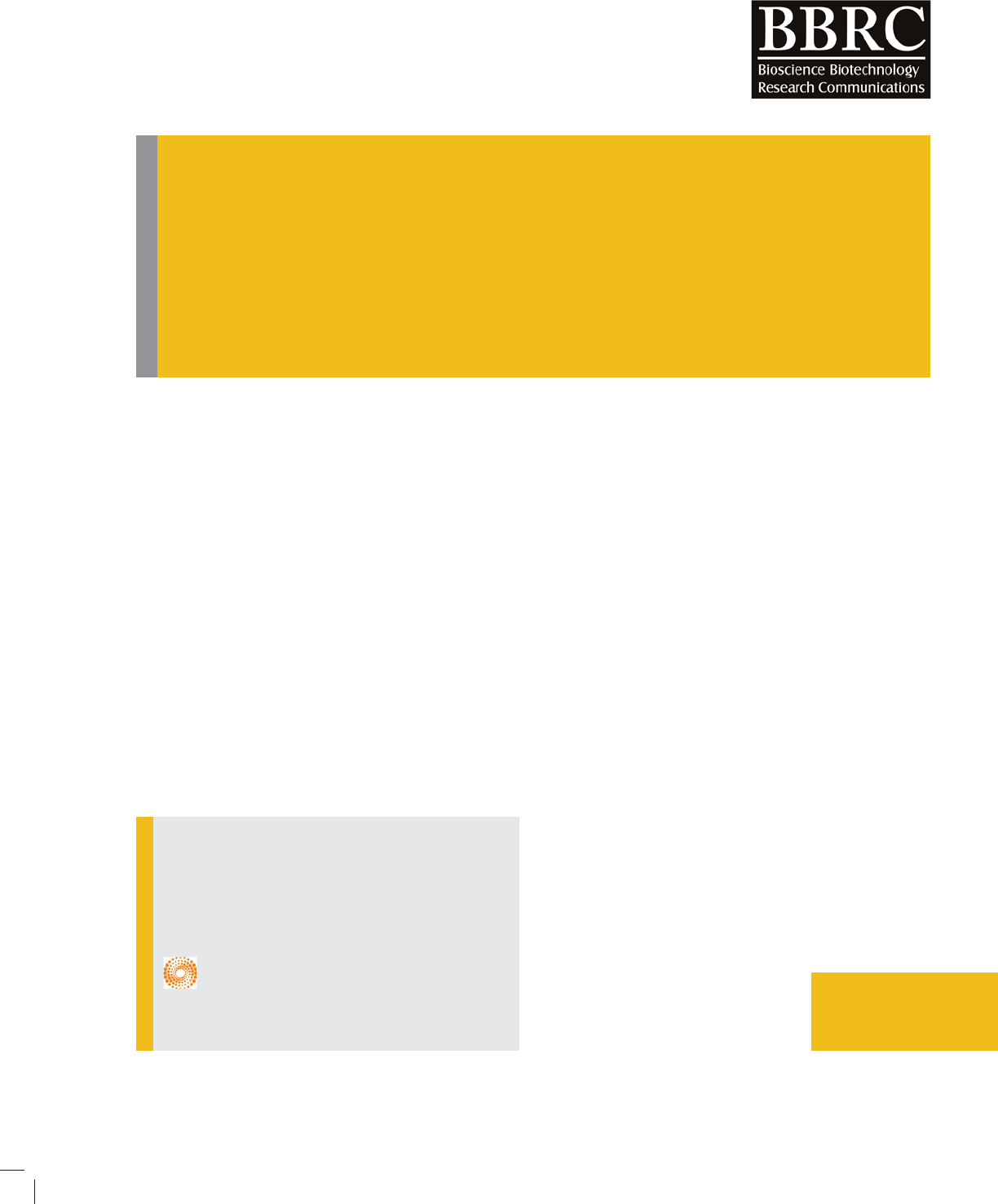
Pharmaceutical
Communication
Biosci. Biotech. Res. Comm. 9(4): 605-614 (2016)
Biological activities of methanolic leaf extract of
Ziziphus mauritiana
Emad Mohamed Abdallah,
1
*, Eman Ramadan Elsharkawy
2,3
and Abdelaziz Ed-dra
4
1
Department of Laboratory Sciences, College of Sciences and Arts, Qassim University, Al-Rass, Saudi Arabia
2
Department of Eco- physiology, Desert Research Center, 15753, Cairo, Egypt
3
Chemical Department, Faculty of Science, Northern Borders University, Saudi Arabia
4
Laboratory of Chemistry-Biology Applied to the Environment, Department of Biology, Moulay Ismail
University, Meknes, Morocco
ABSTRACT
Ziziphus mauritiana is recognized for traditional use in different areas as its fruits are consumed locally in some
parts of Saudi Arabia. However, little is known about the biological activities of the leaves. The current study has
been aimed to evaluate some bioactive properties of the methanol leaf extract of Ziziphus mauritiana. Phytochemi-
cal analysis was performed using colorimetric methods:Disc diffusion, MIC and MBC method was used to determine
the antimicrobial activity; DPPH scavenging activity and reduction capacity were determined spectrophotometrically
for the antioxidant activity, and carrageenan-induced paw edema method using rat models was employed to evalu-
ate the anti-in ammatory activity. The study revealed the presence of some bioactive phytochemical constituents
such as saponins, tannins, alkaloids, phenolic compounds, terpenoids and avonoids. The methanol leaf extract of
Ziziphus mauritiana has signi cant antibacterial activity against Bacillus cereus ATCC 10876 and Proteus vulgaris
(multi-drug resistant isolate); and varied degrees against other bacterial strains but it was not signi cant. The plant
extract also has potent antioxidant (IC50 value of 0.024 g/L competitor to the ascorbic acid and the quercetin, with
observed reducing power of Iron III to Iron II) and anti-in ammatory properties (71.83% reduction in in ammation
at a concentration of 400 mg/kg body weight of rats). The results obtained in the present study suggest that leaves of
Ziziphus mauritiana can be used as a source for functional ingredients for pharmaceutical drug industries.
KEY WORDS: ANTIBACTERIAL, ANTIOXIDANT, ANTI-INFLAMMATORY,
ZIZIPHUS MAURITIANA
605
ARTICLE INFORMATION:
*Corresponding Author: 140208@qu.edu.sa,
emad100sdl@yahoo.com
Received 27
th
Nov, 2016
Accepted after revision 26
th
Dec, 2016
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007
Thomson Reuters ISI ESC and Crossref Indexed Journal
NAAS Journal Score 2015: 3.48 Cosmos IF : 4.006
© A Society of Science and Nature Publication, 2016. All rights
reserved.
