Biotechnological
Communication
Biosci. Biotech. Res. Comm. 10(1): 6-10 (2017)
A mini review on
in vitro
propagation of
Swertia
chirayita
an endangered medicinal plant
Shailja
Department of Biotechnology, Dr. Y. S. Parmar University of Horticulture and Forestry, Nauni,
Solan – 173230 (H.P.), India
ABSTRACT
Swertia chirayita is an endangered indigenous medicinal herb. It is used in traditional medicine to treat various ail-
ments. Swertia chirayita is found in temperate regions of the Himalaya at an altitude of 1200–3000 m from Kashmir
to Nepal, Bhutan and grows in the slopes of moist shady places. The species is valued for its bitterness. The bitterness,
antihelmintic, hypoglycemic and antipyretic properties are attributed to amarogentin, swerchirin, swertiamarin and
other active principles of the herb. Its medicinal usage is reported in Indian pharmaceutical codex, the British and the
American pharmacopoeias and in different traditional systems of medicines such as the Ayurveda, Unani and Siddha.
With the passage of time there is increase in demand of this plant, so it is uprooted in its earlier stage and plant is
becoming endangered. There are some biotechnological methods like in vitro propagation and in vitro conservation
which can protect the medicinal plants to be extinct. These two methods help to produce maximum plants in less
time and conserved the plant for long time. These are also very useful in storing valuable germplasms. This review is
mainly focused on in vitro propagation and conservation of the Swertia chirayita.
KEY WORDS:
SWERTIA CHIRAYITA
,
IN VITRO
PROPAGATION, CONSERVATION, MEDICINAL PLANT, TISSUE CULTURE
6
ARTICLE INFORMATION:
*Corresponding Author: shailjabiotech@gmail.com
Received 2
nd
Jan, 2017
Accepted after revision 12
th
March, 2017
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007 CODEN: USA BBRCBA
Thomson Reuters ISI ESC and Crossref Indexed Journal
NAAS Journal Score 2017: 4.31 Cosmos IF : 4.006
© A Society of Science and Nature Publication, 2017. All rights
reserved.
Online Contents Available at: http//www.bbrc.in/
INTRODUCTION
Swertia chirayita Buch.- Hams. ex Wall.belongs to fam-
ily Gentianaceae. It is commonly known as “Chirata”
and in sanskrit it is called as Anaryatikta, Ardhatikta,
Bhunimba, Chiratika, Chiratitka, Haima, Jvarantaka,
Kairata, Kandatiktaka, Kiranta, Kirataka, Kirata Tikta,
Naditikta, Naipala, Nepalanimba, Nidrari, Ramasenka,
Sannipatha, Sutiktaka, Trinanimba, and Viktaka (Anon,
1982 and Kritikar 1984). It is an indigenous species of
temperate Himalayas. Among many species of Swertia
in India chirayita is the only species which is consid-
ered most important for its medicinal properties (Joshi
and Dhawan, 2005).There is no steadiness in literature
that plant is either annual ( Anon, 1982 and Kritikar
1984), or biennial/pluri- annual (Edwards, 1993). The
plant has long been used for its blood-purifying, anti-
fungal and antihelmintic properties (Pant et al. 2011). S.

Shailja
chirayita plants contain several active constituents such
as xanthones, avonoids, iridoids and secoiridoid glyco-
sides that are responsible for its therapeutics properties
(Kumar and Chandra, 2013).
The major phytochemicals of the bitter-tasting plant
include swertiamarin, amarogentin and mangiferin, a
xanthone C-glucoside (Phoboo et al. 2013). Swertiama-
rin is reported to be effective against hepatitis (Wang
et al. 2011) and shown to exhibit anti-diabetic (Vaidya
et al. 2013), anticancer (Kavimani and Manisenthlku-
mar, 2000) activities. Amarogentin is known to be anti-
diabetic(Phobooet al. 2013), anticancer (Pal et al. 2012)
and anti-arthritic (Saravanan et al. 2014).
The plant has an erect, about 2-3 ft long stem and
the whole plant is bitter in taste. It has lanceolate acute
leaves with orange brown or purplish coloured stem, and
contains large continuous yellowish pith. The roots are
simple, tapering , stout, short and almost 7 cm long.
