissues, and would
have harmful effects a like fractures and loss of bone
marrow function after radiotherapy, (Mego et al., 2013)
osteogenesis associated genes are, RUNX-2, ALP, OCN
and adipogenesis associated genes are PPAR- and C/
EBP, (Mansouri-Tehrani et al., 2015).
It should be noted that apoptosis is a cell death
mechanism with various physical and biological causes.
It plays a major role in many natural and physiologi-
cal processes, as well as in the pathogenesis of various
diseases, (Liu et al., 2013). Apoptosis can be activated
through the extrinsic and mitochondrial dependent
pathway. All paths eventually lead to caspase activation.
(Jilka, et al., 1998) on the other hands probiotics are liv-
ing microorganisms that help to preserve the bene cial
microbial balance in humans or other hosts as drugs or
dietary supplements. Most probiotics belong to a group
of lactic acid producing bacteria (Lactobacilli, Strep-
tococci, and Bi dobacterium). Some of the inhumane
strains are used in the fermentation of dairy products,
while others are human intestinal biochemistry, (Weiss
et al., 2011). Lactobacilli and Bi dobacterium are
generally known as probiotics due to their bene cial
effects on health and include various effects, such as
deprivation and inhibition of pathogens in the intes-
tine, increased integration of intestinal epithelium and
modulation of the host immune system both locally
and systemically, (Dobrzy
n
´
ska et al., 2015). So, based
on the above, we decided to use probiotics as a diet to
reduce the effects of radiation on the bone marrow and
reduce apoptosis.
MATERIAL AND METHODS
36 male Wistar rats with weighting of 220 ± 220 g were
purchased from the Tehran University of Medical Sciences
Pharmaceutical Research Center in 6 groups at the animal
house for four weeks prior to exposure to radiation. The
groups were kept in special cages under constant ambient
conditions at 22 ± 2 ° C and the light was adjusted for 12
hours of light and 12 hours of darkness. Water and spe-
cial food were provided to animals without restrictions,
except during tests. All experiments were conducted on
the basis of ethical standards for animal behavior.
THERE WERE SIX RATS IN EACH GROUP:
1. Non-radiation and non-probiotic group (control
group)
2. Non-radiation group with Lactobacillus casei pro-
biotics
3. Without radiation and with probiotic consumption
of Lactobacillus acidophilus
4. Group with irradiation and no probiotic consump-
tion
5. Radiation and probiotic Lactobacillus casei group
6. Radiation and probiotic group Lactobacillus aci-
dophilus
GAVAGE
0.1 g of each bacterium is equivalent to 10
10
CFU / g, the
amount of each serving was set for each group. Then for
each rat, the calculated amount of each drug was dis-
solved in one ml of PBS buffer (pH 7.2) and Daily, once
for Lactobacillus acidophilus and three times for Lacto-
bacillus casei, the calculated data was fed to a stomach
rat with a gavage needle. Also, control groups received
1 ml of PBS buffer per day.
IRRADIATION
The mice were anesthetized with ethical standards. At the
center of radiotherapy at Imam Khomeini Hospital in Teh-
ran, 60 cobalt irradiation devices were exposed to 2Gy and
a dose rate of 100 cGy /min in a eld with a size of 34.8
cm in 34.8 cm and an SSD of 80 cm were placed. Mice
were sacri ced by displacement of the neck. The animal
skeletal was washed in 70% ethanol. We discrete the mus-
cle and cut the two ends of the thigh bone. 10 ml syringe
by a 27-degree needle was injected from one end of the
thigh bone and was poured from the other end of thigh
bone into the test tube. Using centrifuge at 1000 rpm for
seven minutes, the solvent phase was discarded. These test
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EFFECTS OF PROBIOTIC IN EXPRESSION OF RUNX-2, ALP, OCN AND CASP-3 GENES 239

Omid Azadbakht et al.
tubes were placed in a freezer at a temperature of minus
80 (-80) degrees.
TOTAL RNA EXTRACTION
We extract a 5mm
3
piece of bone marrow tissue and
added 1 ml of RNA extraction solution and then homog-
enized the mixture with the homogenizer. 200 l of cold
chloroform was added to the solution and the tubes were
shaken vigorously for 15 seconds. Then the tubes were
incubated for 5 minutes in ice. The tubes were centri-
fuged for 15 min at 4 ° C and 12,000 rpm. After centrifu-
gation, three layers were formed in each vial from up
to down including the aqueous phase RNA, the protein
phase in the middle with white color, and the green phe-
nolic phase at the bottom. The upper phase was slowly
detached and transferred to a new 1.5-ml sterile tube.