Online Contents Available at: http//www.bbrc.in/

606 BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Emad, Eman and Abdelaziz
INTRODUCTION
No doubt, plants were the main source of therapeutics
for human since ancient times and until the current era.
Although, pharmaceuticals (mostly synthetic drugs) are
the dominant drugs in modern medicine, but phyto-
medicinal drugs (mostly plant derivatives) are more
popular. The WHO reported that, up to 80% of the world
population is depending on drugs derived from plants
particularly in the developing countries (WHO, 1996).
However, the approach for drug development from plants
is very complicated and expensive, each new drug costs
about 100-360 million US dollars and at least 10 years
of intensive work in a form of multi-disciplinary and
integrated activities including many elds like botany,
chemistry, pharmacology, biomedical sciences, biotech-
nology and even anthropology (Rates, 2001).
If we put in consideration that, there are about
250,000 to 500,000 species of plants on earth, human
and animals are consuming not more than 10% of these
plant species (Abdallah, 2011), that means screening for
bioactivity of medicinal plants is important and worth-
while in order provide the basic knowledge for discovery
of new drugs. Two main constituents are produced from
plants; primary compounds such as sugars, proteins and
chlorophyll; secondary compounds (phytochemicals)
such as avonoids, terpenoids and phenolic compounds;
the bioactive properties of some plants are attributed to
the secondary or phytochemical compounds (Wadood et
al., 2013 and Akhtar et al., 2016).
These bioactive phytochemical compounds have a
great potential in the treatment of many human diseases
such as diabetes, coronary heart diseases, infectious dis-
eases and cancer (Chew et al., 2011). In ammation is a
complex series of events that occurs when any tissue
or organ is injured or damaged by chemicals, micro-
organisms, trauma, foreign bodies, surgery and ionizing
radiation, involving the releases of different bioactive
substances such as histamine, serotonin and the prosta-
glandins (Jackson-Robert and Morrow, 2011).
The classical analgesic drugs, opiates and non-steroi-
dal anti-in ammatory drugs (NSAIDs) all have their ori-
gin in natural products that are used for centuries. Sali-
cin, a bitter glycoside from the willow bark extract are
known since the 18
th
century for its bene cial effect in
fever and pain. The synthetic form, acetylsalicylic acid
(ASA) was introduced into medicine as far back as the
19
th
century. Quite a number of derivatives of ASA and
other newer drugs were discovered but these are associ-
ated with side effects limiting their use. These factors
include allergy, gastric mucosal irritation and/or gas-
tric ulceration due to the acidic nature of most NSAIDs
and inhibition of a mucosal protective prostaglandin
E (PGE). Others include prolonged vascular bleeding,
NSAID-induced nephropathy, salt and water retention
by the kidney as well as the displacement of other drugs
from their protein binding sites due to the greater af n-
ity of NSAIDs for plasma albumin are some of the other
challenges facing this group of drugs (Foster, 1999).
The roots and bark of Ziziphus Leaves and root bark of
the plant are used as the remedy of in ammation by the
local communities of eastern India. While, much work
exists on extracts of leaves in animal model proving the
anti-in ammatory action (Kumar and Sharma, 2010;
Soliman 2011; Goyal et al., 2012 and 2013 Ashraf et al.
(2015).
Ziziphus mauritiana from family Rhamnaceae
commonly known as Ber or Indian jujube. The genus
Ziziphus is very common plant found in many places in
the world. Ziziphus mauritiana is a tropical shrub native
to Indian Subcontinent, The Southeast Asia, Iran and
some regions of Africa. Various parts of Ziziphus mau-
ritiana are used for nutritional and medical purposes.
However, leaves are employed traditionally as astringent
and anti-typhoid (Akhtar et al., 2016, Naja , 2013). In
literature, many studies reported that Ziziphus mauri-
tiana have some medical bene ts such as antioxidant,
anti-microbial, anti-diarrheal, anti-diabetic, hepato-
protective and anti-cancer (Lim, 2013). The current study
aimed to evaluate in vitro antibacterial, antioxidant and
in vivo anti-in ammatory activity of the methanol leaf
extract of Ziziphus mauritiana growing in Saudi Arabia.
MATERIAL AND METHODS
PLANT MATERIAL
Leaves of Ziziphus mauritiana (Fig 1.) was collected in
March 2016 from the garden of College of Sciences and
Arts at Al-Rass, after veri cation and authentication by
Dr. Wail Elsadig Abdalla (Plant taxonomist), voucher
specimen was deposited in the herbarium of the depart-
ment of laboratory sciences, College of Sciences and
Arts at Al-Rass. Qassim University. Collected leaves were
washed thoroughly with tap water, rinsed again in dis-
tilled water, and then dried in shade for up to one week.
The dried leaves were crushed to ne powder using a
blender (GEEPAS®, GCG 292) and kept until used.
EXTRACTION
About 300 g of the dried powder of the leaves of Ziziphus
mauritiana L. was macerated in 1.5 liter of 80% metha-
nol (v/v) (HPLC grade, Fisher Scienti c,UK ) to serve as
a hydroalcoholic solvent and left for up to 3 days in a
dark tighten container at room temperature (32-37
o
C)
with frequent soaking. In the 4
th
day, the macerate was

BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
607
Emad, Eman and Abdelaziz
ltered using Whatman lter papers No.1 (Whatman
International Ltd, UK), the ltrate was put in the incu-
bator (BINDER GmbH, Germany) at 45
o
C and allowed
to evaporate for up to 10 days, till getting a semi-solid
extract, which was used for the experiments.
PRELIMINARY PHYTOCHEMICAL ANALYSIS
Leaves of Ziziphus mauritiana were qualitatively
screened for some bioactive phytochemical constituents
by means of Colorimetric qualitative tests. The crude of
methanolic extract was used to detect the presence of
saponins and anthraquinones (Stahl, 1973), alkaloids
(Yusuf et al., 2014), tannins (Ashfaq et al., 2012), Terpe-
noids and avonoids (Mujeeb et al., 2014) and Phenolic
compounds (Clarke, 1975).
