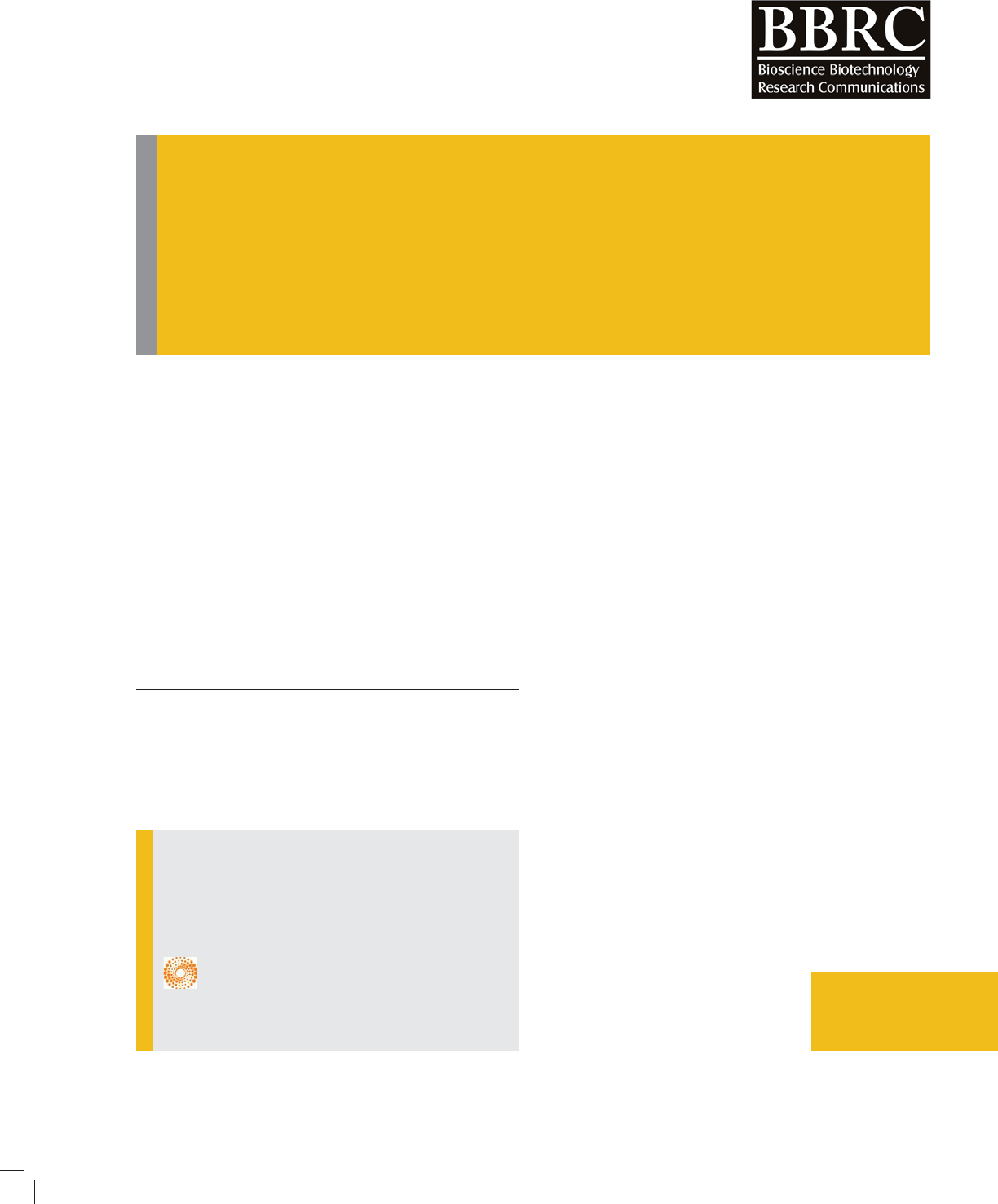
Medical
Communication
Biosci. Biotech. Res. Comm. 9(4): 643-652 (2016)
A common transition in multi-drug resistance gene and
risk of breast cancer: A genetic association study with
an in silico-analysis
Davood Kheirkhah
1
and Mohammad Karimian
2
*
1
Department of Pediatrics, Kashan University of Medical Sciences, Kashan
2
Anatomical Sciences Research Center, Kashan University of Medical Sciences, Kashan
ABSTRACT
We have investigated the correlation of multi-drug resistance- c.3435T>C common gene transition with breast cancer
risk in Asian populations by an updated meta-analysis which followed by an in silico approach. In a meta-analysis
approach, we collected all related studies. For this purpose, we used common electronic databases such as PubMed,
Google Scholar and Science Direct. We employed bioinformatics to evaluate molecular effects of c.3435T>C transi-
tion. Our data revealed that there is no signi cant association between the polymorphism and breast cancer within
Asian population. But, strati ed meta-analysis revealed that there are a signi cant associations within T vs. C
(OR=1.40, 95%CI=1.14-1.73, P=0.002), TT vs. CC (OR=1.85, 95%CI=1.15-2.98, P=0.011), and TT vs. CC+CT (OR=1.59,
95%CI=1.06-2.37, P=0.024) genetic models. Also, bioinformatics data revealed that c.3435T>C polymorphism could
affect splicing pattern. Based on results, the c.3435T>C transition with effects on RNA splicing pattern is a risk factor
for breast cancer in Iranian populations.
KEY WORDS: BREAST CANCER, GENETIC POLYMORPHISM, RISK FACTOR, META-ANALYSIS
643
ARTICLE INFORMATION:
*Corresponding Author: mdkarimian@yahoo.com
Received 11
th
Nov, 2016
Accepted after revision 26
th
Dec, 2016
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007
Thomson Reuters ISI ESC and Crossref Indexed Journal
NAAS Journal Score 2015: 3.48 Cosmos IF : 4.006
© A Society of Science and Nature Publication, 2016. All rights
reserved.