Equilibrium solution was added to cold isopropanol and
incubated after mixing for 10 min at -20° C, then centri-
fuged for 15 min at 4 ° C and 12,000 rpm. To remove any
impurities, the super uous solution was discarded. The
RNA precipitate was rinsed gently with a milliteratanol;
after adding ethanol 80%, the tubes were slowly gutted
several times and then centrifuged for 10 min at 4 ˚C
and 12,000 rpm. The supernatant was removed slowly
and the sediment was placed for 10-15min at the labora-
tory temperature to dry. RNA deposition added to twenty
microliters of treated water with DEPC. The solution was
placed on a hot plate at 50-55 ° C for 5 minutes to solve
the RNA, then the tubes were kept in - 80 ° C freezer.
EVALUATE RNA QUALITY
To evaluate RNA quality, electrophoresis gel was used.
Before electrophoresis, all devices were treated with
DEPC water. Electrophoresis was performed in 1% aga-
rose gel. The gels were stained with ethidium bromide.
DETERMINE THE CONCENTRATION OF
EXTRACTED RNA
To determine the concentration of RNA, the BioTek
Nano Drop device was used. The OD= 260/280 expresses
the purity of the extracted RNA and has an inverse rela-
tionship with RNA contamination with the protein. The
aforementioned ratio close to 2 represents the absence of
contamination with the protein.
The ratio 260/230 was also used to check out the amount
of RNA contaminated with the materials used for extrac-
tion. It is desirable that this number is also close to 2.
SYNTHESIS OF CDNA
The master mix was made and added to each tube
(all the work was done on ice). One microgram of the
extracted RNA was used to synthesize cDNA. As a result,
the amount of the required RNA was calculated based on
its concentration in the sample and added to the tube.
The tubes were transmitted to the thermocycler, and the
cDNA synthesis reaction was performed according to the
following procedure.
Finally, the cDNA was maintained at -20 ° C.
PRIMER
Primers used in these experiments were designed and
tested using NCBI and Gene Runner software. By the
company Sinoclon with OD about 2 was made as freeze-
dried. The table 3 shows the characteristics of the prim-
ers used in this thesis.
REAL-TIME PCR STEPS
All ingredients were removed from the freezer and let
to melt gently. Master Mix was kept in aluminum foil to
protect it against light. According to the table 4, a mix-
ture of the desired materials was prepared to do real time
PCR with nal volume of 20 microliter. All reactions
were repeated twice. Special microcircuits were put in
a cold box with pins and the ingredients were added to
each of them according to the instructions below. All the
microtubules completely were sterilized to avoid error
in results. To control the contamination of the reaction
during the test, one sample without cDNA was consid-
ered for each gene, as a template called the NTC.
INGREDIENTS FOR REAL TIME PCR
The results of Real Time PCR were obtained from the
Corbett-6000 device.
Data analysis:
the results of the Real Time were analyzed through a
Fafel test. After analysis, one-way analysis of variance
Table 1. Materials for synthesis of cDNA
ingredientsQuantity
RT Buffer (x5)µl2
primer(50µM) oligo dTµl0.5
Primer:Random Hexamer(100µM)µl5/0
Reverse TranscriptaseEnzyme µl0.5
Sterilized water treated with DEPC
The nal volume is
10 L
Table 2. Thermosilocera device program for reverse
transcription reaction
timeTemperature (°c)The type of reaction
15 min37
Synthesis of single-
stranded cDNA
5 s85Enzyme inactivation
240 EFFECTS OF PROBIOTIC IN EXPRESSION OF RUNX-2, ALP, OCN AND CASP-3 GENES BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Omid Azadbakht et al.
Table 3. Characteristics of primers
ODlengthTmSequencesprimer
22259.2F:CAGACCTAGCAGACACCATGAG
OCN
22258.5R:GGACATGAAGGCTTTGTCAGAC
22059F:CGTTTTCACGTTTGGTGGCT
ALP
22058.9R:ACCGTCCACCACCTTGTAAC
22058.1F:GGCCACTTACCACAGAGCTA
RUNX-2
22058.3R:AGGCGGTCAGAGAACAAACT
22059.35F:AGCTGGACTGCGGTATTGAG
CASPASE-3
22057.3R:ATGGCGCAAAGTGACTGGAT
Table 4. Materials for Real Time PCR
MaterialsVolumeFinal concentration
Master Mix 2Xµl10X1
cDNAµl2ng/reaction100
M10forward primerl 8/0M4/0
M 10 reverse primer l 8/0M4/0
water RNase- Freeµl4/6-
(ANOVA) was performed. Because there was a meaning-
ful difference between the different groups, we set up
the Tukey HSD Post-hoc test and compared the different
groups.
