Biotechnological
Communication
Biosci. Biotech. Res. Comm. 10(2): 359-364 (2017)
Antibacterial and antioxidant activity of leaf organic
extracts of local cultivars of
Murraya koenigii
(L.)
Spreng from Tamilnadu
M. Kavitha
School of Biosciences and Technology, VIT University, Vellore-632014, Tamilnadu, India
ABSTRACT
The aim of the present study was to evaluate the antibacterial activity against ve bacterial pathogens and anti-
oxidant activity of three organic extracts prepared from the leaves of local cultivars of Murraya koenigii. Agar well
diffusion was utilized to determine the zone of inhibition. Broth microdilution was employed to nd out the minimal
inhibitory concentration (MIC) and minimal bactericidal concentration (MBC). Anti-oxidant activity was analysed
using 2,2-Diphenyl-1-picryl hydrazyl (DPPH) free radical scavenging assay. The organic extracts displayed promis-
ing antibacterial activity against Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa. Ethyl alcohol
extract presented highest inhibitory activity when compared to hexane and chloroform extracts. In relation to agar
diffusion method, the ethyl alcohol extract displayed lowest MIC and MBC value of 12.5 and 25 mg ml
-1
against
Staphylococcus aureus and same MIC and MBC value of 25 and 50 mg ml
-1
against Escherichia coli and Pseudomonas
aeruginosa. The radical-scavenging activity of ethyl alcohol extract (IC
50
= 30 μg ml
-1
) is very close to that of the
standard compound, gallic acid (IC
50
value of 35 μg ml
-1
). The results suggested that the local cultivars of M. koenigii
carry bene cial health effects in terms of antimicrobial and anti-oxidant activities and could be used as a promising
dietary supplement.
KEY WORDS: MURRAYA KOENIGII; ETHYL ALCOHOL EXTRACT; CARBAZOLE ALKALOID; ANTIBACTERIAL ACTIVITY; ANTIOXIDANT ACTIVITY
359
ARTICLE INFORMATION:
*Corresponding Author: mkavitha1972@gmail.com
Received 27
th
Nov, 2017
Accepted after revision 26
th
Sep, 2017
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007 CODEN: USA BBRCBA
Thomson Reuters ISI ESC and Crossref Indexed Journal
NAAS Journal Score 2017: 4.31 Cosmos IF: 4.006
© A Society of Science and Nature Publication, 2017. All rights
reserved.
Online Contents Available at:
http//www.bbrc.in/
DOI: 10.21786/bbrc/10.3/4

360 ANTIBACTERIAL AND ANTIOXIDANT ACTIVITY OF LEAF ORGANIC EXTRACTS OF LOCAL CULTIVARS BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
M. Kavitha
INTRODUCTION
Spices and herbs which are used in food preparations
traditionally are well known to contain several health
promoting bioactive substances. Herbal medicines are
used alternatively in many countries to cure many ail-
ments (Kamboj, 2000). Herb based medicines are now
marketed as nutraceuticals (health foods) (Brower,
1998). Indian Ayurvedic medicine is believed to be more
than 5000 years old and it was well practiced and well
recorded over the years (Garodia et al., 2007). Herbal
drugs are generally used to prevent and treat various
diseases and ailments or to support health and healing
(De Smet, 1997). The World Health Organization (WHO)
has also recognized the importance of traditional medi-
cine and has set precise guidelines for the evaluation
of the safety, ef cacy, and quality of herbal medicines.
In recent years the research on the medicinal proper-
ties of various traditionally used medicinal plants has
gained considerable interest in order to ascertain their
true medicinal values and also to assess their toxicity
(Newman and Crag, 2007). Some drugs have been dis-
continued due to their toxicity, while others have been
modi ed or combined with additional herbs to counter-
balance side effects (Harish, 2001).
Bioactive principles of plants, also known as secondary
metabolites exert a range of pharmacological activities in
human beings besides serving as defense barriers in plants
(Paul et al., 2015). They are responsible for therapeutic
activities like hypoglycemic, anti-diabetic, anti-oxidant,
anti-microbial, anti-in ammatory, anti-carcinogenic,
anti-malarial, anti-cholinergic, anti-leprosy activities
etc. (Makri and Kintzios, 2007; Negi etal., 2011). Com-
mon bioactive principles present in plants are, alkaloids,
avonoids, polyphenols, terpenoids, coumarins, tannins,
saponins, anthocyanins and glycosides (Paul etal., 2015).