Online Contents Available at: http//www.bbrc.in/
INTRODUCTION
Breast cancer is the most prevalent cancer in women
all over the world, and its occurrence is rising. It is a
multifactorial disease that is caused by complicated
interactions among genetic and environmental factors,
(Jemal et al., 2006 and Nickels et al., 2013). Lately, a
growing amount of research have been focused to assess
the association among genetic factors and breast can-
cer susceptibility. Moreover, individual genetic varia-
tions, especially genetic polymorphisms in enzymes that
metabolize drugs have an important role in metabo-
lism and the fate of drugs. Also, some studies have
showed that genetic polymorphisms of the multi-drug

644 MDR1 GENE AND BREAST CANCER BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Davood Kheirkhah and Mohammad Karimian
resistance1 (MDR1; ABCB1) gene, may be correlated
with cancer risk (Turgut et al., 2007). The MDR1 gene
that encodes a membrane-bound phosphoglycoprotein
(P-gp), is positioned on chromosome 7 with 29 exons.
It functions as an ef ux pump which protects the cells
against numerous elements including amino acids, pro-
teins, organic cations, and others, (Kreile et al., 2013,
Ozdemir et al., 2013, Lee, 2016 and Isvoran et al., 2016 ).
More than 50 variants exist in the MDR1 gene.
Among these, three following single nucleotide poly-
morphisms (SNPs) are more common than others: (i)
C1236T (rs1128503), (ii) G2677T (rs2032582), and (iii)
C3435T (rs1045642; c.3435T>C). Some previous reports
reported that c.3435T>C single nucleotide polymor-
phism in MDR1 gene may contribute in breast cancer
risk (Johnatty et al., 2013 and Tazzite et al., 2016).
In this study, we have investigated the association of
aforementioned transition with breast cancer risk within
Asian populations by a meta-analysis and in silico
approach.
MATERIAL AND METHODS
The literature search was done by both two authors of
this paper (DK and MK). Standard electronic databases
such as PubMed, Google Scholar, and Science Direct
were explored for eligible papers up to Dec, 2016. Key-
words and phrases which used for our search strategy
were as follow: (“multi-drug resistance” or “MDR” or
“ABCB1” or “C3435T” or “rs1045642”), and (“genetic
polymorphism” or “SNP” or “variant” or “single nucleo-
tide polymorphism”) and (“breast cancer”). Citations in
possible eligible papers were also studied as a subsidiary
source to recognize more eligible papers.
Included studies in the meta-analysis were chosen
based on the following criteria: (1) the paper evaluated
the association between the MDR1- c.3435T>C poly-
morphism and breast cancer risk; (2) The total number
of participants in the case and control groups, the geno-
type distribution, or other relevant information could be
extracted from the paper. (3) Paper published in English
language. The exclusion criteria for meta-analysis were
as follow: (1) publication was review, meta-analysis, let-
ter to the editor, or abstract; (2) the paper introduced
either vague information or did not prepare frequencies
of genotypes ; (3) the paper related to ethnicities except
than Asians.
The information from included papers were extracted
by two authors independently (DK and MK). Differences
concerning study selection and data extraction were set-
tled by a discussion. We extracted the following infor-
mation from eligible studies: rst author’s name, publi-
cation year, country, genotyping methods, frequencies of
genotypes in case and control groups, and estimation of
Hardy-Weinberg equilibrium (HWE) in control groups.
At rst, Hardy-Weinberg equilibrium (HWE) for
control group of each study were calculated by a Chi-
squared test. This calculation was performed by had-
2know online web server. A two-tailed p value less than
0.05 (p<0.05) was considered as a statistically signi -
cant difference. All statistical meta-analysis were done
by Comprehensive Meta Analysis program (Biostat, Inc.,
Englewood, NJ, USA) and The Open Meta Analyst soft-
ware (Tufts University, Medford, MA, USA). Pooled odds
ratios (ORs) with 95% con dence intervals (CIs) were
estimated to assess the association of MDR1- c.3435T>C
polymorphism and breast cancer risk. Tests were per-
formed under the following models: 1- Allelic (T vs. C
allele), 2- Homozygous (TT vs. CC), 3- Heterozygous (CT
vs. CC), and 4- Dominant model (TT+CT vs. CC), and 5-
Recessive model (TT vs. CT+CC). The Cochrane Q-test and
I2 score were employed to estimate heterogeneity and
apvalue more than 0.1 was considered as statistically
signi cant. Fixed-effect and random-effects model were
used in the absence and presence of heterogeneity of the
studies, respectively. Also, a strati ed meta-analysis was
performed in Iranian populations. Publication bias was
discovered by funnel plots and Egger’s test (Begg and
Mazumdar, 1994; Egger et al., 1997). Sensitivity analysis
was done to evaluate the magnitude effect of individual
studies on the overall analysis.
IN SILICO ANALYSIS
For bioinformatics analysis, the entire gene sequence of
MDR1 was deduced from NCBI database (Chromosome 7,
NC_000007.14). The location of c.3435T>C SNP on the
MDR1 gene was determined manually. Also, the loca-
tion of this SNP on RNA sequence was found according
to the procedure. Since c.3435T>C mutation is a syn-
onymous SNP, the effects of this transition on protein
structure were not evaluated in the current study. But,
this polymorphism may affect the RNA structure or/and
splicing pattern of MDR1. Therefore, we used some bio-
informatics tools to evaluate of these molecular effects
of c.3435T>C on MDR1. For this purpose, RNAsnp server
was used to evaluate the effects of c.3435T>C on the
RNA structure (http://rth.dk/resources/rnasnp/).
The RNAsnp software works in three methods:
Method 1 is planned to calculate the effect of SNPs
on small RNA sequences (less than 1000 nucleotides);
Method 2 is planned to expect the in uence of SNPs on
lengthy RNA sequence while the nally Method 3 works
as a combination of method 1 and 2 and it is planned to
screen all potential structure-wrecking single nucleotide
polymorphisms in an input sequence using a brute-force
methodology. In plot summary of the RNAsnp output,
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS MDR1 GENE AND BREAST CANCER 645
Davood Kheirkhah and Mohammad Karimian
the local area which identi ed with maximum structural
alteration is highlighted giving to the p-value. If the
p-value is more than 0.2 (p> 0.2) the area is highlighted
in black that shows not signi cant structural alteration is
happened (Sabarinathan et al., 2013). The ASSP (http://
wangcomputing.com/assp/) servers was employed to
explore the impact of c.3435T>C on splice site pattern
of MDR1. ASSP is a nucleotide sequence examination
instrument for the estimation and cataloguing of splice
sites. It works based on constitutive, cryptic, skipped,
and alternative exon isoform splice sites analyses (Wang
and Marín, 2006). ASSP recognizes putative splice sites
by models which known as pre-processing models.