RESULTS AND DISCUSSION
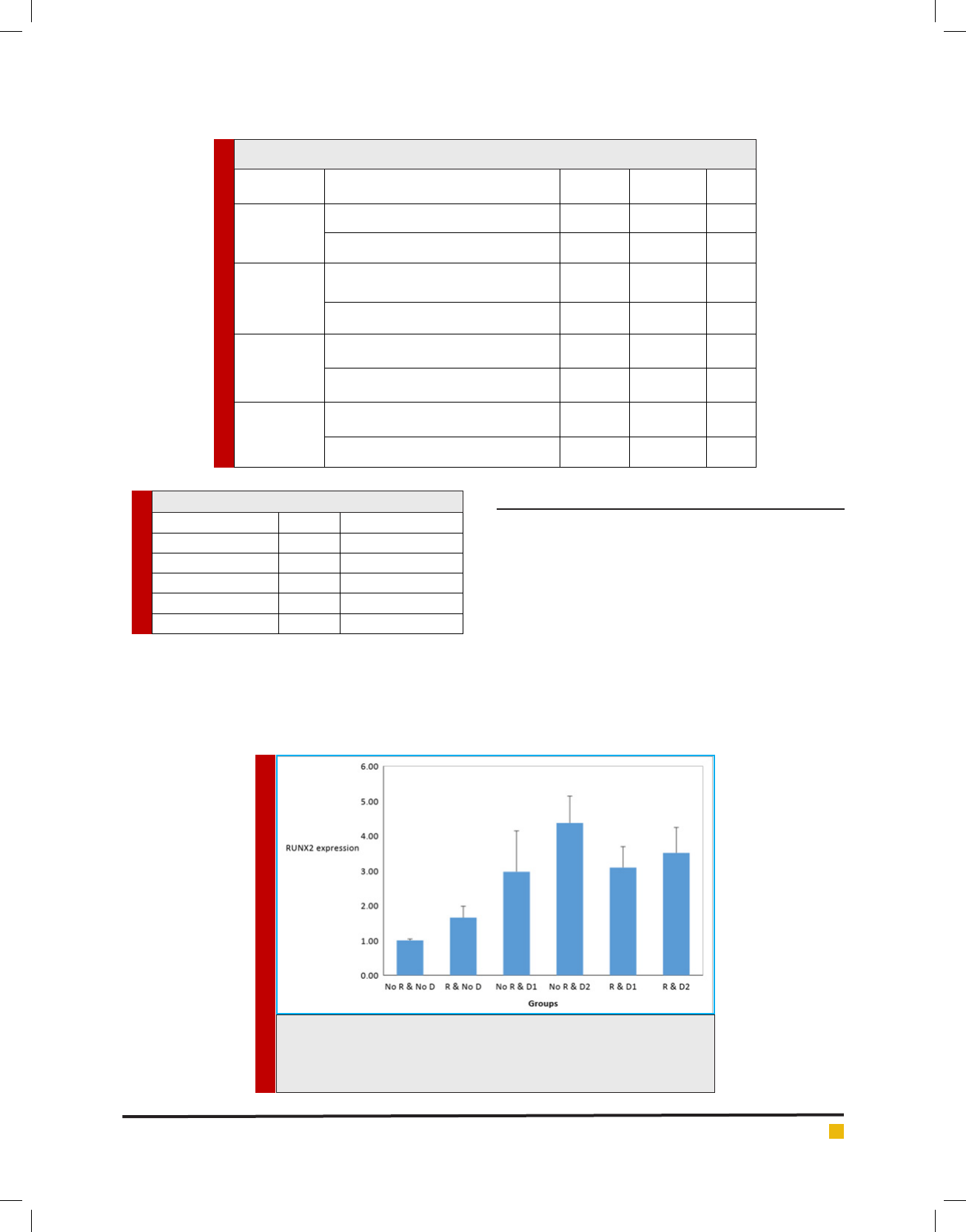
Data analysis results for RUNX-2 gene between different
research groups are shown in gure 1. It can be seen that
all the groups had a higher expression than the control
group ( rst group), while only the second group had no
signi cant difference compared to the control group.
In groups three, four, ve and six, there is a signi -
cant increase compared to the second group. The fourth
group is also the only group that has a signi cant differ-
ence compared to the third group, the difference is also
signi cant. The last signi cant difference was observed
in this chart is for groups ve and four, which expresses
the decline of group ve compared to group four. Data
FIGURE 1. The RUNX-2 gene expression chart in different groups is the
vertical axis of the gene expression and the horizontal axis of the tested
groups. R means radiation, D1 is probiotic Lactobacillus casei and D2 probi-
otic Lactobacillus acidophilus.