The owering & fruiting occurs between July to Sep-
tember. Flowers of Swertia chirayita are in the form of
numerous small, axillary, opposite, lax cymes arranged
as short branches small, stalked, green-yellow, tinged
with purple colour, rotate and tetramerous. The corolla
is twice as long as the calyx and divided near the base
into four ovate–lanceolate segments. The upper sur-
face of the petal has a pair of nectaries covered with
oblong scales and ending as fringes. Fruit is a small,
one-celled capsule with a transparent yellowish peri-
carp. It dehisces from septicidally into two valves. Seeds
are numerous, minute many-sided and angular. Floral
characteristics such as colourful corolla and presence of
nectaries support cross-pollination in the species. Swer-
tia chirayita contains a yellow bitter ophelic acid and
two bitter glucosides chiratin (Joshi and Dhawan., 2005,
Brahmchari et al., 2004 Pant et al., .2010, Chandra et al.,
2012, Kumar and Staden, 2016).
Swertia chirayita is dif cult to propagate on mass
scale via seed owing to non-availability of seeds due to
harvesting of plants before seeds mature. So instead of
going for conventional approaches of, the application of
alternative reproducible micropropagation strategies has
become inevitable for mass propagation and sustainable
utilization of this age-old medicinal plant.
Due to its over
exploitation for different medicinal uses and commercial
purposes its availability is decreasing day by day so it’s
becoming extinct.
S. chirayita conservation status has
been categorized as “critically endangered” (Joshi and
Dhawan, 2005 and Padhan et al. 2015). Developing an in
vitro regeneration protocol for Swertia chirayita is urgent
to promote large-scale production for ex situ conserva-
tion and for satisfying the pharmaceutical needs. Syn-
thetic seed technology is also an applied application of
modern plant biotechnology which offers tremendous
potential for easy handling, micropropagation and plant
germplasm conservation through cryopreservation (Gan-
tait et al. 2015 and Kumar and Staden, 2016).
This article brie y reviews the in vitro propagation
and in vitro conservation of the plant. This is an attempt
to compile and document information on micropropaga-
tion and in vitro conservation of S. chirayita and high-
light the need for research.
IN VITRO
PROPAGATION
Wawrosch et al. (1999) developed as protocol for micro-
propagation of Swertia chirayita. They found that mul-
tiplication by adventitious shoot regeneration from root
explants is most suitable method for the propagation of
Swertia chirayita. A two-step system consisting of an ini-
tial 3 weeks cultivation on modi ed MS medium supple-
mented with 3 μM 6-benzyladenine followed by another
period of 3 weeks in plant growth factor free medium
was used. The pH of all nutrient media was adjusted to
5.8± 0.1. The root explants taken from 6-to 8-week-old
plants are very well suited for the multiplication of Swer-
tia chirayita through regeneration of adventitious shoots.
The explants were cultured on modi ed basal MS medium
with 3 mMBAP for 3 weeks, followed by another 3 weeks
on hormone-free basal medium. An average of 1.9 very
healthy shoots per 5-mm explant were obtained. Dipping
of the shoots in an aqueous solution of NAA (15 ppm) fol-
lowed by 3 to 4 week cultivation period on hormone-free,
half-strength MS medium proved to be the most ef cient
method for rooting of Swertia chirayita.
Chaudhuri et al. (2007) produced genetically uni-
form plants from nodal explants of Swertia chirayita
Buch. Ham. ex Wall. Shoot regeneration was obtained
in shoot inducing medium containing half-strength MS
basal medium supplemented with 0.44 μM 6-BAP and
4.65 μM 6-furfurylaminopurine. The highest number of
shoots, that is 18 shoots per explant were obtained when
medium was again used with 10 mM KNO
3
and 75 mg/l
of casein hydrolysate. The plantlets were successfully
transferred to the eld and produced viable seeds.
Joshi and Dhawan (2007) described the micropro-
pagation of Swertia chirayita through axillary shoot
multiplication from 4 weeks old seedling derived nodal
explants. 4.5 fold multiplication was obtained after
every 4 weeks on MS medium supplemented with 4μM
BAP and 1.5μM 2ip. Rooting was optimized on modi ed
MS medium supplemented with 1μM NAA and 500 mg
of activated charcoal which showed 94% of rooting.