Additionally secondary metabolites also prevent
spoilage and deterioration of food products Panghal
et al., 2011). Murraya koenigii (L.) Spreng popularly
known as curry leaf plant is a familiar spice used in
India for its characteristic avour and aroma. According
to Wealth of India, it is also used in traditional Indian
medicine as analgesic, febrifuge, stomachic, carminative
and for the treatment of dysentery and skin eruptions.
Recent studies on M. koenigii revealed its medicinal val-
ues like antidiarrhoeal, antimicrobial, hepatoprotective,
radical-scavenging, hypoglycemic, and immunomodu-
latory properties (Maa et al., 2016). Phytochemical
analysis of leaves, bark and root showed the presence of
several bioactive constituents like carbazole alkaloids,
-carotene, polyphenols, terpenoids and coumarins
(Malwal and Sarin, 2011; Gahlawat etal., 2014).
The pharmacological activities of M. koenigii as
reported in different studies seem to exhibit a certain
variation which is attributed mainly to the climatic dif-
ferences among locations (Onayade and Adebajo, 2000).
Therefore it has become a common practice to con-
rm that the local cultivars of M. koenigii possess the
expected bene cial effects when consumed regularly.
There are a few reports on phytochemical analysis and
pharmacological activities of M. koenigii from India
(Ningappa et al., 2008; Panghal et al., 2011; Malwal
and Sarin, 2011; Biswas et al., 2012). But to my best
knowledge, in recent years there are no studies on the
antioxidant and antimicrobial activities of M. koenigii
from Tamilnadu, a southern state of India. Therefore the
present study on antibacterial and antioxidant activity
of leaf organic extracts of local cultivars of Murraya
koenigii from Tamilnadu was undertaken.
MATERIALS AND METHODS
2,2-Diphenyl-1-picrylhydrazyl (DPPH) was procured
from Sigma chemicals. Mueller Hinton agar and broth
were purchased from Hi-Media, Mumbai. All other
chemicals used were of analytical grade commercially
available in India.The antibacterial activity of the
organic extracts was tested against ve bacterial strains.
The only Gram positive bacterium included in the study
was Staphylococcus aureus MTCC 96 (ATCC 9144).
Four Gram negative bacteria used were Escherichia coli
MTCC 901 (ATCC 13534), Klebsiella pneumoniae MTCC
109 (ATCC 15380), Salmonella paratyphi MTCC 735 and
Pseudomonas aeruginosa MTCC 8291. All these bacteria
were obtained from Microbial Type Culture Collection
(MTTC), Institute of Microbial Technology, Chandigarh,
India and they were maintained on nutrient agar. M.
koenigii leaves were collected locally from various loca-
tions of Tamil Nadu, India and authenticated by Bot-
anist. Fresh leaves were washed thoroughly and dried
under shade till constant weight. The dried leaves were
ground to powder in a mixer grinder.
100 g of shade dried M. koenigii leaves powder was
extracted with hexane, chloroform and ethanol sepa-
rately at room temperature for 2 days and then the sol-
vents were evaporated using vacuum rotary evaporator
at 50
o
C. The organic extracts thus obtained were dried at
room temperature till constant weight and then stored in
brown bottles at 4
o
C for further study.
Antibacterial activity of the three organic extracts
obtained from M. koenigii was evaluated by agar well
diffusion method on Mueller-Hinton agar (MHA). The
bacterial strains were grown on MHA and suspensions
were prepared to match 0.5 McFarland standard. 100 μl
of the suspension was spread on MHA plates. Wells of
6 mm diameter were punched in the seeded agar using
sterile borer. Test well were loaded with 50 μl extracts

BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS ANTIBACTERIAL AND ANTIOXIDANT ACTIVITY OF LEAF ORGANIC EXTRACTS OF LOCAL CULTIVARS 361
M. Kavitha
(100 mg ml
-1
) and negative control well loaded with
same volume of solvent. Ampicillin and streptomycin
were used as a positive control. The plates were incu-
bated at 37ºC for 24 h. The diameter of inhibition zones
were measured in mm and results were recorded.
Minhibitory concentration (MIC) of the organic
extracts was tested by the standard Clinical and labora-
tory standards institute (CLSI) methods (Wayne, 2008).