MICROORGANISMS
Four pathogenic bacterial strains (Staphylococcus
aureus, Proteus vulgaris, Pseudomonas aeruginosa, and
Klebsiella pneumoniae) and one referenced bacterial
strain (Bacillus cereus ATCC 10876) were used in the
antibacterial activity tests. The pathogenic strains were
obtained from the Department of Pathology and Labo-
ratory medicine, Al-Rass General Hospital, Saudi Ara-
bia, they were identi ed by Dr. Mohamed Algadi. The
sources of these pathogens and the antibiotics sensitiv-
ity pro le are shown in (Table 2). While, the referenced
type culture strain was brought from the department of
laboratory sciences, College of Sciences and Arts at Al-
Rass, Qassim University, Saudi Arabia.
ANTIBACTERIAL ASSAY
The antibacterial activity of the leaves of Ziziphus mau-
ritiana (Methanol extract) was evaluated by disc dif-
fusion method as cited in (Abdallah, 2016) with minor
modi cation. The tested bacterial strains were sub-cul-
tured for 18 h, and then working bacterial samples were
prepared and adjusted to be equivalent 0.5 McFarland
(Approximately 1-2 × 108 CFU/ml). 20 ml of hot auto-
claved Mueller-Hinton agar (Watin-Biolife, KSA), was
poured to a sterile disposable Petri-dish (size 100x15mm)
and left until solidi ed. 100 µl from the bacterial culture
was loaded on the agar and swapped with a sterile cot-
ton swap. Filter paper discs (cut off from Whatman No.1
lter paper) were immersed in the reconstituted extracts
at 400 and 200 mg/ml. these discs were put on the agar
plate, another antibiotic disc was loaded to the plate
(gentamincin 10 µg/disc) to serve as a positive control, a
sterile disc saturated with 80% methanol was also put on
the plate to serve as negative control. The seeded plate
was incubated at 37
o
C for 24 hours, the test was repeated
twice, the mean inhibition zone and standard error of
means were calculated.
MINIMUM INHIBITORY CONCENTRATION
TEST (MIC)
Only the bacterial strains which showed mean inhibition
zone above 10 mm were subjected to MIC test, using
the broth dilution method as reported in (El-Mahmood
and Ameh, 2007) with slight modi cation. A suf cient
quantity of test tubes containing 1 ml of Nutrient broth
(Watin Biolife, KSA) was prepared. 1 ml of the plant
extract (200 mg/ml) was dropped in the rst tube and
mixed well. Serial two fold dilutions were made subse-
quently to get 100, 50, 25, 12.5, 6.25, 3.123 mg/ml. 100
µl of the adjusted bacterial solution (1-2 × 108 CFU/ml)
was put in each of the seven tubes. Positive control tube
(all contents without the extract and with 1 ml of 5mg/
ml Chloramphenicol), and a negative control tube (nutri-
FIGURE 1. The leaves of Ziziphus mauritiana.

608 BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Emad, Eman and Abdelaziz
ent broth and 1 ml methanol) were prepared. All tubes
were incubated for 18 hours at 37
o
C. The lowest dilution
with no obvious growth was considered as MIC.
DETERMINATION OF MINIMUM BACTERICIDAL
CONCENTRATION (MBC)
The MBC test was performed as cited in Doughari (2006)
with minor modi cation. 100 l from the MIC test tubes
that showed no visible growth was loaded in new pre-
viously prepared sterile plates containing nutrient agar
(Watin-Biolife, KSA) and spread over the agar with a
sterile swap. To another 2 agar plates, one was inocu-
lated with Chloramphenicol as a positive control and the
other one was inoculated with 10%DMSO as a negative
control. All cultured plates were incubated overnight at
37
o
C and investigated for bacterial growth. Plates with
no visible growth were considered as the MBC.
DPPH SCAVENGING ACTIVITY
The hydrogen atoms or electrons donation ability of the
plant extract and some pure compounds were measured
from the bleaching of a purple-colored methanol solu-
tion of DPPH (Kubola et al., 2008). Brie y, 1 mL of a
0.08 g/L solution of DPPH radical in methanol was added
to 2 mL of the extract at different concentrations. The
absorbance of the resulting solution was measured after
30 min in dark at 517 nm with a spectrophotometer. The
percentage inhibition of activity was calculated as:
(A blank-A sample)
% Inhibition = × 100
(A blank)
Quercetin and ascorbic acid was used as positive control
and the concentration providing 50% inhibition (IC
50
)
were calculated from the graph of inhibition percentage
plotted against the extract concentration.
REDUCTIVE CAPACITY: IRON (III) TO IRON (II)
The reductive capacity of the extract was determined
using ferric to ferrous iron reduction assay as determined
spectrophotometrically from the formation of Perl’s
Prussian blue colored complex (Dorman et al., 2003).
Brie y, 1 mL of each sample, in methanol, was mixed
with 2.5 mL of phosphate buffer (0.2 mol/L, pH 7.0) and
2.5 mL of potassium hexacyanoferrate K
3
Fe(CN)
6
solu-
tion. After 30 min incubation at 50°C, aliquots (2.5 mL)
of trichloroacetic acid (10%) were added to the mixture.
Then, 2.5 mL of this solution was mixed with distilled
water (2.5 mL) and FeCl
3
(0.5 mL, 0.1%), and the absorb-
ance was measured at 700 nm. The ascorbic acid stand-
ard was used for comparison.
ANTI-INFLAMMATORY STUDY
Anti-in ammatory effect was evaluated in rats using a
carrageenan-induced paw edema (Winter et al., 1962).
Experiments on animals were performed in accord-
ance with the ethical guidelines and regulations set
forth by Faculty of Science, Northern Borders Univer-
sity, Saudi Arabia. Four groups of animals (n=6) were
fasted overnight prior to the study with free access to
water. The 1
st
and 2
nd
groups (normal control and refer-
ence, respectively) were treated orally with the vehicle (5
mL/kg) and Indomethacin (10 mg/kg), respectively. The
extract of Ziziphus mauritiana at 200 and 400 mg/kg
was administered orally to rats of the 3
rd
and 4
th
groups,
respectively. After 30 min, in ammation was induced
by subplantar injection of 1% carrageenan (0.1 mL) in
the left hind paw of all animals. The paw volumes up
to the tibio tarsal joint were measured in mL using a
plethysmometer (Ugo basile, Italy), immediately before
and at 1, 2 and 3 h after carrageenan administration.
Their mean increases in the injected paw volume were
calculated and the percentage inhibition of paw edema
was calculated using the following equation.
Inhibition (%) = {1- (Vt/Vc)}100
Where, Vt and Vc are the mean change in paw volume
of treated and control rats, respectively.