Finally, we used String online web server to predict the
gen-gene interaction network of MDR1.
RESULTS
After screening of studies and as shown in gure 1, total
of 7 eligible papers were included in the meta-analysis
(Tatari et al. 2009; George et al. 2009 ; Taheri et al. 2010;
Wu et al. 2012; Ghafouri et al. 2016; Abuhaliema et al.,
2016; Sharif et al., 2016). The details of included studies
in meta-analysis are given in table 1. This meta-analysis
involved 1819 breast cancer patients and 1929 healthy
controls. There were 4 studies with Iranian populations,
and 3 remained studies are related to Indian, Chinese,
and Jordanian populations. All of seven studies used
polymerase chain reaction - restriction fragment length
polymorphism (PCR-RFLP) method for c.3435T>C SNP
genotyping. Also, the distribution of genotypes in all
of control groups were consistent to Hardy–Weinberg
criteria (Table 1).
Results of association between mentioned SNP and
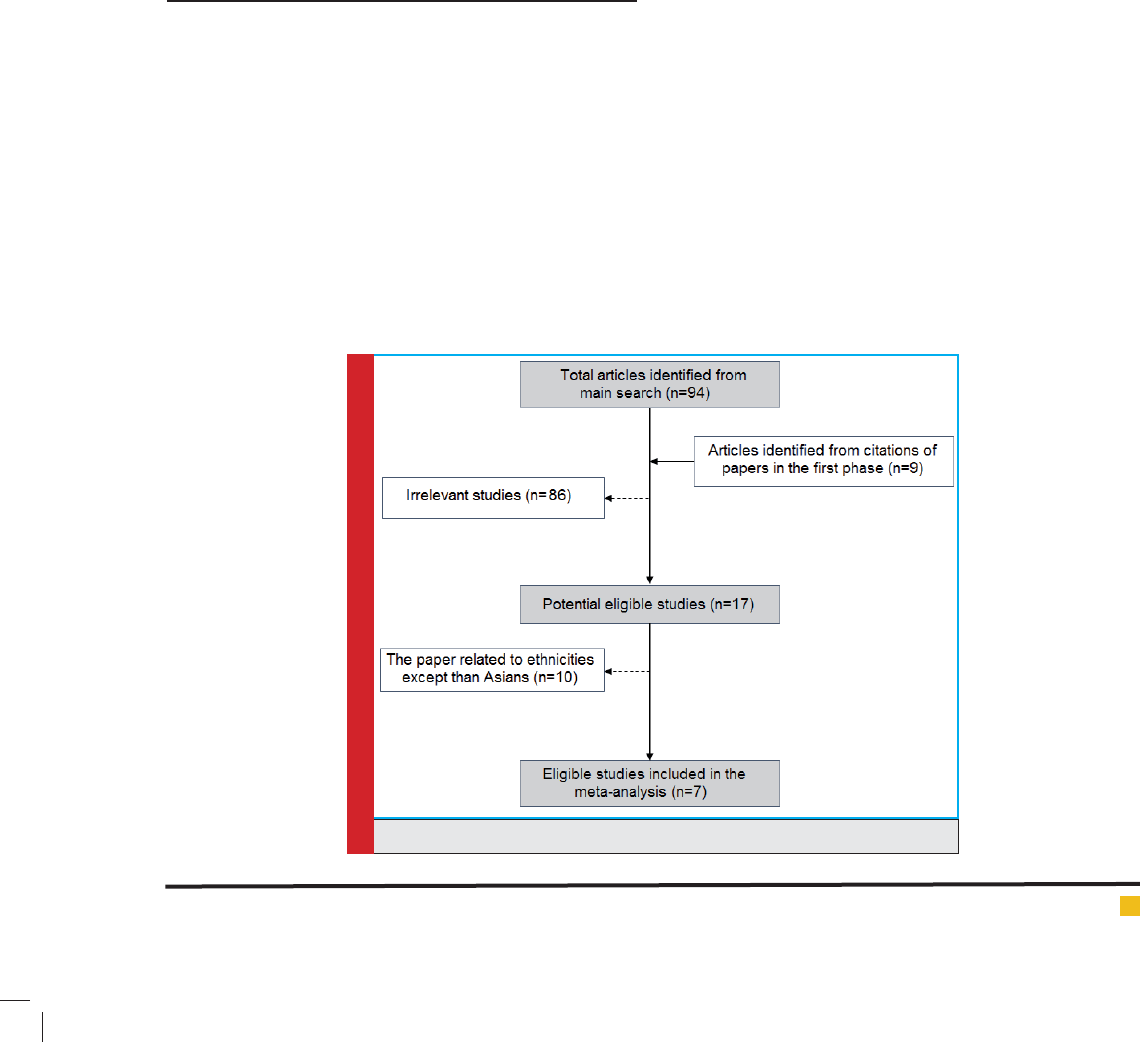
risk of breast cancer are summarized in table 2. Our
data revealed that there are no signi cant associations
between MDR1 c.3435T>C and risk of breast cancer in
Asian population under all of ve genetic models (T vs.