A protocol for plant regeneration through indirect
organogenesis was established by Bisht et al. (2008) for
Swertia angustifolia Buch.-Hams. Callus was induced on
MS basal medium supplemented with cytokinin (Kine-
tin or BA) and auxin (2,4-D/IBA/NAA) from leaf, petiole
and stem explants. Higher concentration of Kinetin and
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS A MINI REVIEW ON
IN VITRO
PROPAGATION OF SWERTIA CHIRAYITA 7
Shailja
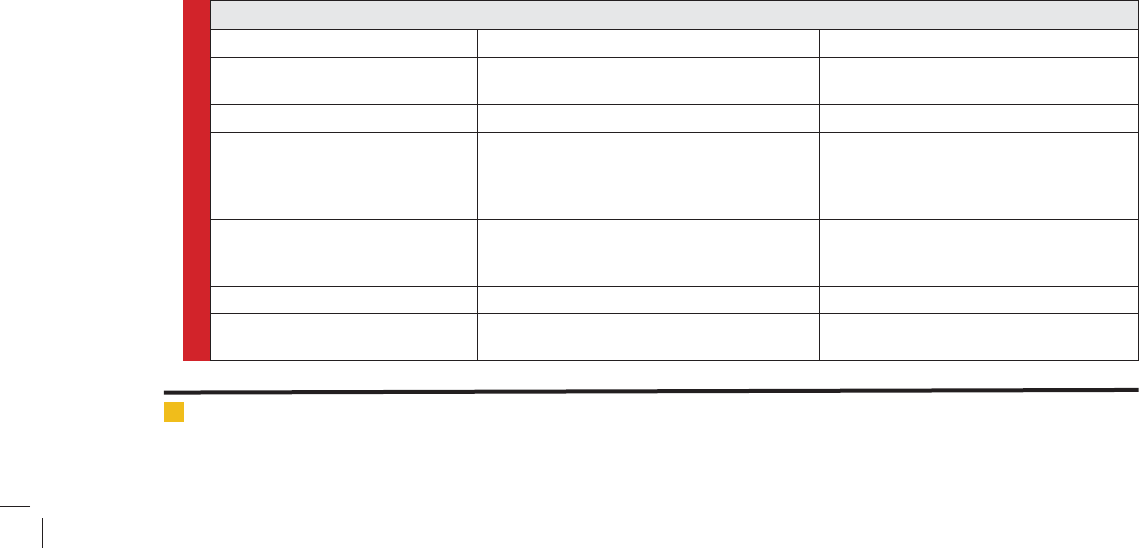
Table 1. list of important bioactive constituents isolated from Swertia chirayita
Active constituents Biological activities References
Amarogentin (chirantin) Topoisomerase inhibition, chemo-preventive
and antileishmanial effects .
[Ray 1996), [Saha and Dass 2005), [Phoboo
et al. 2013]
Amaroswerin Gastro-shielding [Niiho 2005], [Phoboo et al. 2013]
Gentianine Anti-in ammatory, anesthetic, antihistaminic,
anticonvulsant properties, hypotensive,
antipsychotic, lenitive, diuretic, antimalarial,
antiamoebic and antibacterial properties.
[Song Zhen Yu 1958; Geng Tao 1959; Kwak
2005]. [Bhattacharya 1974], [Mansoor and
Malghani MAK, 2005] , [Natarajan et al.,
1974, [Phoboo et al. 2013]
Swerchirin Antimalarial, hypoglycemic, hepatoprotective,
pro-heamatopoitic, and weak chemo preventive
pharmacological effects.
[Arino 1997], [Bajjpai 1991], [Saxena 1996],
[Ya 1999] [Hirkawa1987], [Phoboo et al.
2013].
Swertiamarin Analgesic property [Lei 1982], [Phoboo et al. 2013]
xanthones, avonoids, iridoids and
secoiridoid glycosides
Therapeutic properties Joshi and Dhawan 2005
2,4–D (2.5-3.0 mg/l ) exhibited best callusing in leaf and
better in petiole explants. BA and NAA in the range of
1.5-2mg/l exhibited fast proliferation in callus mass in
both explants.Shoots were regenerated on MS medium
containing BA (1.5– 2.5 mg/l) and IBA or NAA (0.5-1.5
mg/l). Rooting was obtained with full or half MS media
with IBA or NAA (0.5 -1.5 mg/l).