MIC was the lowest concentration of the extract which
gives no visible bacterial growth. Broth microdilution
technique was used to determine the MIC (Elansary
etal., 2012). The organic extracts from M. koenigii were
serially diluted to obtain the dilutions, 200, 100, 50, 25,
12.5, 6.25 and 3.12 mg ml
-1
. 100 μl from each dilution
was transferred to respective wells followed by 90 μl of
double strength Mueller-Hinton broth (MHB). Finally 10
μl respective bacterial suspensions were added to give
nal dilution of 100, 50, 25, 12.5, 6.25, 3.12 and 1.56
mg ml
-1
and the microtiter plate was incubated at 37
o
C
for 24 h. Blank, positive and negative controls were in
place. After incubation, the lowest concentrations of the
extract, which did not show any visual growth of test
organisms, were considered as MICs.
Minimum bactericidal concentration (MBC) of the
organic extracts was also determined by the standard
CLSI methods (Wayne, 2008). MBC was the lowest con-
centration of the extract which kills the initial bacterial
inoculum. Broth microdilution technique was performed
to determine the MBC. The extracts from M. koenigii
were serially diluted to obtain the dilutions, 200, 100, 50,
25, 12.5, 6.25 and 3.12 mg ml
-1
. 100 μl from each dilu-
tion was added to corresponding wells followed by 90 μl
of double strength MHB. At last 10 μl respective bacte-
rial suspensions were transferred to provide nal dilu-
tion of 100, 50, 25, 12.5, 6.25, 3.12 and 1.56 mg ml
-1
and
the plate was incubated at 37
o
C for 24 h. Blank, positive
and negative controls were also included. After incuba-
tion, the wells which did not show any visual growth
of test organisms were considered. 100 μl of the sam-
ple from those wells were transferred aseptically onto
MHA, spread uniformly and the plates were incubated
at 37
o
C for 24 h. After incubation, the concentrations
at which no visible growth was seen were xed as the
MBC.
Free radical scavenging activity of the organic extracts
from M. koenigii was evaluated by its ability to scavenge
DPPH (Elansary etal., 2012). DPPH is a free radical, but
stable in nature. The DPPH solution is initially violet in
color which fades when antioxidants donate hydrogen.
The change in color is monitored using a spectrophotom-
eter and thus the DPPH free radical scavenging activity
is calculated (Molyneux, 2004). A stock solution of 0.1
mM DPPH in ethanol was prepared. Test concentrations
of organic extracts were made in the range of 50-250 μg
ml
-1
in ethanol. Gallic acid was used as standard. 1 ml
of DPPH stock and 3 ml of test samples were mixed and
allowed to react at room temperature for 30 min. The
absorbance was measured at 517 nm using an ultravio-
let visible spectrophotometer (Shimadzu, Japan). Lower
absorbance of the reaction mixture indicated the higher
free radical scavenging activity (Saikia and Upadhyaya,
2011). The percentage of free radical scavenging was
calculated using the following Eq.
where A0 is the absorbance of the DPPH solution and AT
is the absorbance of the test or reference. The percentage
of free radical scavenging was then plotted against the
concentration and a regression equation was obtained to
calculate the IC
50
which is de ned as the total antioxi-
dants necessary to decrease the initial DPPH radical by
50% (Koleva etal., 2002).
All the experiments were done in triplicates. The
values in the tables represent the arithmetic mean and
standard deviation or mean. The standard error was
within experimental limits.
RESULTS AND DISCUSSION
The organic extracts obtained from M. koenigii were less
viscous in nature and brownish in colour. The yield of
the extracts was estimated to be 7, 6.5 and 9.2 % (v/w)
for hexane, chloroform and ethanol respectively. The
yield in the present study was comparable to the yield
reported in other investigations
(Ningappa etal., 2008;
Brind etal., 2014).