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EFFECTS OF PROBIOTIC IN EXPRESSION OF RUNX-2, ALP, OCN AND CASP-3 GENES 241

Omid Azadbakht et al.
FIGURE 2. The OCN gene expression chart in different groups is the vertical
axis of the gene expression and the horizontal axis of the tested groups. R
means radiation, D1 is probiotic Lactobacillus casei and D2 probiotic Lacto-
bacillus acidophilus.
FIGURE 3. The ALP gene expression chart in different groups is the vertical
axis of the gene expression and the horizontal axis of the tested groups. R
means radiation, D1 is probiotic Lactobacillus casei and D2 probiotic Lactoba-
cillus acidophilus.
analysis results for OCN gene between different research
groups are revealed in gure 2 .
Figure 2 shows a signi cant increase in the expres-
sion of the three, four, ve, and six groups relative to
group one. And all groups except group 1 have a signi -
cant increase compared to the second group.
Figure 3 shows data analysis for ALP gene between
different research groups.
The four, ve, and six groups showed a signi cant
increase compared to the control group in Figure 3. It
can also be seen that groups four and ve have a sig-
ni cant increase compared to the second group. The last
result indicated in this chart is the increase in ALP gene
expression in groups four, ve and six compared to the
third group.
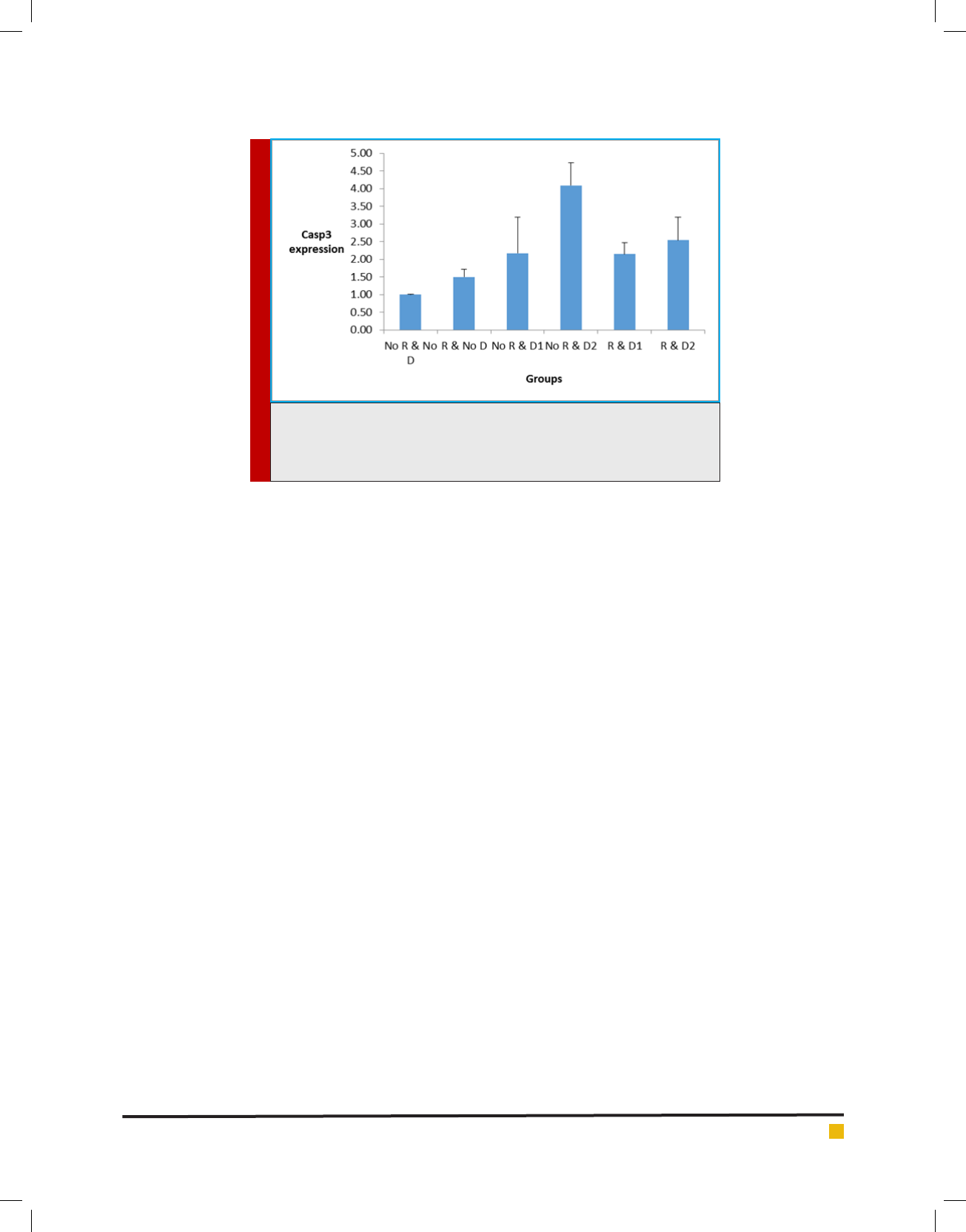
Figure 4 reveals data analysis for CASPASE-3 gene
between different research groups. Increasing the expres-
sion of groups 3, 4 , 5 and 6 was signi cantly different
from that of the control group. Fourth and sixth groups
also had a signi cant increase compared to the second
group. The fourth group is the only group that has sig-
ni cantly increased relative to the third group. The fth
and sixth groups also had a signi cant decrease com-
pared to the fourth group, which is visible on the chart.
