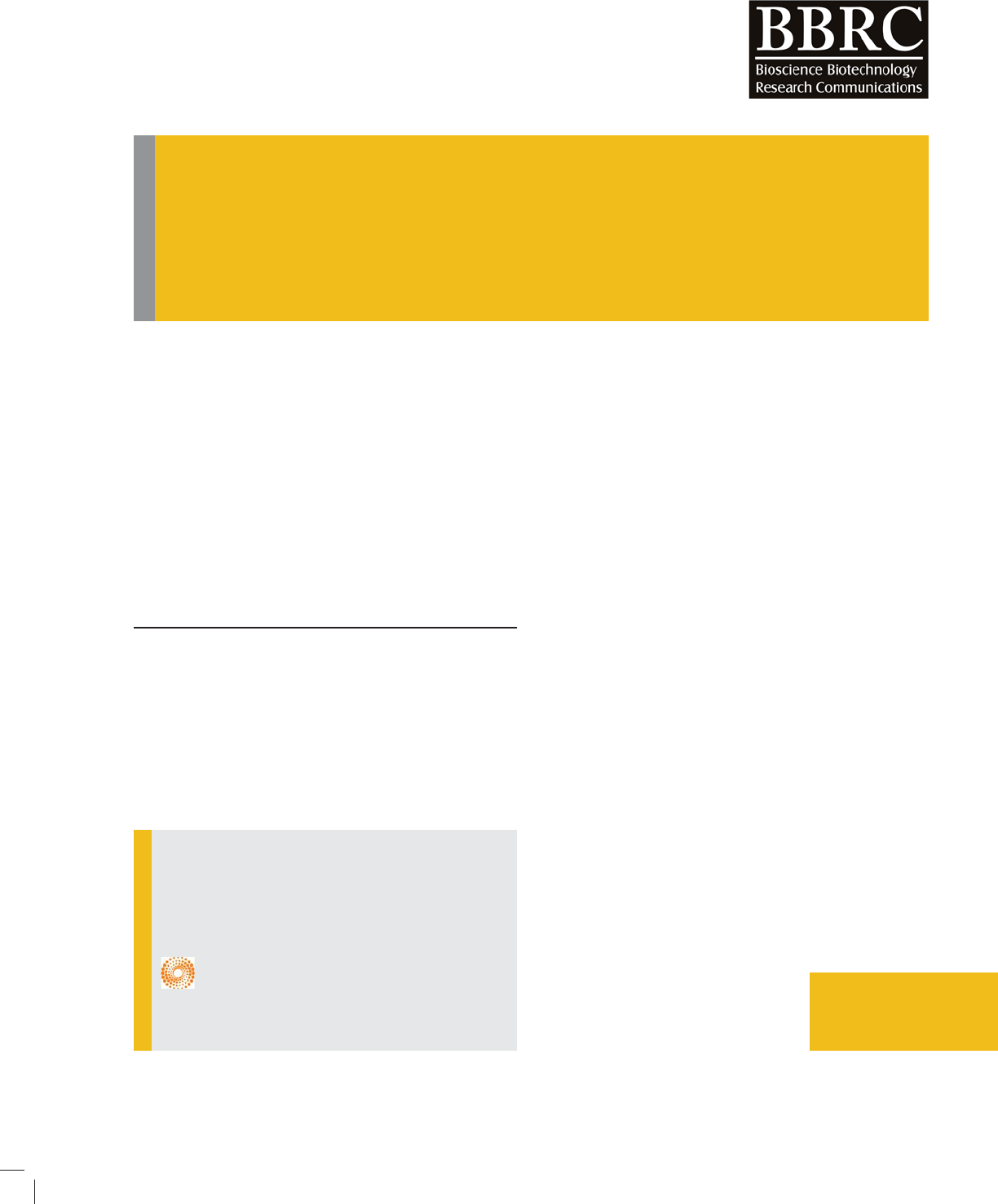
Chemical
Communication
Biosci. Biotech. Res. Comm. 9(4): 712-717 (2016)
Toluene adsorption on C
24
, Si-doped C
24
, and C
20
fullerenes
Mohammad T. Baei
1,
* and Adel Shojaei
2
1
Department of Chemistry, Azadshahr Branch, Islamic Azad University, Azadshahr, Golestan, Iran
2
Department of Physics, Behbahan Branch, Islamic Azad University, Behbahan, Iran
ABSTRACT
T he absorption feasibility of toluene molecule in the C
24
, Si-doped C
24
, and C
20
fullerenes has been studied based on
calculated electronic properties of those fullerenes using Density functional Theory (DFT). It is found that energy of
toluene adsorption upon the pure and Si-doped C
24
fullerenes were in range of -1.80 and -15.72 kJ/mol with slight
changes in their electronic structure. The results showed that the C
24
and Si-doped C
24
fullerenes cannot be used as a
chemical adsorbent or sensor for toluene molecule in nature. Also, silicon doping cannot signi cantly modify both
the adsorption energy and electronic properties of C
24
fullerene to toluene. On the other hand, toluene molecule
exhibits a high sensitivity upon C
20
fullerene, so that the energy gap of the fullerene is changed about 91.21% after
the adsorption process. We concluded that the C
20
fullerene can be served as a reliable material for toluene detection.