Balaraju et al. (2009) established rapid system for
micropropagation of Swertia chirayita Buch. Hams.
ex Wall. using shoot tip explants derived from in vitro
grown seedlings. MS medium containing BAP (1.0 mg/l)
and Kinetin (0.1 mg/l) along with 2% sucrose induced
highest number of multiple shoots per explants. Micro
proliferated shoots were transferred to elongation
medium amended with 0.1 mg/l GA
3
. The highest fre-
quency of rooting was obtained in half MS medium sup-
plemented with 0.1 mg/l NAA.
Wang et al. (2009) investigated the effects of phyto
hormones on shoot regeneration from the leaves of eld
grown Swertia chirayita .The best result obtained in MS
medium supplemented with 13.2μM 6- BAP and 0.54
μM - NAA. The highest rate of shoot regeneration was
96.5% on the medium with 0.54 μM NAA. Adventitious
shoots were transferred on the rooting medium. Root-
ing was optimized on MS medium supplemented with
NAA 5.40μM. Pant et al. (2010) developed an ef cient
protocol for in vitro propagation of Swertia chirayita.
Axillary shoot bud multiplication was achieved using
nodal segments as explants. A combination of BAP 4.4
μM + IAA 2.85 μM + Adenosine sulphate 271.45 μM
proved to be the best giving 11.8 fold multiplication
with average shoot length of 1.9 cm after 4 weeks and
18.5 fold multiplication with mean shoot length 2.6 cm
was observed. Best rooting was observed on MS medium
with IBA 4.90μM. Maximum mean number of root per
shoot 35.3 was observed after 8 weeks.
Pant et al. (2011) described procedure for regeneration
of complete plantlets of Swertia chirayita via indirect
organogenesis. Callus was obtained from in vitro regen-
erated roots on MS medium supplemented with varying
concentrations of BAP and 2,4-D. BAP (13.32 μM) in
combination with 2,4-D (0.90 μM) proved to be the most
effective concentration for callus induction, multiplication
and adventitious shoot regeneration from callus surface.
The optimal hormone combination for shoot multiplica-
tion was shown to be BAP (8.88 μM), IAA (2.85 μM) and
271.45 μM adenine sulphate (Ads). Individual elongated
shoots were rooted on half-strength MS medium supple-
mented with varying concentrations of auxins. Best root-
ing was obtained with MS Medium supplemented with
4.90 μM IBA. In vitro raised plantlets with well developed
shoots and roots were acclimatized successfully.
Jha et al. (2011) carried out in vitro propagation and
conservation of Swertia bimaculata Hook. f. & Thomas.
Seeds were germinated aseptically with low concen-
tration of BA (2.22 μM) or Kinetin (2.32 μM). The best
response of in vitro grown shoots was obtained on MS
medium with BA (2.22 μM), Kinetin (2.32 μM ) and NAA
(0.54μM). The number of shoots were increased to 20.6
on addition of 10 mM potassium nitrate (KNO
3
) in the
medium. Isolated shoots induce 100% rooting on basal
medium with in 5 weeks. Rooted plants were hardened
and transplanted in soil with 80-90% survival rate.
CONSERVATION OF
SWERTIA CHIRAYITA
Over exploitation of plant sources is a normal occurrence
due to its increasing demand. It is mostly used as tradi-
tional drug. The demand of this plant is on rise at both
national and international level due to its multiple uses
which leads to increase over harvesting of wild popula-
tions and ultimately in reduction of population. Accord-
ing to the International Union of Conservation of Nature
(IUCN) criteria, S.chirayita conservation status has been
categorized as “critically endangered” (Joshi and Dhawan,
2005). There are limitations in the use of seed propaga-
8 A MINI REVIEW ON
IN VITRO
PROPAGATION OF SWERTIA CHIRAYITA BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS

Shailja
tion, due to low viability, and low germination percent-
ages (Badola and Pal, 2002; Chandra et al., 2012).