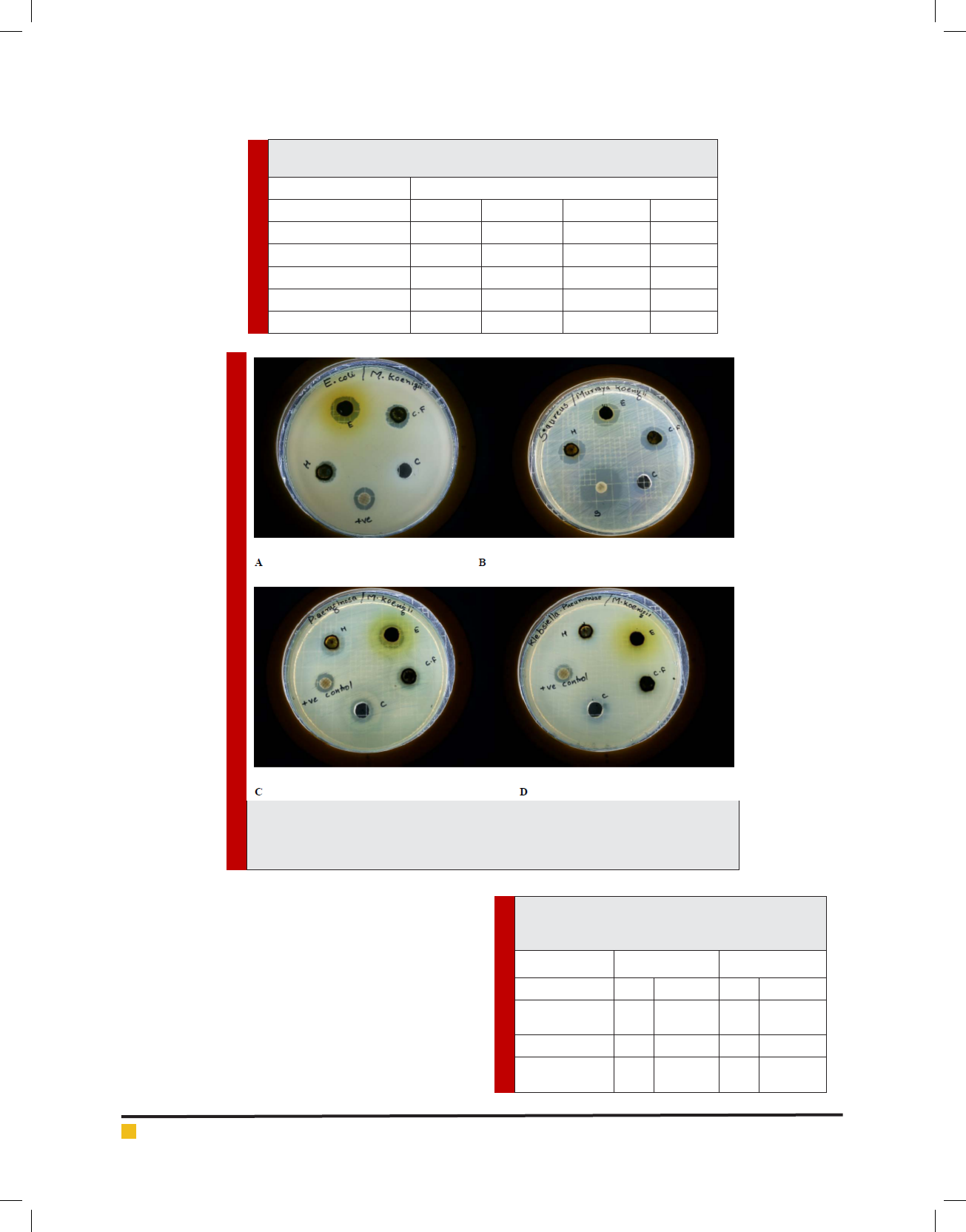
The results of antibacterial activity were presented in
Table 1. Based on the diameter of zone of inhibition,
the three organic extracts exhibited inhibitory activ-
ity against Staphylococcus aureus, Escherichia coli and
Pseudomonas aeruginosa (Figure 1). Klebsiella pneumo-
niae and Salmonella paratyphi were found to be resist-
ant except for chloroform extract which showed a very
faint zone of inhibition. The organic extracts presented a
good potential of antibacterial activity against Escheri-
chia coli and Pseudomonas aeruginosa with the zones of
inhibition well above standard antibiotic. Ethyl alcohol
extract displayed highest inhibitory activity when com-
pared to hexane and chloroform extracts. The results
were comparable to that of previous studies in which the
researchers reported that the various organic extracts of
M. koenigii exhibited signi cant antimicrobial activity
against both Gram positive and Gram negative bacteria
(Panghal etal., 2011; Baskaran etal., 2011). This prop-
erty is attributed to several carbazole alkaloids present
in the organic extracts (Malwal and Sarin, 2011).