In order to better understand the conclusions and con-
clusions about the results of the study, the mechanisms
of the effect of probiotics should be considered rst. The
mechanism of action of these probiotics includes the pro-
duction of inhibitor compounds, competition for binding
sites, competition for food, elimination of tox Enhances
242 EFFECTS OF PROBIOTIC IN EXPRESSION OF RUNX-2, ALP, OCN AND CASP-3 GENES BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Omid Azadbakht et al.
the immune system by increasing the level of cytokines,
immunoglobulins activating macrophages and mononu-
cleosis, and increasing the activity of natural killer cells,
self-immune modulation and the production of TNF
and interleukin 6.in receptors and ultimately boosting the
immune system. The tumor necrosis factor effect has three
pathways: one way reaches to caspase-3, and the second
can be terminated into three different functions, namely
pre-apoptosis, proliferation and cellular differentiation,
and the third pathway is the anti-in ammatory route .
Caspase-3:
All groups under the probiotic diet were more likely to
increase than the control group because of the mecha-
nism of probiotic effects on TNF alpha, which increases
the expression of CASPASE-3. Groups under the Lacto-
bacillus acidophilus probiotic diet, in the presence, and
in the absence of radiation, gave rise to the expres-
sion of the CASPASE-3 gene in comparison to the only
radiation group, since probiotics express the expres-
sion of CASPASE-3 in accordance with the above men-
tioned mechanism. Lactobacillus acidophilus group
has increased expression in lactose-bacillus casei in
the presence and absence of radiation, which probably
indicates a greater effect of Lactobacillus acidophilus
than Lactobacillus casei. The Lactobacillus acidophilus
group has a greater expression than the Lactobacil-
lus acidophilus group with radiation, which indicates
radiation and Lactobacillus acidophilus neutralize each
other. Perhaps radiation in the bone marrow may pro-
duce an immunological status that is more favorable
than the pathway of differentiation and proliferation
cells arrive.
RUNX-2:
All groups under the probiotic diet were more likely to
be exposed than the control group and the radiation
group, as probiotics increase the cellular differentiation
and multiply their pathways. The Lactobacillus acido-
philus group has been shown to have a higher expres-
sion than Lactobacillus casei (both in the presence and
in the absence of radiation), indicating a greater effect
of Lactobacillus acidophilus.
ALP:
Groups that have taken Lactobacillus acidophilus (both in
the presence of radiation and in the absence of radiation)
have a greater expression than the control group, which
is probably due to the effect of Lactobacillus acidophilus.
Lactobacillus casei group has a signi cant increase in
expression in comparison with the control group, prob-
ably due to the synergistic effect of the radiation and pro-
biotics. Because according to the radiation signal path and
the osteoblastic signal path, radiation can ultimately cause
cellular sensitivity so that the cell shows the counter-
effects of radiation. This issue of the effects of signals can
be investigated in the future. Groups that have taken Lac-
tobacillus acidophilus (both in the presence of radiation
and in the absence of it) have a higher expression than
the Lactobacillus casei group; it indicates the greater effect
of Lactobacillus acidophilus. The Lactobacillus casei group
(in the presence of radiation) has a greater expression than
Lactobacillus casei which is due to synergistic effect.
OCN:
All groups under the probiotic diet have increased
expression in both the control group and the radiation
FIGURE 4. The CASPASE-3 gene expression chart in different groups is the
vertical axis of the gene expression and the horizontal axis of the tested
groups. R means radiation, D1 is probiotic Lactobacillus casei and D2 probiotic
Lactobacillus acidophilus.