DRUG PREPARATION
Carrageenan (1% w/v), Indomethacin and the test
extracts were prepared by suspending in 1% Carboxy
methyl cellulose solution.
STATISTICAL ANALYSIS
Values were expressed as means ± S.E. Compari-
sons between means were carried out using one-way
ANOVA followed by LSD (least signi cant difference)
and Tukey multiple comparison tests. The P < 0.05 was
accepted as being signi cant in all types of statisti-
cal tests. SPSS software (version 17) was used to carry
out all.
RESULTS
PHYTOCHEMICAL CONSTITUENTS
The investigation has revealed that methanol leaf
extract of Ziziphus mauritiana contains some bioac-
tive compounds such as saponins, tannins, alkaloids,
phenolic compounds, and terpenoids. Flavonoids and
anthraquinones did not detect in the methanol extract
(Table 1).

BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
609
Emad, Eman and Abdelaziz
Table 1: The phytochemical analysis of 80%
methanol extract (v/v) of Ziziphus mauritiana
leaves.
Phytochemicals Methanol extract
Saponins +++
Tannins ++
Alkaloids ++
Flavonoids +
Terpenoids +
Phenolic compounds ++
Anthraquinones -
+++ = Present in high amount, ++ = moderately present, + =
Trace amounts,
- = Absent.
Table 2: Antibiotics sensitivity pro le of the clinical pathogenic strains and the
referenced bacterial strain.
Bacterial strain Origin Source AK GM CPM TC PRL IMI
Proteus vulgaris Clinical Pus R R R R R S
Pseudomonas aeruginosa Clinical Sputum S S R R R W
Klebsiella pneumonia Clinical Sputum S R R W R S
Escherichia coli Clinical Urine S S S S S S
Staphylococcus aureus Clinical Pus R R R S S S
Bacillus cereus ATCC 10876 - S S R R S S
AK: Amikacin 30 µg, GM: Gentamicin 10 µg, CPM: Cefepime 30 µg, TC: Ticarcillin 75 µg, PRL: Piperacillin
100 µg, IMI: Imipenem 10 µg, R: Resistant, S: Sensitive, (Bacterial inhibition zone ≤ 10 mm considered
sensitive).
Table 3: The antibacterial activity of the methanol leaf extract of Ziziphus mauritiana.
Test Mean zone of growth inhibition (mm)*
Gram-positive Gram-negative
Sa Bc Pv Pa Kp
Methanol extract (400 mg/ml) 7.25 ± 0.75 13.00 ± 0.00** 8.75 ± 0.75** 6.25 ± 0.25 6.50 ± 0.50
Methanol extract (200 mg/ml) 6.50 ± 0.50 12.00 ± 0.00** 7.50 ± 0.50 6.00 ± 0.00 6.50 ± 0.50
Gentamicin (10 g/disc) 7.25 ± 0.75 20.00 ± 0.00** 9.50 ± 0.50** 10.00 ± 0.00** 6.75 ± 0.25
80% Methanol 6.00 6.00 6.00 6.00 6.00
*Disc diameter=6 mm, zone off inhibition is the mean of two replicates ±standard error of means; 6 mm =no inhibition; Sa=
Staphylococcus aureus, Bc= Bacillus cereus ATCC 10876 , Pv= Proteus vulgaris, Pa= Pseudomonas aeruginosa, Kp= Klebsiella
pneumoniae. **Signi cant (p<0.05)
ANTIBACTERIAL PROPERTIES
For the antibacterial testing, both of the multi-drug
resistant (MDR) and non-MDR gram- positive and gram-
negative bacteria were chosen. As shown in (Table 2),
clinical pathogenic strains resistant to three or more
of the tested antibiotics were considered as multi-drug
resistant, which was Proteus vulgaris, Pseudomonas
aeruginosa, Klebsiella pneumoniae and Staphylococcus
aureus. The other bacterial strains considered as non-
MDR, which were Escherichia coli and Bacillus cereus
ATCC 10876.
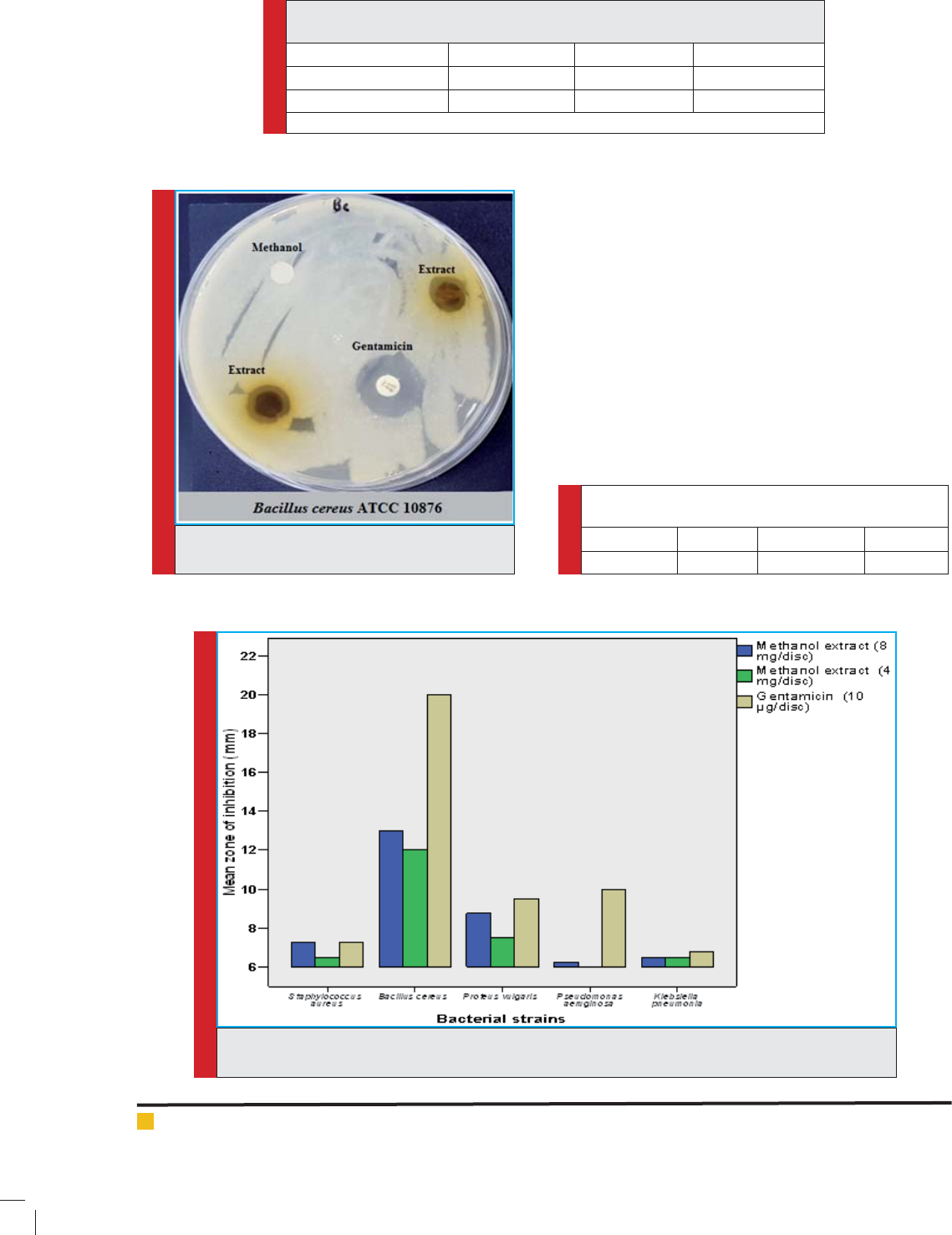
As shown in (Table 3), the disc diffusion test revealed
various antibacterial ef cacies of the plant extract. At
concentration 400mg/ml, Bacillus cereus ATCC 10876,
which is a referenced non-MDR bacterial strain showed
the highest inhibition zone (13.00±0.00 mm) and Pro-
teus vulgaris (8.75± 0.75 mm) which were statistically
signi cant (p<0.05), followed by Staphylococcus aureus
(7.25± 0.75 mm), Klebsiella pneumonia (6.50± 0.50 mm)
and Pseudomonas aeruginosa (6.25± 0.25mm), respec-
tively. There were also various degrees of antibacte-
rial activity at the concentration 200 mg/ml of lesser
strength. Figure (3) illustrates the antibacterial activity
of the methanol leaf extract in comparison with the gen-
tamicin (10µg/ml), after excluding the diameter of the
paper disc (6 mm).
Bacteria with the highest sensitivity to the metha-
nol leaf extract of Ziziphus mauritiana (Figure 2) were
examined for MIC and MBC. The MIC and MBC values of
Bacillus cereus ATCC 10876 and Proteus vulgaris were
25, 100 and 50, 100 mg/ml, respectively (Table 4).
ANTIOXIDANT ACTIVITY
In DPPH assay, the hydrogen donating ability of the
extract was determined by converting the DPPH radical
to nonradical by the reduction process. In this study,
the methanol extract of Ziziphus mauritiana from Saudi
Emad, Eman and Abdelaziz
610 BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Arabia was investigated for the antioxidant activity
with DPPH scavenging assay. The results are shown in
(Table 5).
The IC
50
value was de ned as the concentration of
sample that scavenged 50% of the DPPH. In this study,
the results showed an important antioxidant power of
Ziziphus mauritiana extract compared to the standard
product such as quercetin and ascorbic acid. The result
(table 1) showed that the antioxidant activity of metha-
nol extract of Ziziphus mauritiana has an IC
50
value of
0.024 g/L, which was near to the inhibition capacity of
the ascorbic acid. Then the quercetin has an IC
50
value of
0.008 g/L. For the evaluation of the Reductive capacity
of the methanol leaves extract of Ziziphus mauritiana
Table 4: The minimum inhibitory concentration (MIC) and minimum bactericidal
concentration (MBC) of bacteria showed signi cant sensitivity to the extract.
Microorganism* MIC (mg/ml) MBC (mg/ml) MBC/MIC
Bc 25 100 4
Pv 50 100 2
*Bc= Bacillus cereus ATCC 10876, Pv= Proteus vulgaris
FIGURE 2. Representative photo showing the zone
of inhibition of the extract.
FIGURE 3. The antibacterial activity of the methanol leaf extract of Ziziphus mauritiana compared to the
antibiotic gentamicin.
Table 5: IC
50
(g/L) values of methanol extract of
Ziziphus mauritiana to DPPH assay.
Antioxidant Extract Ascorbic acid Quercetin
IC
50
(g/L) 0.024 g/L 0.017 g/L L

Emad, Eman and Abdelaziz
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
611
(Iron III to Iron II. In the ferric to ferrous iron reduction
assay, the electron donation capacity of the extract was
assessed and compared to that of ascorbic acid, which
known as strong reducing agent. The reducing power of
Ziziphus mauritiana extract increased with the increas-
ing of their concentrations (Figure 4).
ANTI-INFLAMMATORY ACTIVITY
The anti-in ammatory activities were examined in vivo
via inhibition of carragenan induced rat paw edema
method at 200 and 400 mg/kg dose orally. The results of
the anti-in ammatory activity obtained were compared
to those of Indomethacin and those of the control, which
received saline. The evolutions of in ammation for dif-
ferent groups are shown in Table 6.
According to the results (Table 6), the in ammation
caused by carrageenan increases with time and reaches
a maximum of 0.401 ±0.05mL for three hours. From
these results, it appears that the plant extract inhibits
signi cantly the in ammatory response. This inhibition
is gathering of time, or at 1h the difference in paw vol-
ume is measured 0.167 ±0.06 ml, and the time equal to
2 hours the inhibition was 0.197 ± 0.06 mL and showed
no signi cant difference compared to control. At time
equal to 1 hour, the extract represents a highly signi -
cant inhibition of 0. 11 ±0.04 mL and which is very close
to the effect of Indomethacin 0.10 ± 0.05 mL.
Then, this inhibition is continuous over time; one can
deduce that the extract has an anti-in ammatory effect
similar to the effect of Indomethacin. The anti-in am-
matory activity (71.78% reduction in in ammation) was
observed with extract at the concentration of 400mg/kg
body weight when administered orally to rats.
In the 1st hour, the MeOH extract at a dose of 200
mg/kg shows a percent inhibition of 47.62% lower than
that obtained with Indomethacin, at a dose of 400 mg/
kg at the third time the effect of the extract (71.78%) is
almost similar to that of Indomethacin. (75.10%).) inhi-
bition values.
The administration of Ziziphus mauritiana extract at
a dose of 400 mg/kg prevents signi cantly (P <0.05) the
plantar edema in rats from the second hour of treatment.
This suggests the signi cant anti-in ammatory effect of
the extract of the plant, it could be due to the richness
of the methanol extract in bioactive compounds which
are rich in Ziziphus, such as saponins, triterpenoic acids,
fatty acids avonoids and alkaloids.
FIGURE 4. Reducing power of ascorbic acid and methanol extract of Ziziphus
mauritiana.
Table 6: Anti-in ammatory effect of Indomethacin and Extract using carrageenan-induced paw
edema.
Groups Dose (mg/kg) Paw volume (mL) Inhibition %
0 h 1 h 2 h 3 h 0 h 1 h 2 h 3 h
Control 0.0 0.350 0.402 0.420 0.342 - - - -
Indomethacin 10 0.132 0.100 0.068 0.100 62.38 75.10 83.73 70.73
Extrant 200 0.183 0.167 0.197 0.258 47.62 58.51 53.17 24.39
400 0.142 0.113 0.152 0.225 59.52 71.78 63.89 34.15

612 BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Emad, Eman and Abdelaziz
DISCUSSION
The preliminary phytochemical screening showed vari-
ous bioactive ingredients such as saponins, tannins, alka-
loids, phenolic compounds, and terpenoids. The presence
of these components is an indication that this plant has
some medical properties. The ndings of current study
agreed with previous studies, Naja (2013) reported that
leaves of Ziziphus mauritiana revealed the presence of
saponins, phenolic compounds, tannins and glycosides.
Parmar et al. (2012) stated that leaves of Ziziphus mauri-
tiana contain glycosides, saponins, phenols, lignins and
tannins. Plants rich in saponins have anti-in ammatory
activity and strengthen the immune system, Tannins are
antibacterial compounds which damages the bacterial
cell wall (Mainasara et al., 2012) Phenolic compounds,
alkaloids, avonoids, tannins, saponins and glycosides
are good antioxidant compounds and controls the oxi-
dative stress related disorders (Biapa et al., 2007).
The methanol leaf extract of Ziziphus mauritiana
resulted in variable zone of inhibitions. However, Bacil-
lus cereus ATCC 10876 (Gram-positive) and Proteus vul-
garis (Gram-negative) exhibited the highest sensitivity.
Similar studies published that leaves of Ziziphus mauri-
tiana have antibacterial activity against different bacte-
rial strains; Naja (2013) cited that the methanol leaf
extracts of Ziziphus mauritiana exhibited signi cant
activity against Staphylococcus aureus and Escherichia
coli. Ashraf et al. (2015) claimed that the methanol leaf
extract of Ziziphus mauritiana has potent antibacterial
effects against Escherichia coli, Bacillus subtilis and
Staphylococcus aureus. However, in the current study the
other clinical pathogens including MDR and non-MDR
isolates did not show signi cant antibacterial activities.
In addition, it was published that, leaves of Ziziphus
mauritiana have no antibacterial effects against some
bacterial strains.
Mainasara et al. (2012) mentioned that methanol and
ethanol extracts did not show a signi cant effect against
Pseudomonas aeruginosa, Salmonella typhi, Escherichia
coli, Staphylococcus aureus and Streptococcus pyrogenes
at 120 mg/ml. Accordingly, more studies regarding the
mechanism and mode of action of this plant extract
are required. The results also revealed that the ratios of
MBC/MIC values are 4 and 2 for Bacillus cereus ATCC
10876 and Proteus vulgaris. It is considered that, to clas-
sify any plant extract as bactericidal the ratio of MBC/
MIC should be ≤ 4, but if this ratio is > 4, so the plant
extract classi ed as bacteriostatic (Djeussi et al., 2013).
Accordingly, the methanol leaf extract of Ziziphus mau-
ritiana have bactericidal effect on Bacillus cereus ATCC
10876 (Non-MDR referenced bacteria) and bacteriostatic
effect on Proteus vulgaris
(MDR-pathogen). Based on
these interesting ndings, more studies regarding the
mechanism and mode of action of this plant extract are
required in order to understand the nature of these anti-
bacterial constituents. Free radicals, formed as result of
oxidation, are one of the major causes of degenerative
diseases (Phamhuy et al., 2008). Pharmacological evalu-
ation of plant extracts is incomplete without assessment
of their free radical scavenging activity. Therefore, in
the present study, DPPH free radicals scavenging poten-
tial and the ferric reducing antioxidant power (FRAP)
assay of methanol extract of Ziziphus mauritiana were
appraised.
In this study, methanol extract of Ziziphus mauriti-
ana has an IC
50
of 0.024g/l, compared to ascorbic acid
(IC
50
= 0.017 g/L) and Quercetin (IC
50
= 0.008 g/L). This
result is in agreement with a previous study in Malaysia
(Perumal et al., 2011), which reported an IC
50
of Ziziphus
jujuba leaf extract of 20.62 g/mL. Another study in
Pakistan shows an IC
50
of 0.11 mg/mL (Ashraf et al.,
2015).In Nepal, methanol extract of Ziziphus mauriti-
ana has an IC
50
of 47.50 g/mL (Sharma et al., 2015).
Methanol extract of Ziziphus mauritiana has an impor-
tant antioxidant activity because it contains the sub-
stances having an antioxidant action such as ascorbic
acids and avonoids (Cheng et al., 2000; Pawlowska et
al., 2000; Preeti and Tripathi, 2014). The avonoids are
considered as ef cient radical scavengers and found in
almost every plant. In addition, quercetin belongs to the
avonoids family, has a higher antioxidant activity than
ascorbic acid, this results is reinforced by the study of
Wybranowski et al. (2013).
In this study, methanol extract of Ziziphus mauri-
tiana has good signi cant anti-in ammatory, the ratio
of inhibition reached to 71.1% that close to positive
control,The antioxidant and anti-in ammatory activ-
ity could be attributed to the presence of many bioac-
tive compounds in Ziziphus mauritiana. Taking in to
account that, it is plausible to suggest that the anti-
in ammatory activity of extract of Ziziphus mauritiana
involved, partly, synergistic action of alkaloids, avo-
noids, condensed tannins, and saponins.
The cellular and molecular mechanism by which
the -carrageenan induced in ammatory process is
well known. It stimulates the release of histamine and
serotonin from mast cells, starting it with a cascade of
events that produce other mediators that contribute to
the establishment of the acute in ammatory response
(Cuzzocrea et al., 1998). Indeed, the carrageenan-
induced during the early phase (1-2 h) of the in am-
matory response, the production of pro-in ammatory
factors such as histamine, serotonin, leukotrienes, PAF
and prostanoids. These factors cause vascular changes
leading to plasma exudation. In addition to their inhibi-
tion of the production of pro-in ammatory mediators,
secondary metabolites ( avonoid, alkaloid and sapo-

Emad, Eman and Abdelaziz
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
613
nin) inhibit neutrophil recruitment to the pleural cav-
ity through the inhibition of the expression of adhesion
molecules on the endothelial cell wall veins (Middleton
et al., 2000). Flavonoids block the migration of leuko-
cytes to the in ammatory site by inhibiting adhesion
molecules ICAM-1 and VCAM-1, and this regulation by
TNF-. (Tsuda et al., 2002) report that the administra-
tion of cyanidin 3-O--glucoside inhibits in ammation
induced by zymosan. It has been reported indeed that
quercetin blocks adhesion of leukocytes to the endothe-
lial wall of the umbilical veins by inhibiting the expres-
sion of ICAM-1 (Anné et al., 1994). Gallic acid in turn
inhibits leukocyte migration by inhibiting the molecules
VCAM-1 adhesion, ICAM-1 and E-selectin in vascular
endothelial cells, this inhibition is due to inhibition of
IL-1, TNF- and NF-kB (Murase et al., 1999). This agrees
with our results at the high inhibition in the second hour
of the experiment, which is related to the production
of pro-anti-in ammatory compounds of the leaves of
Ziziphus mauritiana.
CONCLUSION
Many pharmaceutical innovations are invented from
natural products. Leaves of Ziziphus mauritiana are rich
in phytochemical ingredients. These ingredients have
antibacterial, antioxidant and anti-in ammatory prop-
erties. Isolation and puri cation of different bioactive
phytochemicals may further yield signi cant antibac-
terial, antioxidant, anti-in ammatory or other curative
properties against different ailments.
FUNDING
None
CONFLICT OF INTEREST
None-declared
REFERENCES
Abdallah, E. M. (2011): Plants: An alternative source for anti-
microbials. J. Appl. Pharm. Sci., 1(6): 16-20.
Abdallah, E. M. (2016): Antibacterial ef ciency of the Suda-
nese roselle (Hibiscus sabdariffa L.), a famous beverage from
Sudanese folk medicine. J. Intercult. Ethnopharmacol., 5(2):
186-190.
Akhtar, N., Ijaz, S., Khan, H. M. S., Uzair, B., Reich, A. and
Khan, B.A. (2016): Ziziphus mauritiana leaf extract emulsion
for skin rejuvenation. Trop. J. Pharm. Res. 15(5): 929-936.
Anné, S., Agarwal, R., Nair, M. P., Schwartz, S. A., Ballow, M.,
Kandaswami, C. and Middleton, E. (1994): Inhibition of endo-
toxininduced expression of intercellular adhesion molecule-1
and of leukocyte adhesion to endothelial cells by the plant
avonoid quercetin. J. Allergy Clin. Immunol., 93: 276.
Ashfaq, M., Shah, K. W., Ahmad, S. and Singh, D. (2012): Pre-
liminary phytochemical screening of alcoholic and aqueous
extracts of Mentha longifolia Linn. leaves. Int. J. Biol. Pharm.
Res., 3(3): 384-386.
Ashraf, A., Sarfrz, R. A., Anwar, F., Shahid, S.A. and Alkharfy,
K. M. (2015): Chemical composition and biological activities
of leaves of Ziziphus mauritiana L. native to Pakistan. Pak. J.
Bot., 47(1): 367-376.
Biapa, P. C.,Agbor, G. A.,Oben, J. E. and Ngogang, J. Y. (2007):
Phytochemical studies and antioxidant properties of four
medicinal plants used in Cameroon. Afr. J. Trad. Complement.
Altern. Med.,4(4):495-500.
Cheng, G., Bai, Y., Zhao, Y., Tao, J., Liu, Y., Tu, G., Ma, L., Liao,
N..and Xu, X. (2000): Flavonoids from Ziziphus jujuba Mill
var. spinasa. Tetrahedron., 56: 8915-8920.
Chew, Y. L., Chan, E. W. L., Tan, P. L., Lim, Y. Y., Stanslas, J. and
Goh J. K. (2011): Assessment of phytochemical content, poly-
phenolic composition, antioxidant and antibacterial activities
of Leguminosae medicinal plants in Peninsular Malaysia. BMC
Compl. Altern. Med., 11:12. DOI:10.1186/1472-6882-11-12
Clarke, E. G. C. (1975): Isolation and identi cation of Drugs.
The pharmaceutical Press, London, UK.
Cuzzocrea, S., Zingarelli, B., Hake, P., Salzman, A. L. and
Szabó, C. (1998): Antiin ammatory effects of mercaptoethyl-
guanidine, a combined inhibitor of nitric oxide synthase and
peroxynitrite scavenger, in carrageenan-induced models of
in ammation.Free Radic. Biol. Med.,24(3):450–459.
Djeussi, D. E., Noumedem, J. A., Seukep, J. A., Fankam, A.
G., Voukeng, I. K., Tankeo, S. B., Nkuete, A. H. and Kuete.
V. (2013): Antibacterial activities of selected edible plants
extracts against multidrug-resistant Gram-negative bacteria.
BMC Complement. Altern. Med., 13: 164.
Dorman, H. J. D., Peltoketo, A., Hiltunen, R. and Tikkanen,
M. J. (2003): Characterisation of the antioxidant properties of
de-odourised aqueous extracts from selected Lamiaceae herbs.
Food Chem., 83: 255-262.
Doughari, J. H. (2006): Antimicrobial Activity of Tamarindus
indica Linn. Trop. J. Pharm. Res., 5(2): 597-603.
Duh, P. D., Du, P. C. and Yen, G. C. (1999): Action of metha-
nolic extract of mung hulls as inhibitors of lipid peroxida-
tion and non-lipid oxidative damage. Food Chem. Toxicol., 37:
1055-1061.
El-Mahmood, A. M. and Ameh, J. M. (2007): In-vitro anti-
bacterial activity of Parkia biglobosa (Jacq) root, bark extract
against some microorganisms associated with Urinary tract
infections. Afri. J. Biotech., 6(11): 195-200.
Gordon, M. H. (1990): The mechanism of antioxidant action
in vitro. In: Foods antioxidants. Pp 1-18 (Ed) B. J. F. Hudson,
London: Elsevier Applied Science, London UK.
Goyal, M., Sasmal, D. and Nagori, B.P. (2012): Analgesic and
anti-in ammatory activity of ethanolic extract of Zizyphus
nummularia. Res. J. Med. Plant, 6:521–528.

Emad, Eman and Abdelaziz
614 BIOLOGICAL ACTIVITIES OF THE METHANOL LEAF EXTRACT OF
ZIZIPHUS MAURITIANA
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Goyal, M., Ghosh, M., Nagori, B. P. and Sasmal, D. (2013):
Analgesic and anti-in ammatory studies of cyclopeptide alka-
loid fraction of leaves of Ziziyphus nummularia. Saudi J. Biol.
Sci., 20:365–371.
Jackson-Robert, L. and Morrow, J. D. (2011): Analgesic-Anti-
pyretic and Anti-in ammatory agents and drugs employed in
the treatment of gout. In: Goodman and Gilman’s the Phar-
macological Basis of Therapeutics. Pp. 687-732 (Ed) Hardman,
J.G., L.E. Limbird and A.G. Gilman, McGraw-Hill Medical Pub-
lishing Division 2011 New York USA.
Kubola, J. and Siriamornpun, S. (2008): Phenolic content and
antioxidant activities of bitter gourd (Momordica charantia L.)
leaf stem and fruit fraction extracts in vitro. Food Chem., 110:
881-890.
Kumar, S., Garg, V. K. and Sharma, P. K.. (2010): A review of
Ziziphus nummularia. Pharmacologyonline, 2:565–574.
Lim, T. K. (2013): Edible Medicinal and Non-Medicinal Plants:
Fruits, Springer Science+Business Media Dordrecht. 605-613.
DOI 10.1007/978-94-007-5653-3_31
Mainasara, M. M., Aliero, B. L., Aliero, A. A. and Yakubu, M.
(2012): Phytochemical and Antibacterial Properties of Root
and Leaf Extracts of Calotropis procera. Nigerian J. Basic Appl.
Sci., 20(1): 1-6.
Middleton, E. J. R., Kandaswami, C. and Heoradies T. C. (2000):
The effects of plant avonoids on mammalian cells: implica-
tions for in ammation, heart disease, and cancer. Pharmacol.
Rev., 52: 673-751.
Mujeeb, F., Bajpai, P. and Pathak, N. (2014): Phytochemical
evaluation, antimicrobial activity, and determination of bio-
active components from leaves of Aegle marmelos. BioMed
Res. Inter. Hindawi Publishing Corporation. Article ID 497606.
http://dx.doi.org/10.1155/2014/497606
Murase, T., Kume, N., Hase, T., Shibuya, Y., Nishizawa, Y.,
Tokimitsu, I. and Kita, T. (1999): Gallates Inhibit Cytokine-
Induced Nuclear Translocation of NF-kB and Expression of
Leukocyte Adhesion Molecules in Vascular Endothelial Cells.
Arterioscler Thromb. Vasc. Biol., 19:1412-1420.
Naja , S. (2013): Phytochemical screening and antibacterial
activity of leaf extract of Ziziphus mauritiana Lam. Int. Res. J.
Appl. Basic Sci., 4 (11): 3274-3276.
Parmar, P., Bhatt, S., Dhyani, S. and Jain, A. (2012): Phytochem-
ical studies of the secondary metabolites of Ziziphus mouritiana
Lam. Leaves. Int. J. Curr. Pharm. Res., 4(3): 153-155.
Pawlowska, A. M., Camangi, F., Bader, A. and Braca, A. (2000):
Flavonoids of Zizyphus jujuba and Zizyphus spina-christi (L)
Wild (Rhamnaceae) fruits. Food Chem., 112: 858- 862.
Perumal, S., Mahmud, R., Piaru, S. P., Cai, L. W. and Ramana-
than, S. (2012): Potential Antiradical Activity and Cytotoxic-
ity Assessment of Ziziphus mouritiana and Syzygium polyan-
thum.Int. J. pharm.,8(6), 535-541.
Phamhuy, L. A., He, H. and Pham-Huy, C. (2008): Free radicals,
antioxidants in disease and health. Int. J. Biomed. Sci., 4(2):
89-96.
Preeti and Tripathi S. (2014): Ziziphus jujuba: a phytophar-
macological review. Int. J. Res. Develop. Pharm. Life Sci., 3(3):
959-966.
Rates, S. M. K. (2001): Plants as source of drugs. Toxicon., 39:
603-0613.
Sharma, K. R., Kalauni, S., Awale, S. and Pokharel, Y. R. (2015):
In vitro Free Radical Scavenging Activity of Methanol Extracts
of Some Selected Medicinal Plants of Nepal.Austin J. Biotech-
nol. Bioeng.,2: 1-5.
Soliman, Y. H. (2011): Topical Anti-in ammatory and wound
healing activities of herbal gel of Ziziphus nummularia L. (F.
Rhamnaceae) leaf extract. Int. J. Pharm., 7:862–867.
Stahl, E. (1973): Drug analysis by Chromatography and Micros-
copy. Ann Arbor Scienti c Publishers, Inc., Michigan, USA.
Tsuda, T., Horio, F. and Osawa, T. (2002): Cyanidin 3-O--D-
Glucoside Attenuates the Hepatic Ischemia-Reperfusion Injury
through a Decrease in the Neutrophil Chemoattractant Produc-
tion in Rats. J. Nut. Sci. Vitam., 48 (2):134-141.
Wadood, A., Ghufran, M., Jamal, S. B., Naeem, M., Khan,
A., GhaffarR. and Asnad (2013): Phytochemical Analysis of
Medicinal Plants Occurring in Local Area of Mardan. Biochem.
Anal. Biochem. 2:144.
WHO (1996): WHO Guideline for the Assessment of herbal
medicines. WHO Expert Committee on speci cation for phar-
maceutical preparation. Technical Report series no. 863,
Geneva.
Winter, C. A., Risley, E. A. and Nuss, G. W. (1962): Carra-
geenan-induced edema in hind paws of the rat as an assay
for anti-in ammatory drugs. In: Proceedings of the Society
for Experimental Biology and Medicine, New york, N.Y., 111:
544–547.
Wybranowski, T., Ziomkowska, B. and Kruszewski, S. (2013):
Antioxidant properties of
avonoids and honeys stud-
ied by optical spectroscopy methods. Med. Biol. Sci., 27(4):
53-58.
Yusuf, A. Z., Zakir, A., Shemau, Z., Abdullahi, M. and Hal-
ima, S. A. (2014): Phytochemical analysis of the methanol
leaves extract of Paullinia pinnata lin. J. Pharm. Phyto., 6(2):
10-16.