C: OR= 1.22, 95%CI= 0.86-1.75, P= 0.267; TT vs. CC:
OR= 1.35, 95%CI= 0.72-2.55, P= 0.353; CT vs. CC: OR=
1.00; 95%CI= 0.72-1.38; P= 0.993; CT+TT vs. CC: OR=
1.07, 95%CI= 0.73-1.56, P= 0.724; TT vs. CC+CT: OR=
1.26, 95%CI= 0.80-1.99, P= 0.323) (Figure 2). When we
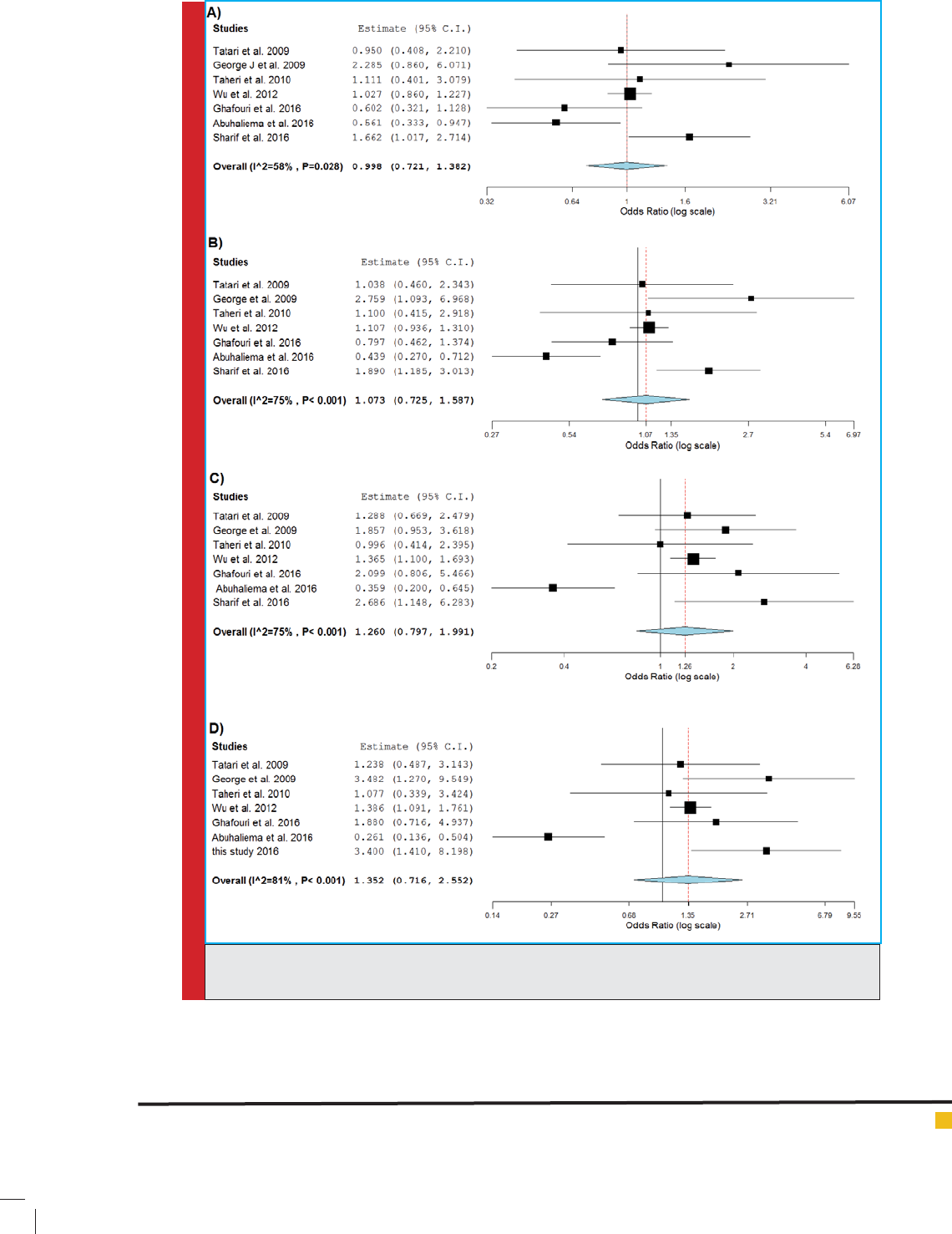
performed a strati ed meta-analysis in Iranian popula-
tions, we found a signi cant association between MDR1
c.3435T>C and breast cancer risk in Iranian populations
under three genetic models (T vs. C: OR= 1.40, 95%CI=
1.14-1.73, P= 0.002; TT vs. CC: OR= 1.85, 95%CI= 1.15-
2.98, P= 0.011; TT vs. CC+CT: OR= 1.59, 95%CI= 1.06-
2.37, P= 0.024) (Figures 3 and 4).