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EFFECTS OF PROBIOTIC IN EXPRESSION OF RUNX-2, ALP, OCN AND CASP-3 GENES 243

Omid Azadbakht et al.
group because probiotic activates the second alpha-TNF
pathway, resulting in differentiation and proliferation.
CONCLUSION
This study shows, the use of probiotics increase the
expression of osteogenic genes and apoptosis, except in
cases where this increase was not signi cantly expressed.
According to the results and analysis, probiotic Lactoba-
cillus acidophilus has a greater effect on Lactobacillus
casei. That is, this probiotic increased the expression of
the two groups of radiation and control. Although it also
expands the gene for apoptosis. In general, the use of
probiotics in this study increased the expression of oste-
ogenic genes and apoptosis, except in cases where this
increase was not signi cantly expressed. In some stud-
ies, we observed that radiation combined with two pro-
biotics has different effects of synergistic and inhibitory
effects for two probiotics, which is probably due to the
effect of various probiotics in their signal paths, which
is not exactly clear and can be tracked and researched.
ACKNOWLEDGMENTS
This study has been supported by Tehran University of
Medical Sciences.
Con ict of interest: There is no con ict of interest.
Funding: This study was funded by Tehran University of
Medical Sciences (grant number: 33517).
Ethical approval: All applicable international, national,
and/or institutional guidelines for the care and use of
animals were followed.
REFERENCES
Alwood, J. S., Shahnazari, M., Chicana, B., Schreurs, A.,
Kumar, A., Bartolini, A., Globus, R. K. (2015). Ionizing radia-
tion stimulates expression of pro-osteoclastogenic genes in
marrow and skeletal tissue. Journal of Interferon & Cytokine
Research, 35(6), 480-487.
Britton, R. A., Irwin, R., Quach, D., Schaefer, L., Zhang, J., Lee,
T., McCabe, L. R. (2014). Probiotic L. reuteri treatment prevents
bone loss in a menopausal ovariectomized mouse model. Jour-
nal of cellular physiology, 229(11), 1822-1830.
Cai, S., Kandasamy, M., Rahmat, J. N., Tham, S. M., Bay, B. H.,
Lee, Y. K., & Mahendran, R. (2016). Lactobacillus rhamnosus GG
activation of dendritic cells and neutrophils depends on the dose
and time of exposure. Journal of immunology research, 2016.
Chen, M., Ona, V. O., Li, M., Ferrante, R. J., Fink, K. B., Zhu, S.,
Hersch, S. M. (2000). Minocycline inhibits caspase-1 and cas-
pase-3 expression and delays mortality in a transgenic mouse
model of Huntington disease. Nature medicine, 6(7), 797.
Ciorba, M. A., & Stenson, W. F. (2009). Probiotic Therapy in
RadiationInduced Intestinal Injury and Repair. Annals of the
New York Academy of Sciences, 1165(1), 190-194.
Ciorba, M. A., Riehl, T. E., Rao, M. S., Moon, C., Ee, X., Nava,
G. M., Stenson, W. F. (2012). Lactobacillus probiotic protects
intestinal epithelium from radiation injury in a TLR-2/cyclo-
oxygenase-2-dependent manner. Gut, 61(6), 829-838.
Collins, F. L., Irwin, R., Bierhalter, H., Schepper, J., Britton,
R. A., Parameswaran, N., & McCabe, L. R. (2016). Lactobacil-
lus reuteri 6475 increases bone density in intact females only
under an in ammatory setting. PLoS One, 11(4), e0153180.
Dasika, G. K., Lin, S.-C. J., Zhao, S., Sung, P., Tomkinson, A.,
& Lee, E. Y. P. (1999). DNA damage-induced cell cycle check-
points and DNA strand break repair in development and tumo-
rigenesis. oncogene, 18(55), 7883.
Demers, M., Dagnault, A., & Desjardins, J. (2014). A rand-
omized double-blind controlled trial: impact of probiotics
on diarrhea in patients treated with pelvic radiation. Clinical
Nutrition, 33(5), 761-767.
Deng, H., Li, Z., Tan, Y., Guo, Z., Liu, Y., Wang, Y., Bai, Y. (2016).
A novel strain of Bacteroides fragilis enhances phagocytosis and
polarises M1 macrophages. Scienti c reports, 6, 29401.
Dobrzy
n
´
ska, M. M., Gajowik, A., & Radzikowska, J. (2015). The
effect of in vivo resveratrol supplementation in irradiated mice
on the induction of micronuclei in peripheral blood and bone
marrow reticulocytes. Mutagenesis, 31(4), 393-399.
Fernandes, K. R., Ribeiro, D. A., Rodrigues, N. C., Tim, C.,
Santos, A. A., Parizotto, N. A., Renno, A. C. (2013). Effects of
low-level laser therapy on the expression of osteogenic genes
related in the initial stages of bone defects in rats. Journal of
biomedical optics, 18(3), 038002.
Gamian, A. (2014). Distinct immunomodulation of bone mar-
row-derived dendritic cell responses to Lactobacillus plan-
tarum WCFS1 by two different polysaccharides isolated from
Lactobacillus rhamnosus LOCK 0900. Applied and environ-
mental microbiology, 80(20), 6506-6516.
Gaur, U., & Aggarwal, B. B. (2003). Regulation of proliferation,
survival and apoptosis by members of the TNF superfamily.
Biochemical pharmacology, 66(8), 1403-1408.
Gerbitz, A., Schultz, M., Wilke, A., Linde, H.-J., Schölmerich, J.,
Andreesen, R., & Holler, E. (2004). Probiotic effects on experi-
mental graft-versus-host disease: let them eat yogurt. Blood,
103(11), 4365-4367.
Giralt, J., Regadera, J. P., Verges, R., Romero, J., de la Fuente,
I., Biete, A., Guarner, F. (2008). Effects of probiotic Lactobacil-
lus casei DN-114 001 in prevention of radiation-induced diar-
rhea: results from multicenter, randomized, placebo-controlled
nutritional trial. International Journal of Radiation Oncology
Biology Physics, 71(4), 1213-1219.
González Pereyra, M., Dogi, C., Torres Lisa, A., Wittouck, P.,
Ortíz, M., Escobar, F., Torres, A. (2014). Genotoxicity and
cytotoxicity evaluation of probiotic Saccharomyces cerevisiae
RC016: a 60day subchronic oral toxicity study in rats. Journal
of applied microbiology, 117(3), 824-833.
244 EFFECTS OF PROBIOTIC IN EXPRESSION OF RUNX-2, ALP, OCN AND CASP-3 GENES BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS

Omid Azadbakht et al.
Górska, S., Schwarzer, M., Jachymek, W., Srutkova, D., Brzozo-
wska, E., Kozakova, H., &
Hall, E., & Giaccia, A. (2006). Radiobiology for the Radiologist
Lippincott Williams & Wilkins: Philadelphia.
Hayashi, M., Morita, T., Kodama, Y., Sofuni, T., & Ishidate
Jr, M. (1990). The micronucleus assay with mouse peripheral
blood reticulocytes using acridine orange-coated slides. Muta-
tion Research Letters, 245(4), 245-249.
Huang, B., Guang, M., Ye, J., Gong, P., & Tang, H. (2015). Effect
of increasing doses of -radiation on bone marrow stromal
cells grown on smooth and rough titanium surfaces. Stem cells
international, 2015.
Ji, J., Hu, S.-L., Cui, Z.-W., & Li, W.-F. (2013). Probiotic Bacil-
lus amyloliquefaciens mediate M1 macrophage polarization in
mouse bone marrow-derived macrophages. Archives of micro-
biology, 195(5), 349-356.
Jilka, R. L., Weinstein, R. S., Bellido, T., Par tt, A. M., & Manol-
agas, S. C. (1998). Osteoblast programmed cell death (apopto-
sis): modulation by growth factors and cytokines. Journal of
Bone and Mineral Research, 13(5), 793-802.
Khoshtabiat, L., & Mahdavi, M. (2015). The Role of Oxidative
Stress in Proliferation and Cell Death. Journal of Mazandaran
University of Medical Sciences, 25(127), 130-145.
Li, J., Yan, M., Wang, Z., Jing, S., Li, Y., Liu, G., Fan, Z. (2014).
Effects of canonical NF-B signaling pathway on the pro-
liferation and odonto/osteogenic differentiation of human
stem cells from apical papilla. BioMed research international,
2014.
Liu, C., Zhang, C., Mitchel, R. E., Cui, J., Lin, J., Yang, Y., Cai,
J. (2013). A critical role of toll-like receptor 4 (TLR4) and its’
in vivo ligands in basal radio-resistance. Cell death & disease,
4(5), e649.
Mansouri-Tehrani, H.-A., Rabbani-Khorasgani, M., Hosseini,
S. M., Mokarian, F., Mahdavi, H., & Roayaei, M. (2015). Effect
of supplements: Probiotics and probiotic plus honey on blood
cell counts and serum IgA in patients receiving pelvic radio-
therapy. Journal of research in medical sciences: the of cial
journal of Isfahan University of Medical Sciences, 20(7), 679.