362 ANTIBACTERIAL AND ANTIOXIDANT ACTIVITY OF LEAF ORGANIC EXTRACTS OF LOCAL CULTIVARS BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
M. Kavitha
FIGURE 1. MHA plates exhibiting zone of inhibition of M. koenigiiorganic extracts
against (A) Escherichia coli, (B) Staphylococcus aureus, (C) Pseudomonas aeruginosa
and (D) Klebsiella pneumoniae.
Table 1. Antibacterial activity of M. koenigii organic extracts by Agar well
diffusion method
Te st organism Zone of Inhibition (in mm)
Hexane Chloroform Ethyl alcohol Standard
Staphylococcus aureus 15.5±0.5 16.23±0.87 16.33±0.57 23.16±0.76
Escherichia coli 11.5±0.86 12.83±0.28 15.83±0.28 11.83±0.76
Klebsiella pneumoniae 0 7.83±0.76 0 11.86±0.8
Salmonella paratyphi 0 7.5±0.5 0 14.83±076
Pseudomonas aeruginosa 13.66±0.76 13.66±0.76 14.83±0.76 12.9±0.85
The results of MIC and MBC of ethyl alcohol extract
were summarized in Table 2. Relative to the results of agar
diffusion method, the extract displayed lowest MIC and
MBC value of 12.5 and 25 mg ml
-1
respectively against
Staphylococcus aureus. The extract produced same MIC
and MBC value of 25 and 50 mg ml
-1
against Escherichia
coli and Pseudomonas aeruginosa. The results are equi-
table to the results presented by Panghal etal. in which
the organic extracts prepared from the leaves of M.
koenigii manifested slightly better MIC values against
the clinical isolates (Panghal et al., 2011). Zinc oxide
Table 2. Minimum Inhibitory Concentration and
Minimum Bactericidal Concentration of M. koenigii
organic extracts
Test organism MIC (mg ml
-1
) MBC (mg ml
-1
)
Test Standard Test Standard
Staphylococcus
aureus
12.5 3.12 25 6.25
Escherichia coli 25 25 50 50
Pseudomonas
aeruginosa
25 25 50 50

BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS ANTIBACTERIAL AND ANTIOXIDANT ACTIVITY OF LEAF ORGANIC EXTRACTS OF LOCAL CULTIVARS 363
M. Kavitha
nanoparticles synthesized using the leaf extract of M.
koenigii displayed almost similar results to the present
study (Elumalai et al., 2015). Antioxidant protein iso-
lated from the leaves of M. koenigii also exhibited com-
parable results (Ningappa etal., 2010). The variation in
the antimicrobial activity of M. koenigii organic extracts
speci ed in various studies may be due to the differences
in chemical composition which can result from the cli-
matic differences among locations (Paul, 2015). Though
several compounds like tocopherol, -carotene, lutein
and alkaloids (Khanum etal., 2000) are reported in the
leaf organic extracts, carbazole alkaloids are accounted
for the antimicrobial activity (Ramsewak et al.,
1999).
The results of free radical-scavenging ability of
organic extracts of M. koenigii measured by the DPPH
assay were given in Table 3. Ethyl alcohol extract dis-
played highest radical scavenging activity with the
IC
50
value of 30 μg ml
-1
than the rest two extracts. The
radical-scavenging activity of ethyl alcohol extract is
very close to that of the standard, gallic acid exhibiting
the IC
50
value of 35 μg ml
-1
. This is in agreement with
other studies on anti-oxidant activity of leaf extracts
of M. koenigii (Rao etal., 2007; Ningappa etal., 2008;
Kusuma etal., 2011). The antioxidant activity of organic
extracts from M. koenigii is attributed to the presence of
polyphenolic compounds and carbazole alkaloids (Rao
etal., 2007; Garodia etal., 2007). Ethyl alcohol extract
exhibited greater radical-scavenging activity than the
other two extracts and this can be inferred as ethanol is
effective in extracting polyphenols (Singh etal., 2011).
CONCLUSION
As substantiated in the study, the ethyl alcohol extract
from the leaves of M. koenigii, grown locally to ful-
l culinary needs of Indian cuisine displayed excel-
lent antimicrobial activity against both Gram positive
and negative bacteria and also exhibited a quality free
radical scavenging activity. Climate of the location and
growing season greatly in uence the chemical compo-
sition of
M. koenigii and thus have an impact on the
biological activities. The above study con rmed that the
local cultivars of M. koenigii possess bene cial health
effects in terms of antimicrobial and anti-oxidant activi-
ties. Consequently, local cultivars of M. koenigii could
be effectively used as spice and dietary supplement,
however further study on molecular mechanism behind
the role of various constituents on radical scavenging
and antimicrobial inhibition needs to be done.
REFERENCES
Baskaran, C., Rathabai, V. and Kanimozhi, D. (2011) Screening
for antimicrobial activity and phytochemical analysis of vari-
ous leaf extract of
Murraya koenigii. International Journal of
Research in Ayurveda and Pharmacy, 2, 1807
–1810.
Biswas, A.K., Chatli, M.K. and Sahoo, J. (2012) Antioxidant
potential of curry (Murraya koenigii L.) and mint (Mentha
spicata) leaf extracts and their effect on colour and oxidative
stability of raw ground pork meat during refrigeration storage.
Food Chemistry, 133, 467–472.
Brind, L., Misra, A. and Srivastava, S. (2014) Evaluation of
central nervous system stimulating and analgesic activities of
Murraya koenigii leaves. Journal of Acute Medicine, 4, 81–85.
Brower, V. (1998) Nutraceuticals: Poised for a healthy slice of
the healthcare market? Nature Biotechnology, 16, 728–731.
De Smet, P.A. (1997) The role of plant derived drugs and herbal
medicines in healthcare drugs. Drugs, 54, 801–840.
Elansary, H.O., Salem, M.Z.M., Ashmawy, N.A. and Yacout,
M.M. (2012) Chemical composition, antibacterial and antioxi-
dant activities of leaves essential oils from Syzygium cumini
L., Cupressus sempervirens L. and Lantana camara L. Egyptian
Journal of Agricultural sciences, 4, 144–152.
Elumalai, K., Velmurugan, S., Ravi, S., Kathiravan, V. and
Ashokkumar, S. (2015) Bio-fabrication of zinc oxide nanopar-
ticles using leaf extract of curry leaf (Murraya koenigii) and
its antimicrobial activities. Materials Science in Semiconductor
Processing, 34, 365–372.
Gahlawat, D.K., Jakhar, S. and Dahiya, P. (2014) Murraya
koenigii (L.) Spreng: an ethnobotanical, phytochemical and
Table 3. Antioxidant activity of M. koenigii organic extracts measured in term of DPPH
radical scavenging ability
Concentration
(μg ml-1) DPPH scavenging activity (%) IC50 value (μg ml-1)
Ethyl alcohol Hexane Chloroform Gallic acid Gallic acid = 35
Ethyl alcohol = 30
Hexane = 22
Chloroform = 27
20 12 9 9 13
40 23 16 21 28
60 35 25 33 42
80 48 34 42 55
100 62 45 54 68

M. Kavitha
364 ANTIBACTERIAL AND ANTIOXIDANT ACTIVITY OF LEAF ORGANIC EXTRACTS OF LOCAL CULTIVARS BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
pharmacological review. Journal of Pharmacognosy and Phy-
tochemistry, 3, 109–119.
Garodia, P., Ichikawa, H., Malani, N., Sethi, G. and Aggar-
wal, B.B. (2007) From ancient medicine to modern medicine:
ayurvedic concepts of health and their role in in ammation
and cancer. Journal of the Society for Integrative Oncology,
5, 25–37.
Harish, P. (2001) Herbal drugs. Current Science India, 81, 15.
Kamboj, V.P. (2000) Herbal medicine. Current Science India,
78, 35–39.
Khanum, F., Anilakumar, K.R., Sudarshan Krishna, K.R.,
Viswanathan, K.R. and Santhanam, K. (2000). Anticarcino-
genic effects of curry leaves in dimethylhydrazine-treated rats.
Plant Foods for Human Nutrition, 55, 347–355.
Koleva, I.I.,van Beek, T.A.,Linssen, J.P.,de Groot, A. and Evs-
tatieva, L.N. (2002) Screening of plant extracts for antioxidant
activity: a comparative study on three testing methods. Phyto-
chemical Analysis, 13, 8–17.