Biotechnology offers new means of conservation
of Swertia chirayita. Synthetic seed production is one
of them.In this method somatic embryos are encap-
sulated in a suitable matrix like sodium alginate along
with insectisides, fungicides and herbicides. Kumar
et al. (2014) reported on synthetic seed production and
plant regeneration of S.chirayita from somatic embryos.
However, studies are required to improve this technol-
ogy so that it can be used on large scale.. Cryopreserva-
tion this is also one another method of conservation. In
this method, the cells are preserved in the frozen state.
The germplasm is stored at a very low temperature using
liquid nitrogen (at-196ºC). The cells stay in completely
inactive state and thus can be conserved for long peri-
ods. Certain compounds like- DMSO (dimethyl sulfoxide),
glycerol, ethylene, propylene, sucrose, mannose, glucose,
praline, acetamide are added during the cryopreservation.
These are called cryoprotectants and prevent the damage
caused to cells by reducing the freezing point and super
cooling point of water., (Ara et al., 2000; Sharma et al.,
2013a; Perveen and Anis, 2014; Gantait et al., 2015)
CONCLUSION AND FUTURE PERSPECTIVES
This review article revealed the morphogenetic potential
of leaves of Swertia chirayita as a source for micro-
propagation. The explants can be easily and regularly
obtained from established shoot cultures and do not
require disinfection treatment hence being ideal for
germplasm exchange and cryopreservation. Normal leaf
culture establishment for a number of plant species have
the ability to accumulate secondary metabolites and
plays important role in pharmaceuticals. In vitro conser-
vation was done by cryo conservation. The government
has imposed total ban on collection or removal of plant-
ing materials of this important species from their natural
populations but this is not possible without the support
of local healers. There should be awareness among local
peoples to control the overexploitation. Scientists can-
not conserve this species without the help of local heal-
ers. There are many institutes and universities where
the research work is going on but that work should be
explored at higher level so that other researchers gain
experience from that and help in conserving endangered
medicinal plants otherwise this species also become
extinct. Biotechnological approaches are required in
future also to promote its medicinal use.
REFERENCES
Anon.(1982) In The Wealth of India: Raw Materials,CSIR, New
Delhi. vol. X: Pages 78–81
Ara H., Jaiswal U., and Jaiswal V.S. (2000) Synthetic seed:
prospects and limitations. Current Science Vol. 78: Pages
1438–1444.
Arino A. (1997) The extraction of yellow gentian root (Gen-
tianalutea L.) Zeitschrift fur Leben smitteluntersuchung und-
Forschung, Vol.205: Pages 295-299
Badola H. K. and Pal M.(2002) Endangered medicinal plant
species in Himachal Pradesh. Current Science Vol 83: Pages
797–798.
Bajpai M.B. (1991) Hypoglycemic effect of swerchirin from the
hexane fraction of Swertia chirayita. Planta Me Vol. 57:Pages
102-104
Balaraju K, Agastian P. and Ignacimuthu S.(2009) Micropro-
pagation of Swertia chirata Buch. Hams. ex Wall : a critically
endangered medicinal herb. Acta Physiologiae PlantarumVol
31No 3 : Pages 487- 494
Bhattacharya S. K. (1974) Letter: Chemical constituents of gen-
tianaceae. XI. Antipsychotic activity of gentianine. J Pharm
Sci Vol .63: Pages 1341-1342
Bisht S. S., Bhandari S. and Bisht N. S. (2008) In vitro organo-
genesis in Swertia angustifolia . Journal of Hill Research Vol
21 No. 2: Pages 41-46
Brahmchari G., Mandal S., Gangopadhyay A., Gorai D., and
Mukhopadhyay B.(2004) Swertia (Gentiananceae):chemical
and pharmalogical aspects.Chem. Biodiversity Vol. 1:Pages
1627-1651
Chandra S., Kumar V., Bandopadhyay R.,and Sharma M.
M.(2012) SEM and elemental studies of Swertia chirayita: a
critically endangered medicinal herb of temperate Himalayas.
Current Trends Biotechnology Pharm. Pages 6381–6388.
Chandra S., Kumar V., Bandopadhyay R.,and Sharma
M.M.(2012) SEM and elemental studiesof Swertia chirayita
critically endangered medicinal herb of temperate Himalayas.