Mariman, R., Kremer, B., Koning, F., & Nagelkerken, L. (2014).
The probiotic mixture VSL# 3 mediates both pro-and anti-
in ammatory responses in bone marrow-derived dendritic
cells from C57BL/6 and BALB/c mice. British Journal of Nutri-
tion, 112(7), 1088-1097.
Mego, M., Holec, V., Drgona, L., Hainova, K., Ciernikova, S.,
& Zajac, V. (2013). Probiotic bacteria in cancer patients under-
going chemotherapy and radiation therapy. Complementary
therapies in medicine, 21(6), 712-723.
Meltz, M. L., Reiter, R. J., Herman, T. S., & Kumar, S. (1999).
Melatonin and protection from whole-body irradiation: sur-
vival studies in mice. Mutation Research/Fundamental and
Molecular Mechanisms of Mutagenesis, 425(1), 21-27.
Michelin, S., del Rosario Perez, M., Dubner, D., & Gisone, P.
(2004). Increased activity and involvement of caspase-3 in
radiation-induced apoptosis in neural cells precursors from
developing rat brain. Neurotoxicology, 25(3), 387-398.
Mozdarani, H., & J Vessal, N. (1993). Cimetidine Can Modify
The Effects Of Whole Body ‘y Irradiation On Lymphohemat-
opoietic System. Medical Journal of The Islamic Republic of
Iran (MJIRI), 7(2), 95-99.
Norizadeh Tazehkand, M., Topaktas, M., & Yilmaz, M. B. (2016).
Assessment of chromosomal aberration in the bone marrow
cells of Swiss Albino mice treated by 4-methylimidazole. Drug
and chemical toxicology, 39(3), 307-311.
Porter, A. G., & Jänicke, R. U. (1999). Emerging roles of cas-
pase-3 in apoptosis. Cell death and differentiation, 6(2), 99.
Rodríguez, M. L., Martín, M. M., Padellano, L. C., Palomo, A.
M., & Puebla, Y. I. (2010). Gastrointestinal toxicity associated
to radiation therapy. Clinical and Translational Oncology,
12(8), 554-561.
Salva, S., Marranzino, G., Villena, J., Agüero, G., & Alvarez, S.
(2014). Probiotic Lactobacillus strains protect against myelo-
suppression and immunosuppression in cyclophosphamide-
treated mice. International immunopharmacology, 22(1), 209-
221.
Seal, M., Naito, Y., Barreto, R., Lorenzetti, A., Safran, P., &
Marotta, F. (2007). Experimental radiotherapyinduced enteri-
tis: A probiotic interventional study. Journal of digestive dis-
eases, 8(3), 143-147.
Spyropoulos, B. G., Misiakos, E. P., Fotiadis, C., & Stoidis, C. N.
(2011). Antioxidant properties of probiotics and their protec-
tive effects in the pathogenesis of radiation-induced enteritis
and colitis. Digestive diseases and sciences, 56(2), 285-294.
Spyropoulos, B. G., Theodoropoulos, G., Misiakos, E. P., Stoidis,
C. N., Zapatis, H., Diamantopoulou, K., Machairas, A. (2013).
The effect of synbiotics on acute radiation-induced diarrhea
and its association with mucosal in ammatory and adaptive
responses in rats. Digestive diseases and sciences, 58(9), 2487-
2498.
Steeve, K. T., Marc, P., Sandrine, T., Dominique, H., & Yannick,
F. (2004). IL-6, RANKL, TNF-alpha/IL-1: interrelations in bone
resorption pathophysiology. Cytokine & growth factor reviews,
15(1), 49-60.
Vejdani, R. , Zali, M. R. (2003). Probiotics and the mechanism
of their effect on the prevention and treatment of Human dis-
eases. Shahid Beheshti University of Medical Sciences.Tehran.
Iran, 27(4) 319-330.
Wang, Y., Zhu, G., Wang, J., & Chen, J. (2016). Irradiation alters
the differentiation potential of bone marrow mesenchymal
stem cells. Molecular medicine reports, 13(1), 213-223
Weiss, G. , Christensen, H. R. , Zeuthen, L.H. , Vogensen, F.K. ,
& Jakobsen, M. (2011). Lactobacilli and bi dobacteria induce
differential interferon-b pro les in dendritic cells. Cytokine,
56(2), 520–530.
Yavropoulou, M. P., & Yovos, J. G. (2007). The role of the Wnt
signaling pathway in osteoblast commitment and differentia-
tion. Hormones-Athens-, 6(4), 279.
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS EFFECTS OF PROBIOTIC IN EXPRESSION OF RUNX-2, ALP, OCN AND CASP-3 GENES 245