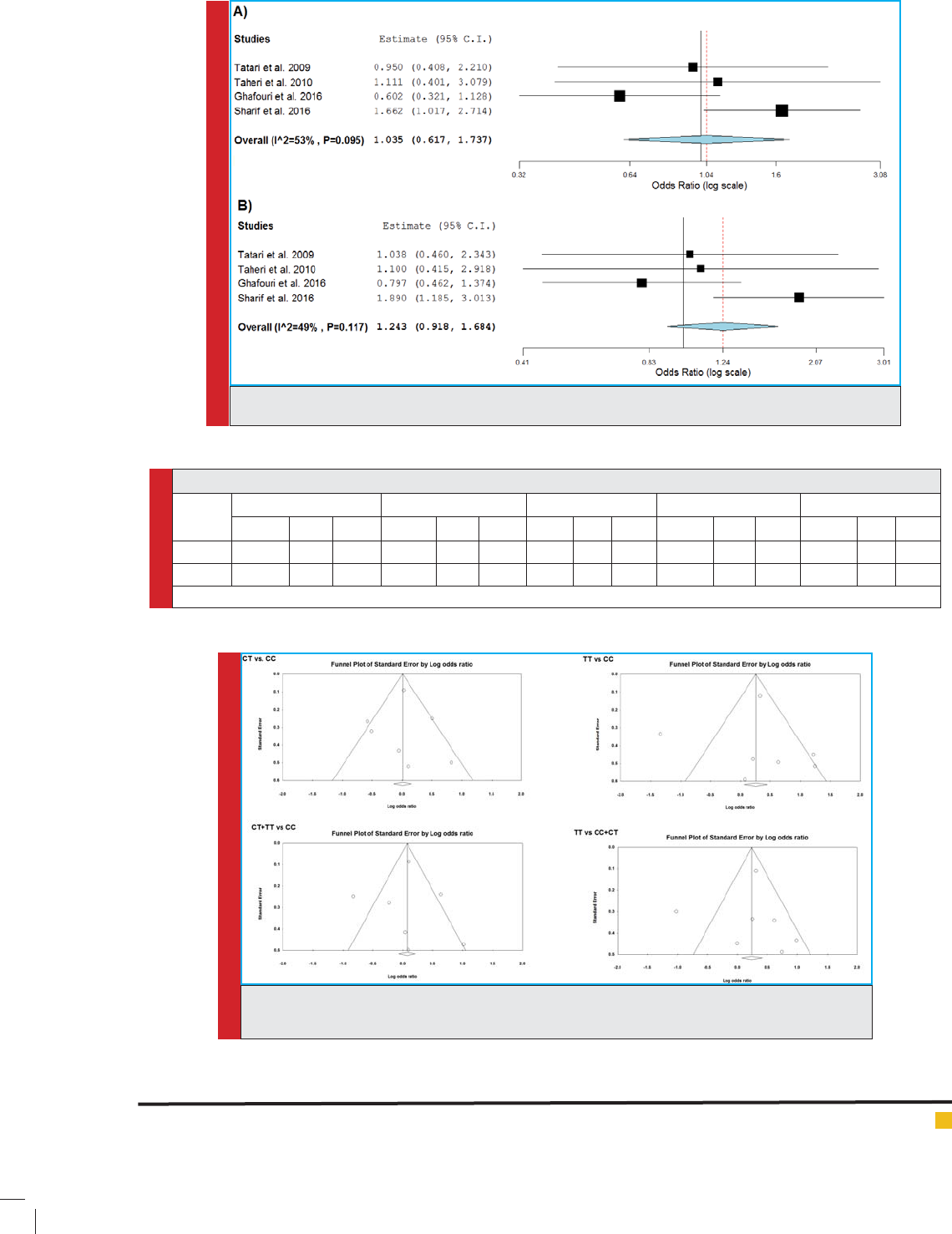
The results of heterogeneity and publication bias
are detailed in table 3. Our data revealed that there are
true heterogeneities in overall analysis under all of ve
genetic models (T vs. C: P
heterogeneity
< 0.001; I
2
=88%; TT
vs. CC: P
heterogeneity
< 0.001; I
2
=81%; CT vs. CC: P
heterogeneity
=
0.028; I
2
=58%; CT+TT vs. CC: P
heterogeneity
< 0.001; I
2
=75%;
TT vs. CC+CT: P
heterogeneity
< 0.001; I
2
=75%). Whereas we
observed only a signi cant heterogeneity in strati ed
analysis for Iranian population under CT vs. CC model
(P
heterogeneity
= 0.095; I
2
=53%). Publication bias was deter-
mined by Egger’s test and funnel plot. As shown in table
3 there are no publication biases in this meta-analysis
in overall analysis (T vs. C: P
Egger
= 0.780; TT vs. CC: P
Eg-
FIGURE 1. Study identi cation owchart.

646 MDR1 GENE AND BREAST CANCER BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Davood Kheirkhah and Mohammad Karimian
Table 1: Characteristics of included studies in meta-analysis
Country
Genotype frequencies
PHWE Genotyping method Author (Reference)Controls Cases
CC CT TT CC CT TT
Iran 12 45 20 16 57 33 0.11 PCR–RFLP Tatari et al. 2009
India 15 32 21 8 39 39 0.67 PCR–RFLP George et al. 2009
Iran 10 27 13 10 30 14 0.55 PCR–RFLP Taheri et al. 2010
China 440 624 180 388 565 220 0.08 PCR–RFLP Wu et al. 2012
Iran 141 50 9 75 16 9 0.11 PCR–RFLP Ghafouri et al. 2016
Jordan 40 65 45 68 62 20 0.10 PCR–RFLP Abuhaliema et al., 2016
Iran 79 53 8 61 68 21 0.82 PCR-RFLP Sharif et al., 2016
HWE: Hardy–Weinberg equilibrium, PCR-RFLP: Polymerase chain reaction-restriction fragment length polymorphism. A Hardy–
Weinberg equilibrium in the control group with P <0.05 did not satisfy the Hardy-Weinberg equilibrium.
Table 2: Association results of MDR1 c.3435T>C and breast cancer risk in the meta-analysis
Group T vs. C TT vs. CC CT vs. CC CT+TT vs. CC TT vs. CC+CT
OR
(95% CI)
POR
(95% CI)
POR
(95% CI)
POR
(95% CI)
POR
(95% CI)
P
Asian 1.22
(0.86-1.75)
0.267 1.35
(0.72-2.55)
0.353 1.00
(0.72-1.38)
0.993 1.07
(0.73-1.56)
0.724 1.26
(0.80-1.99)
0.323
Iranian 1.40
(1.14-1.73)
0.002 1.85
(1.15-2.98)
0.011 1.04
(0.62-1.74)
0.895 1.24
(0.92-1.68)
0.160 1.59
(1.06-2.37)
0.024
OR: odds ratio, CI: con dence interval
ger
= 0.944; CT vs. CC: P
Egger
= 0.989; CT+TT vs. CC: P
Egger
=
0.991; TT vs. CC+CT: P
Egger
= 0.947) and strati ed analy-
sis (T vs. C: P
Egger
= 0.125; TT vs. CC: P
Egger
= 0.296; CT
vs. CC: P
Egger
= 0.600; CT+TT vs. CC: P
Egger
= 0.592; TT vs.
CC+CT: P
Egger
= 0.604). This data were con rmed by sym-
metrical funnel plots (Figure 5). To assess the strength
of the association results, sensitivity analysis was done
by eliminating one study at a time and recalculating
the summary ORs. The summary ORs continued stable,
representing that our meta-analysis is not signi cantly
in uenced by an individual study (data not shown).
The data from ASSP online web server showed that
the c.3435T>C polymorphism alters splice site pattern
of the MDR1 gene (Table 4). The data revealed that the
score of constitutive donor splice site at location 515
is 6.381 for 3435TT genotype whereas this score for
3435CC genotype is 5.066. In addition Alt./Cryptic, con-
stitutive, and con dence scores for genotype at location
507 are 0.652, 0.334, and 0.487, respectively. Whereas
these values for 3435CC are 0.628, 0.335, and 0.434,
respectively. In addition, all of mentioned values are dif-
ferent between 3435TT and 3435CC genotypes at loca-
tion 515 (0.914, 0.057, and 0.937 for TT genotype and
0.820, 0.136, and 0.834 for CC genotype).
To evaluate the effects of c.3435T>C SNP on mRNA
structure of MDR1, we used the RNAsnp server. For this
purpose, we entered the entire coding sequence of the
gene into RNAsnp main window. Then we entered the
code of SNP in a section of server entitled “SNP details”.
The data from RNAsnp revealed that c.3435T>C could
not signi cantly affect RNA structure of MDR1 gene
(Folding Window=3235-3635, Local region=3434-3483,
distance=0.0965, p-value=0.2983). Also, we found that
the minimum free energy of the optimal secondary
structure of global TT genotype sequence is -113.70 kcal/
mol, while this is -114.60 kcal/mol for CC genotype.
DISCUSSION
Breast cancer is known as the most common malignancy
among women in both developing and developed coun-
tries. This disorder is a rising issue that in uences about
12.5% of women during their life. The rate of this malig-
nancy is on the growth (Slamon et al., 1987; Soleimani
et al., 2016). The occurrence of breast cancer in Asian
women is in overall lower than other ethnicity. But, all
health statistics showed that this malignancy has been
quickly growing in current decades in Asia (Matsuno et
al., 2007).
The reasons of breast cancer are weakly recognized.
Some factors including family history, age, lifestyle,
geographical variation, and genetic factors may increase
the risk of breast cancer. Genetic variations are widely
considered as important risk factors for breast cancer
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS MDR1 GENE AND BREAST CANCER 647
Davood Kheirkhah and Mohammad Karimian
FIGURE 2. Forest plot for Asian meta-analysis. A) CT vs. CC model; B) CT+TT vs. CC model; C) TT vs.
CC+CT model; D) TT vs. CC model.

Davood Kheirkhah and Mohammad Karimian
648 MDR1 GENE AND BREAST CANCER BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
FIGURE 3. Forest plot for Iranian populations. A) T vs. C model; B) TT vs. CC model; C) TT vs. CC+CT
model.
susceptibility in exact populations (Haimov-Kochman et
al., 2002). Recognition of some crucial single nucleotide
polymorphisms, which impacts the gene expression and
protein function, it might be suitable to predict suscep-
tibility of breast cancer (Lian et al., 2012).
In this study, we investigate the association of MDR1
c.3435T>C polymorphism and breast cancer risk in Asian
population by a meta-analysis which followed by a bio-
informatics approach. For example Tatari et al. (2009)
reported a signi cant association between MDR1-3435T
allele and risk of breast cancer, whereas Taheri et al.
(2010) didn’t discover any association. Therefor meta-
analysis as an in uential statistical instrument can help
researchers to nd out more precise conclusions. Our
meta-analysis revealed that there are no signi cant
association between mentioned SNP and breast cancer
risk in overall Asian analysis. But, strati ed meta-anal-
ysis in Iranian population showed a signi cant associa-
tion between MDR1 c.3435T>C transition and risk of
breast cancer in some genetic model. Also, after strati-
fying analysis we found that the heterogeneity become
disappear. Therefore, it may be a source of heterogeneity
among the studies. The different results in various stud-
ies may be due to environmental factors, genetic back-
grounds and lifestyles.
Some mechanisms explain the role of MDR1 gene in
breast cancer risk. MDR1 gene also known as MDR1 is
a member of the MDR1 superfamily that expresses P-gp
protein, which is an ATP-dependent ef ux pump that
permits the human cells to remove poisons and carcino-
genic agents (Kreile et al., 2013). Some studies proposed
that c.3435T>C transition may impact the risk of some
cancers such as breast carcinoma (Ikeda et al., 2015).
Really, this synonymous transition (Ile1145Ile) effects
Davood Kheirkhah and Mohammad Karimian
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS MDR1 GENE AND BREAST CANCER 649
FIGURE 5. Funnel plot for Asian meta-analysis. A) CT vs. CC model; B) TT vs. CC model; C) CT+TT
vs. CC model; D) TT vs. CC+CT model.
FIGURE 4. Forest plot for Iranian populations. A) CT vs. CC model; B) CT+TT vs. CC model.
Table 3: Results of heterogeneity and publication bias for the association of MDR1 c.3435T>C and breast cancer risk
Group T vs. C TT vs. CC CT vs. CC CT+TT vs. CC TT vs. CC+CT
Ph I2 Pe Ph I2 Pe Ph I2 Pe Ph I2 Pe Ph I2 Pe
Asian < 0.001 88% 0.780 < 0.001 81% 0.944 0.028 58% 0.989 < 0.001 75% 0.991 < 0.001 75% 0.947
Iranian 0.140 45% 0.125 0.335 12% 0.296 0.095 53% 0.600 0.117 49% 0.592 0.351 8% 0.604
Ph: Pheterogeneity (p< 0.1) was considered as a signi cant difference. Pe: PEgger (p< 0.05) was considered as a signi cant difference

650 MDR1 GENE AND BREAST CANCER BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Davood Kheirkhah and Mohammad Karimian
stability of protein (Fung et al., 2014) and leads to apop-
tosis modi cation or cellular damage witch these are
important for development of cancer (Johnstone et al.,
2000).
Our previous in silico studies revealed that SNPs may
affect gene expression (Jamali et al., 2016) mRNA struc-
ture (Karimian et al., 2015; Rayganet al., 2016) and pro-
tein structure and function (Karimian and Hosseinzadeh
Colagar, 2016a; Nikzad et al., 2015; Karimian and Hos-
seinzadeh Colagar, 2016b).
The SNP which was studied in this study is a syn-
onymous polymorphism, and then it has no effect on
the peptide sequence of the protein. But, it could impact
the mRNA structure or splicing pattern. So it is pos-
sible to apply bioinformatics tools to assess the harmful
properties of MDR1- c.3435T>C polymorphism on the
splicing pattern and mRNA structure. So that we used
in silico tools to evaluate these effects. Our data from
in silico analysis revealed that c.3435T>C could not
affect the mRNA structure. Though c.3435T>C substi-
tution leads to a decrease in minimum free energy for
mutant type but this change is not signi cant. Therefore,
it is expected that pathogenicity of c.3435T>C may arise
from its effect on RNA splicing.
There are some limitations in our meta-analysis that
should be mentioned. For instance, lack of original data,
such as smoking, BMI, age, and etc. may affect the accu-
racy of the association of the SNP and risk of breast
cancer. Also, this meta-analysis did not cover a large
number of Asian countries. In conclusion, the present
study suggests that the MDR1- c.3435T>C transition
might be correlated with risk breast cancer in Iranian
population. But, studies with larger sample size and con-
sidered to gene-environment and gene-gene interactions
are essential to approve our results.
REFERENCES
Abuhaliema, A.M., Yousef, A.M., El-Madany, N.N., Bulatova,
N.R., Awwad, N.M., Yousef, M.A., & Al Majdalawi, K.Z. (2016)
In uence of Genotype and Haplotype of MDR1 (C3435T,
G2677A/T, C1236T) on the Incidence of Breast Cancer-a Case-
Control Study in Jordan.Asian Paci c Journal of Cancer Pre-
vention17(1), 261-266.
Egger, M., Davey Smith, G., Schneider, M., Minder, C. (1997)
Bias in metaanalysis detected by a simple, graphical test. BMJ
315, 629-634.
Fung, K. L., Pan, J., Ohnuma, S., Lund, P. E., Pixley, J. N., Kim-
chi-Sarfaty, C.& Gottesman, M. M. (2014) MDR1 synonymous
polymorphisms alter transporter speci city and protein sta-
bility in a stable epithelial monolayer.Cancer Research74(2),
598-608.
George, J., Dharanipragada, K., Krishnamachari, S., Chan-
drasekaran, A., Sam, S. S., & Sunder, E. (2009) A single-
nucleotide polymorphism in the MDR1 gene as a predictor of
response to neoadjuvant chemotherapy in breast cancer.Clini-
cal Breast Cancer9(3), 161-165.
Ghafouri, H., Ghaderi, B., Amini, S., Nikkhoo, B., Abdi, M., &
Hoseini, A. (2016) Association of ABCB1 and ABCG2 single
Table 4: ASSP prediction results for 3435TT and 3435CC genotypes
Positio
(bp)
Putative
splice site
Sequence Score
Intron
GC
Activations
Con dence
Alt./
Cryptic
Constitutive
A) 3435TT Genotype
458 Alt. isoform/
cryptic acceptor
ttgactgcagCATTGCTGAG 6.509 0.586 0.696 0.300 0.569
507 Alt. isoform/
cryptic acceptor
ggtgtcacagGAAGAGATTG 2.614 0.543 0.652 0.334 0.487
515 Alt. isoform/
cryptic donor
GGAAGAGATTgtgagggcag 6.381 0.486 0.914 0.057 0.937
569 Constitutive
donor
ACTGCCTAATgtaagtctct 13.099 0.429 0.103 0.852 0.879
B) 3435CC Genotype
458 Alt. isoform/
cryptic acceptor
ttgactgcagCATTGCTGAG 6.509 0.586 0.696 0.300 0.569
507 Alt. isoform/
cryptic acceptor
ggtgtcacagGAAGAGATCG 2.614 0.543 0.628 0.355 0.434
515 Alt. isoform/
cryptic donor
GGAAGAGATCgtgagggcag 5.066 0.486 0.820 0.136 0.834
569 Constitutive
donor
ACTGCCTAATgtaagtctct 13.099 0.429 0.103 0.852 0.879

Davood Kheirkhah and Mohammad Karimian
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS MDR1 GENE AND BREAST CANCER 651
nucleotide polymorphisms with clinical ndings and response
to chemotherapy treatments in Kurdish patients with breast
cancer.Tumor Biology 37(6), 7901-7906.
Haimov-Kochman, R., Lavy, Y., Hochner-Celinkier, D. (2002)
Review of risk factors for breast cancer-what’s new? Harefuah
141, 702-708.
Ikeda, M., Tsuji, D., Yamamoto, K., Kim, Y. I., Daimon, T.,
Iwabe, Y. & Nakamichi, H. (2015) Relationship between ABCB1
gene polymorphisms and severe neutropenia in patients with
breast cancer treated with doxorubicin/cyclophosphamide
chemotherapy.Drug Metabolism and Pharmacokinetics,30(2),
149-153.
Isvoran, A., Louet, M., Vladoiu, D. L., Craciun, D., Loriot, M.
A., Villoutreix, B. O., & Miteva, M. A. (2016) Pharmacogenom-
ics of the cytochrome P450 2C family: impacts of amino acid
variations on drug metabolism. Drug Discovery Today. Doi:
10.1016/j.drudis.2016.09.015.
Jamali, S., Karimian, M., Nikzad, H., & Aftabi, Y. (2016) The
c.− 190 C> A transversion in promoter region of protamine1
gene as a genetic risk factor for idiopathic oligozoospermia.
Molecular Biology Reports 43, 795-802.
Jemal, A., Siegel, R., Ward, E., Murray, T., Xu, J., Smigal, C., &
Thun, M. J. (2006) Cancer statistics, 2006.CA: a Cancer Jour-
nal for Clinicians,56(2), 106-130.
Johnatty, S. E., Beesley, J., Gao, B., Chen, X., Lu, Y., Law, M.
H. & Fereday, S. (2013) ABCB1 (MDR1) polymorphisms and
ovarian cancer progression and survival: a comprehensive
analysis from the Ovarian Cancer Association Consortium
and The Cancer Genome Atlas. Gynecologic Oncology, 131(1),
8-14.
Johnstone, R. W., Rue i, A. A., & Smyth, M. J. (2000) Multiple
physiological functions for multidrug transporter P-glycopro-
tein?Trends in Biochemical Sciences,25(1), 1-6.
Karimian, M., Hosseinzadeh Colagar, A. (2016a) Methionine
synthase A2756G transition might be a risk factor for male
infertility: evidences from seven case-control studies. Molecu-
lar and Cellular Endocrinology 425, 1–10.
Karimian, M., Hosseinzadeh Colagar, A. (2016b) Association
of C677T transition of the human methylenetetrahydrofolate
reductase (MTHFR) gene with male infertility. Reproduction
Fertility and Development 28, 785-794.
Karimian, M., Nikzad, H., Azami-Tameh, A., Taherian, A.,
Darvishi, F. Z., & Haghighatnia, M. J. (2015) SPO11-C631T
gene polymorphism: association with male infertility and an
in silico-analysis. Journal of Family and Reproductive Health
9, 155-163.
Kreile, M., Rots, D., Piekuse, L., Cebura, E., Grutupa, M., Koval-
ova, Z., & Lace, B. (2013) Lack of association between poly-
morphisms in genes MTHFR and MDR1 with risk of childhood
acute lymphoblastic leukemia. Asian Paci c Journal of Cancer
Prevention: APJCP, 15(22), 9707-9711.
Lee, I. H. (2016) Molecular Prognostic and Predictive Assays
in Breast Cancer: A Practical Review. Pathology Case
Reviews,21(1), 4-10.
Lian, H., Wang, L., Zhang, J. (2012) Increased risk of breast can-
cer associated with CC genotype of Has-miR-146a Rs2910164
polymorphism in Europeans. PLoS One 7, e31615.
Matsuno, R. K., Anderson, W. F., Yamamoto, S., Tsukuma, H.,
Pfeiffer, R. M., Kobayashi, K., ... & Levine, P. H. (2007) Early-
and late-onset breast cancer types among women in the United
States and Japan.Cancer Epidemiology Biomarkers & Preven-
tion,16(7), 1437-1442.
Nickels, S., Truong, T., Hein, R., Stevens, K., Buck, K., Beh-
rens, S. & Gaudet, M. (2013) Evidence of gene-environment
interactions between common breast cancer susceptibility loci
and established environmental risk factors.PLoS Genet,9(3),
e1003284.
Nikzad, H., Karimian, M., Sareban, K., Khoshsokhan, M., &
Colagar, A. H. (2015) MTHFR-Ala222Val and male infertility:
a study in Iranian men, an updated meta-analysis and an in
silico analysis. Reproductive Biomedicine Online 31, 668-680.
Ozdemir, S., Uludag, A., Silan, F., Atik, S. Y., Turgut, B., &
Ozdemir, O. (2013) Possible roles of the xenobiotic transporter
P-glycoproteins encoded by the MDR1 3435 C> T gene poly-
morphism in differentiated thyroid cancers. Asian Paci c Jour-
nal of Cancer Prevention, 14(5), 3213-3217.
Raygan, F., Karimian, M., Rezaeian, A., Bahmani, B., & Behjati,
M. (2016) Angiotensinogen-M235T as a risk factor for myo-
cardial infarction in Asian populations: a genetic association
study and a bioinformatics approach. Croatian Medical Jour-
nal 57, 351-362.
Sabarinathan, R., Tafer, H., Seemann, S. E., Hofacker, I. L.,
Stadler, P. F., & Gorodkin, J. (2013) The RNAsnp web server:
predicting SNP effects on local RNA secondary structure.
Nucleic Acids Research 41(W1), W475-W479.
Sharif, A., Kheirkhah, D., Sharif M.R., Karimian M., Karim-
ian, Z. (2016) ABCB1-C3435T polymorphism and breast cancer
risk: a case-control study and a meta-analysis. JBUON, 21(6),
1433-1441.
Slamon, D. J., Clark, G. M., Wong, S. G., Levin, W. J., Ullrich,
A., & McGuire, W. L. (1987) Human breast cancer: correlation
of relapse and.Science,3798106(177), 235.
Soleimani, Z., Kheirkhah, D., Sharif, M. R., Sharif, A., Karimian,
M., & Aftabi, Y. (2016) Association of CCND1 Gene c. 870G> A
Polymorphism with Breast Cancer Risk: A Case-ControlStudy
and a Meta-Analysis.Pathology & Oncology Research, 1-11.
Taheri, M., Mahjoubi, F., Omranipour, R. (2010) Effect of MDR1
polymorphism on multidrug resistance expression in breast
cancer patients. Genetics and Molecular Research 9, 34-40.
Tatari, F., Salek, R., Mosaffa, F., Khedri, A., & Behravan, J.
(2009) Association of C3435T single-nucleotide polymor-
phism of MDR1 gene with breast cancer in an Iranian popula-
tion.DNA and Cell Biology,28(5), 259-263.
Tazzite, A., Kassogue, Y., Diakité, B., Jouhadi, H., Dehbi, H.,
Benider, A., & Nadi , S. (2016) Association between ABCB1
C3435T polymorphism and breast cancer risk: a Moroccan
case-control study and meta-analysis. BMC Genetics, 17(1),
126.

Davood Kheirkhah and Mohammad Karimian
652 MDR1 GENE AND BREAST CANCER BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Turgut, S., Yaren, A., Kursunluoglu, R., et al. (2007) MDR1
C3435T polymorphism in patients with breast carcinoma.
Archives of Medical Research 38, 539-544.
Wang, M., & Marín, A. (2006) Characterization and prediction
of alternative splice sites. Gene 366(2), 219-227.
Wu, H., Kang, H., Liu, Y., Tong, W., Liu, D., Yang, X. & Sha,
X. (2012) Roles of ABCB1 gene polymorphisms and haplo-
type in susceptibility to breast carcinoma risk and clinical
outcomes. Journal of Cancer Research and Clinical Oncol-
ogy,138(9), 1449-1462.