Kusuma, I.W., Kuspradini, H., Arung, E.T., Aryani, F., Min, Y.H.,
Kim, J.S., Kim, Y.U. (2011). Biological activity and phytochem-
ical analysis of three Indonesian medicinal plants, Murraya
koenigii, Syzygium polyanthum and Zingiber purpurea. Jour-
nal of Acupuncture and Meridian studies, 4, 75–79.
Maa, Q.G., Xu, K., Sang, Z.P., Wei, R.R., Liu, W.M., Su, Y.L.,
Yang, J.B., Wang, A.G., Ji, T.F. and Li, L.J. (2016) Alkenes
with anti-oxidative activities from Murraya koenigii (L.)
Spreng. Bioorganic and Medicinal Chemistry Letters, 26, 799–
803.
Makri, O. and Kintzios, S. (2007) Ocimum sp. (basil): Botany,
cultivation, pharmaceutical properties, and biotechnology.
Journal ofHerbsSpices andMedicinalPlants, 13, 123–150.
Malwal, M. and Sarin, R. (2011) Antimicrobial ef cacy of Mur-
raya koenigii (Linn.) Spreng. root extracts. Indian Journal of
Natural Products and Resources, 2, 48
–51.
Molyneux, P. (2004) The use of stable free radical diphenylpic-
ryl-hydrazyl (DPPH) for estimating antioxidant activity. Song-
klanarin Journal of Science and technology, 26, 211–219.
Negi, J.S., Singh, P. and Rawat, B. (2011) Chemical constitu-
ents and biological importance of swertia: a review. Current
Research in Chemistry, 3, 1–15.
Newman, D.J. and Crag, G.M. (2007) Natural products as
sources of new drugs over the last 25 years. Journal of Natural
Products, 70, 461–477.
Ningappa, M.B., Dhananjaya, B.L., Dinesha, R., Harsha, R. and
Srinivas, L. (2010) Potent antibacterial property of APC protein
from curry leaves (Murraya koenigii L.). Food Chemistry, 118,
747–750.
Ningappa, M.B., Dinesha, R. and Srinivas, L. (2008) Antioxi-
dant and free radical scavenging activities of polyphenol-
enriched curry leaf (Murraya koenigii L.) extracts. Food Chem-
istry, 106, 720–728.
Onayade, O.A. and Adebajo, A.C. (2000) Composition of the
oil of Murraya koenigii growing in Nigeria. Journal of Herbs
Spices and Medicinal Plants, 7, 59–66.
Panghal, M., Kaushal, V. and Yadav, J.P. (2011) In vitro antimi-
crobial activity of ten medicinal plants against clinical isolates
of oral cancer cases. Annals of Clinical Microbiology and Anti-
microbials, 10, 21.
Paul, C.C., Chiedozie, O.I. and Ferdinand, N.M. (2015) Bioactive
principles from medicinal plants. Research Journal of Phyto-
chemistry, 9, 88–115.
Ramsewak, R.S., Nair, M.G., Strasburg, G.M., De Witt, D.L. and
Nitiss, J.L. (1999). Biologically active carbazole alkaloids from
Murraya koenigii. Journal of Agricultural and Food Chemistry,
47, 444–447.
Rao, L.J.M., Ramalakshmi, K., Borse, B.B. and Raghavan, B.
(2007). Antioxidant and radical-scavenging carbazole alka-
loids from the oleoresin of curry leaf (Murraya koenigii
Spreng). Food Chemistry, 100, 742–747.
Saikia, L.R. and Upadhyaya, S. (2011) Antioxidant activity,
phenol and avonoid content of some less known medicinal
plants of Assam. International Journal of Pharma and Bio-
sciences, 2, 383–388.
Singh, A.P., Wilson, T., Luthria, D., Freeman, M.R., Scott, R.M.,
Bilenker, D., Shah, S., Somasundaram, S. and Vorsa, N. (2011).
LC-MS-MS characterization of curry leaf avonols and anti-
oxidant activity. Food Chemistry, 127, 80–85.
Wayne, P.A. (2008) Performance standards for antimicrobial sus-
ceptibility testing; eighteenth informational supplement, CLSI
document M100-18, Clinical and Laboratory Standards Institute.