Curr.Trends.Biotechnol.Pharm. Pages 6381–388
Chassot P. (2000) Phylogenetic position of the genus Swertia
(Gentiananceae) in the subtribe Swertiinae .American Journal
of Botany. 87(suppl):Pages 118-119
Chaudhuri R. K., Pal A. and Jha T. B. (2007) Production of
genetically uniform plants from nodal explants of Swertia chi-
rayita Buch. Ham. Ex Wall: an endangered medicinal herb. In
Vitro Cellular and Developmental BiologyPlantVol.43 No.5:
Pages 467-472
Edwards D.M.(1993)The marketing of non-timber forest prod-
uct from the Himalayas : The trade between East Nepal and
India rural development Forestry Network. Rural Development
For Network.Pages1-21
Gantait S., Kundu S., Ali N., and Sahu N.C.(2015)Synthetic
seed production of medicinal plants:a review on in uence of
explants, encapsulation agent and matrix. ActaPhysiol.Plant
37,98.doi:10.1007/s11738-015-1847-2
Gaur R.D. (1999) Flora of the District Garhwal North West
Himalaya (with ethnobotanical notes)Transmedia, Srinagar
(Garhwal) Page208
Geng Tao. (1959)Journal of Physiology. Vol. 23:Page 203
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS A MINI REVIEW ON
IN VITRO
PROPAGATION OF SWERTIA CHIRAYITA 9

Shailja
Hirkawa K. (1987) Chemo preventive action of xanthone deriv-
atives on photosensitized DNA damage. Photochemical Photo-
biology Vol .81: Pages 314-319
Jha T. B., Dafadar A. and Chaudhuri K. R. (2011) Somatic
embryogenesis in Swertia chirata Buch. Ham. Ex Wall: a
multipotent medicinal herb. Asian Journal of Biotechnology
Vol 3: Pages 186-193
Joshi P and Dhawan V.(2005) Swertia chirayita –an overview.
Current Sciences.vol.89 No. 4:Pages 635-640
Joshi P, and Dhawan V. (2007) Axillary multiplication of Swer-
tia chirayita: a critically endangered medicinal herb of temper-
ate Himalayas. In Vitro Cellular and Developmental Biology
PlantVol. 43 No.6: Pages 631-638
Kavimani S. and Manisenthl kumar K. T. (2000) Effect of metha-
nolic extract of Enicostemma littorale on Dalton’s ascitic lym-
phoma. Journal of Ethnopharmacology Vol. 71 :Pages 349–352
Kirtikar K. R. and Basu B. D. (eds)(1984) Indian Medicinal
Plants, Allahabad.vol. III: Pages 1664–1666
Kumar V. and Chandra S. (2013) Ef cient regeneration and
antioxidant activity of the endangered species Swertia chi-
rayita. International Journal of Pharma and Bio Sciences Vol
4 No. 4:Pages 823-833
Kumar V. and Staden J. V.(2016)
A Review of Swertia chi-
rayita (Gentianaceae)as aTraditional Medicinal Plant. Frontiers
in Pharmacology Vol. 6(308):Pages1-14
Kwak W. J. (2005) Effects of gentianine on the production
of proin ammatory cytokines in male Sprague-Dawley rats
treated with lipopolysaccharide (LPS) Biol Pharm Bull, Vol. 28:
Pages750-753
Lei Wei Ya (1982) Swertia marins central inhibitory effects.
Journal of Chinese Materia Medica, Vol 13: Page 368
Mansoor A and Malghani MAK. (2005) .Diuretic effect of Gen-
tiana olivieri and its alkaloid gentianine .Botany. m plants.
August 13-17, Austin, Texas. Oral presentation
Natarajan P. N., Wan A. S., and Zsaman V. (1974) Antimalarial,
antiamobeic and toxicity tests on gentianine. Planta Med. Vol
.25: Pages 258-260
Niiho Y. (2005) Gastro protective effects of bitter principles
isolated from Gentian root and Swertia herb on experimentally
induced gastric lesions in rats. Journal of Natural Medicine,
Vol. 60:Page 888
Padhan J. K., Kumar V., Sood H., Singh T. R. and Chauhan R.
S. (2015) Contents of therapeutic metabolites in Swertia chi-
rayita correlate with the expression pro les of multiple genes
in corresponding biosynthesis pathways. Phytochemistry DOI
10.1016.