KEY WORDS: C
24
AND C
20
FULLERENE, TOLUENE, SENSOR, DFT STUDY
712
ARTICLE INFORMATION:
*Corresponding Author: Baei52@yahoo.com
Received 22
th
Oct, 2016
Accepted after revision 10
th
Dec, 2016
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007
Thomson Reuters ISI ESC and Crossref Indexed Journal
NAAS Journal Score 2015: 3.48 Cosmos IF : 4.006
© A Society of Science and Nature Publication, 2016. All rights
reserved.
Online Contents Available at: http//www.bbrc.in/
INTRODUCTION
Toluene, a kind of important volatile organic compound
(VOC), is used in many kinds of industries, such as paint-
ing, printing, coating, automotive, and petrochemical
industries. Emission of the toluene from these industries
causes air pollution and the environment, odor prob-
lem, ammability problem and affects human health.
Due to these toxicological effects, air contaminated with
toluene needs to be treated before it can be released
to atmosphere. Therefore, adsorption and detection of
toluene molecule has high importance in environmental
systems, (Chang et al 2000, Vandenbroucke et al 2011).
In recent years, a wide variety of investigations have
been done upon the adsorption of toluene. For example,
the adsorption of toluene on ZSM5 and mordenite zeo-
lites modi ed with Cs was investigated both theoretically
and experimentally. Toluene removal by oxidation reac-
tion in spray wet scrubber has been also studied. It has
been shown that the highest toluene removal ef ciency

Mohammad T. Baei and Adel Shojaei
was of 91.7%. Their results showed that the sequence to
prepare the catalyst affected the adsorption and plasma
catalytic of adsorbed toluene, (Chungsiriporn et al 2006,
Serra et al 2012 and Qin et al 2016).
After the synthesis of fullerene C60 by Kroto et al
(1985) fullerenes have attracted great interest because of
their physical and chemical properties and applications
in nanomaterials and biomedical science, (Akasaka and
Nagase 2002 and Muthukumar and Larsson 2008). Also,
they play a fundamental role in medical sciences, chem-
istry, biology, materials, electronics, and related elds
(Senapati et al 2004, Yoon et al 2009, Chamberlain et
al 2011).
Among the smaller fullerenes, C20 and C24 fuller-
enes are a favorable candidate for examining in mol-
ecule electronic devices, nanotechnology, and biomedi-
cal engineering. Liang Xu et al (2012) have studied the
interaction between empty C24 fullerene and the small-
est amino acid (glycine). Their results showed that the
glycine molecule is energetically favorable to interact
on the C24 fullerene through the amino nitrogen active
site. Also, orientation effects on the electronic transport
properties of C24 fullerene were studied by Wen-Kai
Zhao et al (2013) between the electrodes (Au–C24–Au).
Their ndings showed the application of the C24 fuller-
ene in the eld of single molecular devices or nanometer
electronics. Prinzbach et al (2000) have synthesized C
20
fullerene by using C
20
H
20
. They replaced the hydrogen
atoms with bromine atoms, and then debrominated to
produce C
20
fullerene in gas phase.
An et al (2011) have tried to stabilize the highly per-
fect I
h
symmetry C
20
fullerene cage by placing interstitial
atoms at the center of the fullerene using rst-principles
density functional theory (DFT). They was also investi-
gated the transport properties of C
20
fullerene and the
endohedral Li@C
20
metallofullerene coupled to three-
dimensional electrode system using DFT methods . Using
different DFT methods, the transformation processes
from the physisorption state to the chemisorption state
of a H
2
molecule in C
20
fullerene and B-doped fullerene
C
19
B system was investigated.
In previous study, we have studied the chemical func-
tionalization of C
20
fullerene with NO
2
molecule. In sum-
mary, there are few studies on the C20 and C24 fuller-
enes and further study on the structures is of important
duties. On the other hand, the doped C
20
and C
24
fuller-
enes show dramatic changes in electronic properties
with respect to their pristine. Therefore; the aim of this
study was to investigate the ability of toluene adsorp-
tion onto C
24
, Si-doped C
24
, and C
20
fullerenes, to deter-
mine whether the fullerenes are applicable for ltering
or sensing toluene molecule (An et al., 2010, Tian et al
2011 and Baei 2013).
COMPUTATIONAL METHODS
In this study, the adsorption of toluene on the C
24
, Si-
doped C
24
, and C
20
fullerenes are considered. C
24
fullerene
with a D
6
d symmetric form consisting of two 6-mem-
bered rings joined by twelve 5-membered rings and C
20
fullerene with an I
h
symmetric form consisting of 12
pentagons and 30 bonds are selected for this purpose.
DFT calculations at the level of B3LYP (Becke 1993),
with the standard 6-31G* basis set were carried out on
the C
24
, Si-doped C
24
fullerenes and the PBE (Perdew and
Ernzerhof,1996) level in GAMESS package, (Schmidt et
al 1996) with the standard 6-31G* basis set were per-
formed on the C
20
fullerene. For the C
20
fullerene, the PBE
functional show better results with respect to B3LYP
method of (Perdew and Ernzerhof, 1996). These meth-
ods were used to calculate the adsorption energy (E
ad
)
of toluene on the surface of the fullerenes using the fol-
lowing equations:
E
ad
= E
toluene/C24
– [E
C24
+ E
toluene
] +
BSSE
Eq. (1)
E
ad
= E
toluene/Si-doped C24
– [E
Si-doped C24
+ E
toluene
] +
BSSE
Eq. (2)
E
ad
= E
toluene/C20–
[E
C20
+ E
toluene
] +
BSSE
Eq. (3)
Where E
toluene/C24
and E
toluene/C20
are the total energy of
complex of adsorbed toluene on the C
24
and C
20
fuller-
enes. E
C24
, EC
20
, and E
toluene
are the total energy of the
pure C
24
and C
20
fullerenes and toluene molecule. E
toluene/
Si-doped C24
is the total energy of complex of adsorbed tolu-
ene on the Si-doped C
24
fullerene. E
Si-doped C24
is the total
energy of Si-doped C
24
fullerene, and
BSSE
is the BSSE
correction. The negative values of E
ad
reveal that the
adsorption is exothermic. For the structures, the geom-
etry optimization, natural bond orbital (NBO)
density
of states (DOS), energies, and frontier molecular orbital
(FMO) were calculated according to Glendening et al
(1998). The energy gap (E
g
) of the optimized structures
was obtained by the energy difference between the high-
est occupied molecular orbital (HOMO) and the lowest
un-occupied molecular orbital (LUMO). Also, Fermi level
energy (E
FL
) and work function (
) of the considered
structures are calculated.
RESULTS AND DISCUSSION
The optimized structure of C
24
, Si-doped C
24
, and C
20
fullerenes are shown in Fig. 1. The C-C bond lengths of
pure C
24
and C
20
fullerenes are in the range of 1.36-1.53
and 1.44-1.51 Å. The electronic property analysis based
on DOS shows a HOMO-LUMO gap (E
g
) of 1.82, 1.63,
and 0.74 eV for the C
24
, Si-doped C
24
, and C
20
fullerenes,
respectively. E
g
of the C
24
and C
20
fullerenes are very
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS TOLUENE ADSORPTION ON C
24
, SI-DOPED C
24
, AND C
20
FULLERENES 713
Mohammad T. Baei and Adel Shojaei
close to the results of Liang Xu et al (2012) and Kumar
et al (2011) respectively. Change of E
g
of the Si-doped
C
24
fullerene is about 10.44% with respect to the pristine
model, suggesting that the electronic properties of C
24
fullerene is not very sensitive on the Si adsorption.
TOLUENE ADSORPTION ON THE C
24
, SI-DOPED
C
24
, AND C
20
FULLERENES
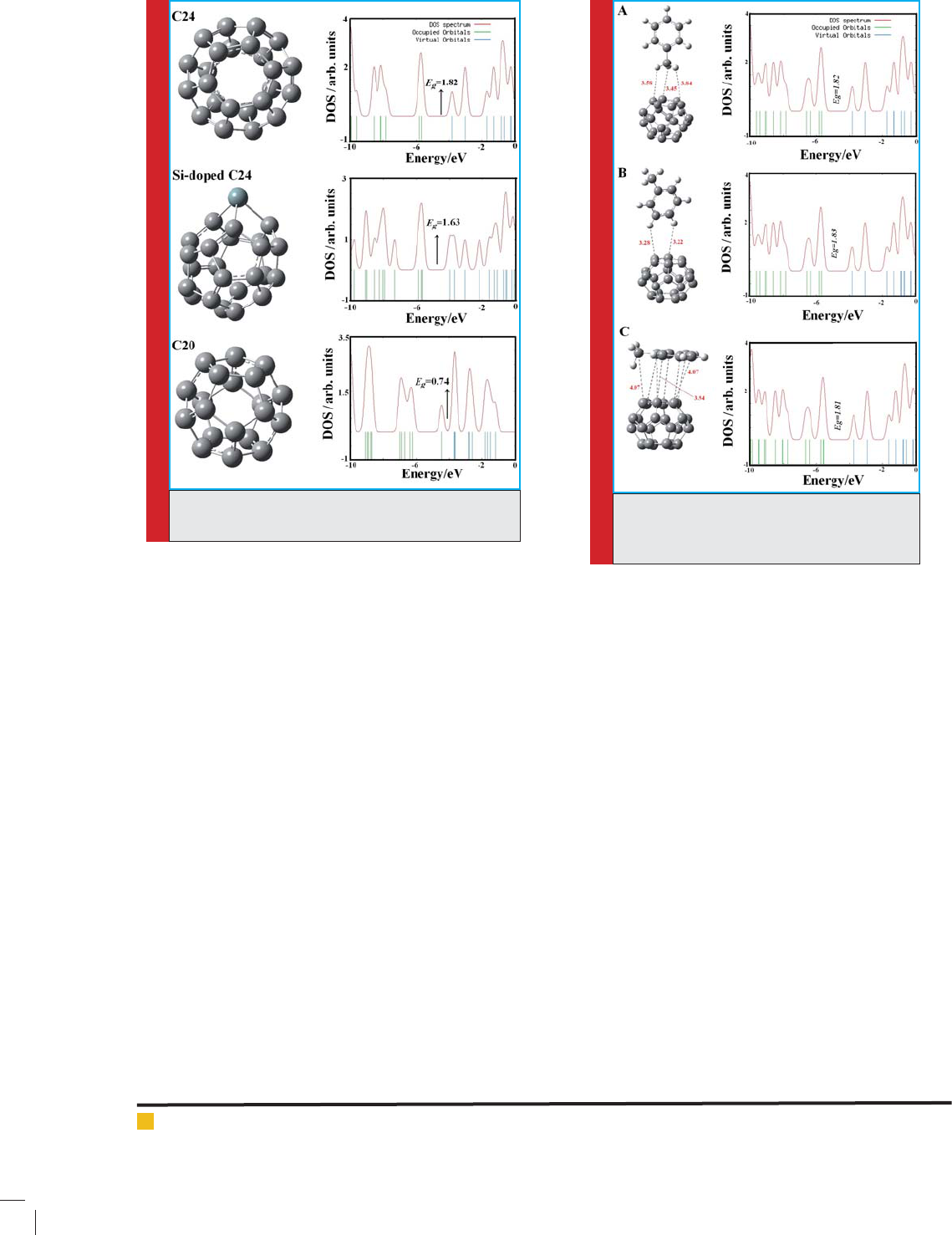
First, we computed the optimized structures of the indi-
vidual toluene molecule and C24, Si-doped C24, and C20
fullerenes. Then, for investigation of toluene adsorp-
tion on the out surface of the fullerenes, several pos-
sible initial adsorption geometries including the parallel
and perpendicular orientations of toluene molecule close
to the surface of the fullerenes are considered. After
full structural optimization without any constraints,
the three most stable structures are obtained upon the
relaxation processes for toluene adsorption on the C
24
fullerene which are shown in Figs. 2 and their electronic
properties are shown in Table 1.
In Con gurations A and B in Fig. 2, the hydrogen
atoms of toluene is interacted perpendicular to 5 and
6-membered rings of the C atoms of the fullerene with
the minimum distance of about 3.45 to 3.22 Å, respec-
tively. Calculated E
ad
values of the con gurations are
about -1.80 and -3.22 kJ/mol, respectively and a maxi-
mum NBO charge of 0.01|e| is transferred from the
fullerene to the toluene. The structural parameters of
the con gurations upon the adsorption process remain
unchanged. The results show that the Con gurations
have a weak interaction between toluene molecule and
the C
24
fullerene. Also, in the con gurations, the in u-
ence of toluene adsorption on the electronic properties
of the fullerene was investigated and almost remains
unchanged. HOMO and LUMO energies, energy gap (E
g
),
Fermi level energy (E
FL
), and work function (
) of the
con gurations are shown in Table 1.
The E
FL
in a molecule is approximately middle of E
g
and
for a semiconductor is de ned as the energy dif-
ference between the E
FL
and the LUMO [24] which is
important in eld emission applications. The important
sensing mechanisms in nanostructure devices is change
of E
g
the nanostructure and subsequently change of its
conductivity upon the adsorption process as per Zhou
et al (2010). Therefore, it is very important to calculate
the DOS of the C
24
fullerene in the con gurations before
and after toluene adsorption. DOS for the con gurations
is shown in Fig. 2. In comparison with the pure model,
FIGURE 1. Optimized Structures of C
24
, Si-doped C
24
,
and C
20
fullerenes and their density of states (DOS).
FIGURE 2. Different models of toluene adsorp-
tion on the C
24
fullerene and their density of
states (DOS). Distances are in Å.
714 TOLUENE ADSORPTION ON C
24
, SI-DOPED C
24
, AND C
20
FULLERENES BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS

Mohammad T. Baei and Adel Shojaei
FIGURE 3. Toluene adsorption and its density of states (DOS) on
the Si-doped C24 fullerene.
their E
g
values remain almost unchanged (changed by
about 0.55 %). Also, for the con gurations, Fermi level
energy (E
FL
), and work function (
) values remain almost
unchanged (changed by about 1.10 %). The results indi-
cate that the toluene adsorption via these con gurations
has no sensible effects on the electronic properties of the
fullerene.
In Con gurations C in Fig. 2, the hydrogen atoms of
toluene is interacted parallel to 5 and 6-membered rings
of the C atoms of C
24
fullerene with the minimum dis-
tances of about 3.94 Å. The E
ad
value of the con gura-
tion is about -3.22 kJ/mol. In comparison with the pure
C
24
fullerene, its E
g
, E
FL
, and
values remain almost
unchanged (see Table 1).
The above results show that C
24
fullerene cannot be a
potential ef cient adsorbent or sensor for adsorption or
determine of toluene from environments systems. There-
fore, to solve this problem, introduced various func-
tional groups and or the doping methods, which enables
chemical covalent bonding between the fullerene and
foreign atoms or molecules. Pure silicon can be doped
with other elements to adjust its electrical response by
controlling the number and charge of current carriers.
Such control is necessary for transistors (Cui and Lieber
(2001), solar cells, semiconductor detectors, and other
semiconductor devices which are used in electronics and
other high-tech applications.
Therefore, doping of C
24
fullerene by Si atoms may
be able to yield changes in the interactions between the
fullerene and foreign atoms or molecules. Fig. 3 shows
adsorption con guration of toluene molecule and its
density of states (DOS) on the C
24
fullerene doped with Si
atom. The E
ad
value of the con guration is about -15.72
kJ/mol, which is stronger than that in the pure C
24
states.
Nevertheless, the adsorption con guration is in the
range of physisorption and cannot be used as potential
ef cient adsorbent for adsorption of toluene. For fur-
ther study, the changes of E
g
, E
FL
, and
of the con-
guration is shown in Table 1. The values do not show
notable changes. Therefore, the C
24
fullerene doped with
Si atom cannot be a potential ef cient sensor for deter-
mine of toluene molecule. Also, Silicon doping cannot
Table 1: Adsorption energy (Ead) of toluene on C
24
, Si-doped C
24
, and C
20
fullerenes, HOMO energies (EHOMO), LUMO energies
(ELUMO), HOMO–LUMO energy gap (Eg), Fermi level energy (EFL), and work function (
) for the studied systems.
Structure Ead(kJ/mol) EHOMO(eV) ELUMO(eV) Eg(eV) aΔEg(%) bQT|e| EFL(eV) (eV) cΔ%
Toluene - -6.40 0.14 6.54 - - -3.13 3.27 -
C
24
- -5.64 -3.82 1.82 - - -4.73 0.91 -
Fig. 2A -1.80 -5.68 -3.86 1.82 0.00 -0.01 -4.77 0.91 0.00
Fig. 2B -3.22 -5.68 -3.85 1.83 0.55 -0.01 -4.76 0.92 1.10
Fig. 2C -3.18 -5.57 -3.76 1.81 0.55 0.00 -4.66 0.90 1.10
Si-doped C
24
- -5.62 -3.99 1.63 - - -4.80 0.82 -
Fig. 3 -15.72 -4.38 -3.02 1.36 -16.53 -0.01 -3.70 0.68 17.04
C
20
- -4.45 -3.71 0.74 - - -4.08 0.37 -
Fig. 4A -6.26 -4.49 -3.76 0.73 0.72 0.00 -4.12 0.37 0.00
Fig. 4B -60.16 -4.60 -3.18 1.41 91.21 0.17 -3.89 0.70 91.21
a
The change of Eg of C
24
, Si-doped C
24
, and C20 fullerenes after toluene adsorption
b
QT is de ned as the total natural bond orbital charges on the toluene molecule (positive values show charge transfer from toluene molecule to the
fullerenes)
c
The change of work function of C
24
, Si-doped C
24
, and C
20
fullerenes upon toluene adsorption
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS TOLUENE ADSORPTION ON C
24
, SI-DOPED C
24
, AND C
20
FULLERENES 715
Mohammad T. Baei and Adel Shojaei
signi cantly improve both the adsorption energy and
electronic properties of C
24
fullerene to toluene.
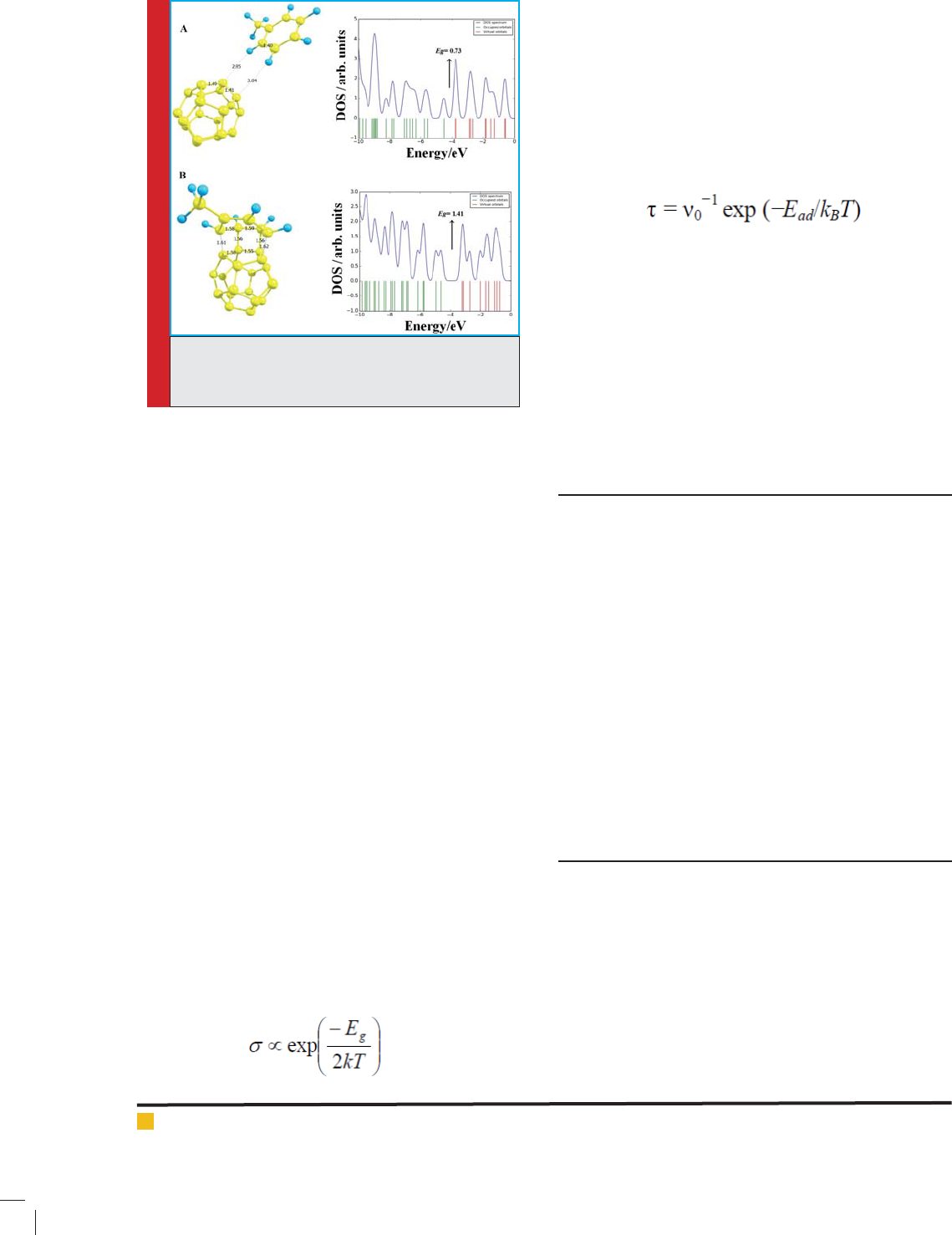
In the next step, the in uence of the toluene adsorp-
tion on the electronic properties of the C
20
fullerene was
studied. After full structural optimization, the two most
stable structures are obtained upon the relaxation pro-
cesses which are shown in Fig. 4. In Con gurations A
and B in Fig. 4, the toluene is interacted perpendicular
and parallel to surface of the fullerene. The E
ad
value of
toluene in Fig. 4A is -6.26 kJ/mol (a weak interaction)
and in comparison with the pure C
20
fullerene, its E
g
, E
FL
,
and
values remain almost unchanged (see Table 1).
The results showed that in this con guration, C
20
fuller-
ene cannot be a potential ef cient adsorbent or sensor
for adsorption or determine of toluene molecule. How-
ever, the E
ad
value of toluene in Fig. 4B is -60.16 kJ/
mol and also, the results show that the toluene adsorp-
tion through this con guration has sensible effects on
the electronic properties of the fullerene. In this state,
the E
g
and
values are changed about 91.21% after the
toluene adsorption. The important sensing mechanisms
in nanostructure devices is change of E
g
the nanostruc-
ture and subsequently change of its conductivity upon
the adsorption process, (Zhou et al 2010). Therefore, the
presence of the toluene molecule can be detected by
computing the conductivity change of the C
20
fullerene
before and after the toluene adsorption. This behavior
can be explained according to the following equation,
(Li 2006).
Eq. (4)
Where is the electric conductivity of the structure
and k is the Boltzmann’s constant. According to equa-
tion (4), smaller E
g
at a special temperature leads to the
larger electric conductivity. Therefore, the considerable
change in E
g
of the C
20
fullerene shows the high sensitiv-
ity of electronic properties of C
20
fullerene towards the
toluene molecule. Also, it is well known that one of the
most important factors in sensor devices is their recov-
ery time () that can be described as:
Eq. (5)
Where k
B
is the Boltzmann’s constant, T is the tempera-
ture, and
0
is the attempt frequency. According to equa-
tion (5), more negative E
ad
will prevent the recovery of
the device. In other words, very strong interactions are
not favorable in sensor devices due to long recovery
times (). However, the E
ad
value of the con guration
is -60.16 kJ/mol (Table 1) that is not too large to hinder
the recovery of the fullerene. The results show the high
sensitivity of the fullerene towards toluene molecule and
can be used as toluene sensor.
CONCLUSION
We have investigated the toluene adsorption on C
24
, Si-
doped C
24
, and C
20
fullerenes using DFT calculations. The
results show that toluene molecule presents a weak phys-
ical adsorption with the pure and Si-doped C
24
fullerene
and the fullerenes are not a suitable adsorbent for tolu-
ene molecule. In addition, the results suggest that the
pure and Si-doped C
24
fullerene has low sensitivity to the
presence of toluene and the in uence of toluene adsorp-
tion on the electronic properties of the fullerenes remain
almost unchanged. On the other hand, toluene molecule
exhibits a high sensitivity upon C
20
fullerene, so that the
energy gap of the fullerene is changed about 91.21%
after the adsorption process. The results show the high
sensitivity of C
20
fullerene towards toluene molecule and
therefore can potentially be used for toluene sensors.
ACKNOWLEDGEMENTS
The authors wish to acknowledge the nancial sup-
port received from Islamic Azad University, Azadshahr
Branch.
REFERENCES
AkasakaT.S. Nagase A. (2002) New, Family of Carbon Clusters,
Kluwer Academic Publisher, Dordrecht, The Netherlands
An YP., C. L. Yang, M. S. Wang, X. G. Ma, D. H. Wang (2010)
First-principles study of transport properties of endohedral
FIGURE 1. Adsorption con gurations of toluene
molecule on the C
20
fullerene and their density of
states (DOS).
716 TOLUENE ADSORPTION ON C
24
, SI-DOPED C
24
, AND C
20
FULLERENES BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS

Mohammad T. Baei and Adel Shojaei
Li@C
20
metallofullerene, Current Applied Physics, 2010, 10,
260–265
An YP., C. L. Yang, M. S. Wang, X. G. Ma, D. H. Wang (2011)
Geometrical and Electronic Properties of the Clusters of C
20
Cage Doped with Alkali Metal Atoms, J. Clust. Sci.22, 31–39
Baei MT (2013) First-Principles Study of NO
2
Adsorption on C
20
Fullerene, Heteroatom Chemistry 24 (6), 516–522
Becke AD (1993) The role of exact exchange J. Chem. Phys.,
1993, 98, 5648
Chang C, C. Lee, Y. Wu, and F. Jeng (2000) Assessment of the
strategies for reducing volatile organic compound emissions
in the automotive industry in Taiwan, Resour, Conserv. and
Recycl, 34 117-128
Chungsiriporn J., C. Bunyakan, R. Nikom (2006) Toluene
removal by oxidation reaction in spray wet scrubber: experi-
mental, modeling and optimization, J. Sci. Technol., 28(6)
1265-1274
Cui Y., C. M. Lieber (2001) Highly polarized assembled using
silicon nanowire building blocks Science, 291, 851
Glendening ED., A.E. Reed, J.E. Carpenter, F. Weinhold,
NBO(1998) Version 3.1 TCI, University of Wisconsin, Madison
Kim C., B. Kim, S.M. Lee, C. Jo, Y.H. Lee (2002) Electronic
structures of capped carbon nanotubes under electric elds,
Phys. Rev. B. 65, 165418.
Kroto, HW., J.R. Heath, S.C. O’Brien, R.F. Curl, R.E. Smalleym
(1985) C60: Buckmin- sterfullerene, Nature (London), 318, 162
Kumar R., A. Rani, Structure and electronic properties of H
n
@
C
20
molecule (2011) Physica B 406, 1173–1177
Li S. (2006) Semiconductor Physical Electronics, 2nd ed.,
Springer, USA,
Liang Xu, Li Chao, Li Feng, Li Xiaojun, Tao Shuqing (2012)
Spectrochimica Acta Part A: Molecular and Biomolecular
Spectroscopy, 98, 183–189
Muthukumar K.. J. A.Larsson (2008) Explanation of the dif-
ferent preferential binding sites forCeandLainM2@C80(M¼Ce,
La)–a density functional theory prediction, J. Mater. Chem. 18,
3347
Perdew JP., K. Burke, M. Ernzerhof (1996) Generalized gradi-
ent approximation made simple, Physical Review Letters 77,
3865–3868
Prinzbach H., A. Weiler, P. Landenberger, F. Wahl, J. Worth,
L.T. Scott, M. Gelmont, D. Olevano, B. Issendorff (2000) Gas-
phase production and photoelectron spectroscopy of the small-
est fullerene, C
20
, Nature, 407, 60-63
Qin C., C., X. Huang, X. Dang, J. Huang, J. Teng, Z. Kang
(2016) Toluene removal by sequential adsorption-plasma cata-
lytic process: Effects of Ag and Mn impregnation sequence on
Ag-Mn/-Al
2
O
3
Schmidt M., K. Baldridge, J. Boatz, S. Elbert, M. Gordon, J.
Jensen, S. Koseki, N. Matsunaga, K. A. Nguyen, S. Su, T.L.
Windus, M. Dupuis, J.A. Montgomery Jr (1993) J. Comput.
Chem., 1993 14, 1347
Senapati L., J. Schrier, K. B. Whaley(2004) Electronic Trans-
port, Structure, and Energetics of Endohedral Gd@C
82
Metal-
lofullerenes Nano Lett. 4, 2073–2078
Serra RM E. E. Miró, P. Bolcatto, A. V. Boix (2012) Experimen-
tal and theoretical studies about the adsorption of toluene on
ZSM5 and mordenite zeolites modi ed with Cs, Microporous
and Mesoporous Materials, 147 17–29
Tian C., K Fen Zhao, M. Shan Wang K (2011)Transformation
mechanism of a H
2
molecule from physisorption to chemisorp-
tions in pristine and B-doped C
20
fullerenes, Chemical Physics
Letters 511, 393–398
Vandenbroucke, AM R. Morent, N. De Geyter (2011)
Non-
thermal plasmas for non-catalytic and catalytic VOC
abatement. J. Hazard. Matter. 195 30-54.
Yo on M., S. Yang, Z. Zhang (2009) J. Chem. Phys., 2009,
131(6), 64707. Chamberlain TW, N. R. Champness, M. Schrder,
A. N. Khlobystov (2011) Chem: Eur. J., 2011, 17, 668–
674
Zhao WK., Chuan-Lu Yang, Jing-Fen Zhao, Mei-Shan Wang,
Xiao-Guang Ma (2012) Physica B, 2012, 407, 2247–2253
Zhou X., W.Q. Tian, X.-L. Wang (2010) Adsorption sensitiv-
ity of Pd-doped SWCNTs to small gas molecules, Sensors and
Actuators B: Chemical 151, 56–64.
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS TOLUENE ADSORPTION ON C
24
, SI-DOPED C
24
, AND C
20
FULLERENES 717