Pal D., Sur S., Mandal S., Das A., Roy A. , Das S. and Panda
C. K. (2012)
Prevention of liver carcinogenesis by amarogentin through
modulation of G1/S cell cycle check point and induction of
apoptosis. Carcinogenesis Vol.33 :Pages2424–2431
Pant M., Bisht P. and Gusain M. P. (2011) In vitro propagation
through root derived callus culture of Swertia chirata Buch.
Ham. ex Wall. African Journal of Biotechnology Vol 11 No 29
:Pages 7408-7416
Pant M., Bisht P., and Gusain M. P. (2010) In vitro propagation
through axillary bud culture of Swertia chirata Buch. Ham. ex
Wall: an endangered medicinal herb. International Journal of
Integerative Biology Vol. 10 No 1:Pages 48-53
Perveen S., and Anis M. (2014) Encapsulation of internode
regenerated adventitious shoot buds of Indian Sirisin alginate
beads for temporary storage and two fold clonal plant produc-
tion. Acta Physiology Plantareum Vol. 36, Pages 2067–2077.
doi: 10.1007/s11738-014-1584-y
Phoboo S., Pinto M. D. S., Barbosa A. C. L., Sarkar D. , Bhow-
mik P. C., Jha P. K. and Shetty K. (2013) Phenolic-linked bio-
chemical rationale for the anti-diabetic properties of Swertia
chirayita (Roxb. ex Flem.) Karst. Phytotherapy Research Vol.
27 :Pages 227–235
Ray S. (1996) Amarogentin, a naturally occurring secoiridoid
glycoside and a newly recognized inhibitor of topoisomer-
ase1 from Leishmania donovani J Nat Prod, Vol. 59: Pages
27-29
Saha P., and Das S. B. (2005) Fraction of Swertia chirata pre-
vent carcinogenic risk due to DMBA Exposure. Indian Journal
of Medical Research, 22-27 Poster Presentation
Saravanan S., Hairul Islam V. I., Prakash Babu N., Pandikumar
P., Thirugnana sambantham K., Chellappandian M ., Simon
D. R., Gabriel C., Paulraj M. and Ignacimuthu S. (2014) Swer-
tia marin attenuates in ammation mediators via modulating
NF-jB/I jB and JAK2/STAT3 transcription factors in adjuvant
induced arthritis. European Journal of Pharmaceutical Sci-
ences Vol. 56: Pages70–86
Saxena A. M. (1996)Mode of action of three structurally differ-
ent hypoglycemic agents: a comparative study. Indian Journal
of Experimental Biology Vol. 34: Pages 351-355
Sharma N., Varshney V. K., Kala R. P., Bisht B., and Sharma
M.(2013) Antioxidant capacity and total phenolic content of
Swertia chirayita (Roxb.ex Fleming) H. Karst. In Uttarakhand.
Int.J.Pharm.Sci.Rev.Res. Vol 23:Pages 259–261.
Song Zhen Yu. (1958) Journal of Physiology. Vol. 22 :Page 201
Vaidya H., Goyal R. K. and Cheema S. K. (2013) Anti-diabetic
activity of swertiamarin is due to an active metabolite, gentia-
nine, that upregulates PPAR-c gene expression in 3T3-L1 cells.
Phytotherapy Research Vol. 27: Pages 624–627
Wang Li, An Lizhe, Hu Yan Ping, Wei Lixin and Li Yi. (2011)
In uence of phytohormones and medium on the shoot regen-
eration from leaf of Swertia chirata Buch. Ham. ex Wall in vitro.
African Journal of BiotechnologyVol 8 No 11: Pages 2513-
2517
Wawrosch C., Maskay N., and Koop B. (1999) Micropropaga-
tion of the threatened Nepalese medicinal plant Swertia chi-
rata Buch-Ham. Ex Wall. Plant Cell Report Vol. 18 No 12:
Pages 997-1001
Ya B. Q. (1999) Protective effect of swerchirin on hematopi-
oesis in 60 Co-irrdiated mice. Phytomedicine Vol. 6: Pages
85-88
10 A MINI REVIEW ON
IN VITRO
PROPAGATION OF SWERTIA CHIRAYITA BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS